
ijʵ��С����̽��Na2CO3��NaHCO3�����ʣ�����ʵ������ʢ�����ֹ�����Լ�ƿ��ʧ�˱�ǩ�����ǣ������ȶԹ���A��B���м�����ͨ��ʵ���������̽����
��1���ֱ���ȹ���A��B�����ֹ���A���Ȳ�����������ʹ����ʯ��ˮ����ǡ�A���ȷֽ�Ļ�ѧ����ʽΪ_________________________________��
��2����ȡ���ֹ����2 g���ֱ��������С�ձ��У��ٸ���10 mL ����ˮ���������¶ȱ仯�����������ܽ⣬�ָ������£���������Һ�и�����2�η�̪��Һ��
�ٷ���Na2CO3������ȫ�ܽ⣬��NaHCO3������ʣ�࣬�ɴ˵ó�����___________________��
��ͬѧ�������ձ��л��۲쵽�����������У�ʢ��Na2CO3���ձ��г��ֵ�������________________������ĸ��ţ���
A����Һ�¶��½� B����Һ�¶����� C�������̪���dz��ɫ D�������̪��ʺ�ɫ
��3����ͼ��ʾ�������������õ�װ�â�͢��зֱ����ҩƷ���������ڵĹ���ͬʱ�����Թ��С�
�����Թ��о��������壬________������ķ�Ӧ�̶ȸ�Ϊ���ҡ�
�ڷ�Ӧ����������������ͣ��ָ������£�����˵����ȷ����__________��
A��װ�â����������ϴ� B��װ�â����������ϴ�
C������������������������� D�����������������ݹ������
��4��ͬѧ�ǽ����ֹ���ֱ����Ƴ�0.5 mol��L-1����Һ��������·������Է�Ӧ��������Ԥ�⣺
| ʵ�鷽�� | Ԥ������ | Ԥ������ |
| ����1����2 mL Na2CO3��Һ�еμ�1 mL 0.5 mol��L-1CaCl2��Һ | �а�ɫ���� | Na2CO3��Һ�е�CO32-Ũ�Ƚϴ�����CaCl2������Ӧ_________��д���ӷ���ʽ���� |
| ����2����2 mL NaHCO3��Һ�еμ�1 mL 0.5 mol��L-1CaCl2��Һ | �ް�ɫ���� | NaHCO3��Һ�е�CO32-Ũ�Ⱥ�С��������CaCl2��Ӧ�� |
��1��2NaHCO3  Na2CO3 + CO2��+ H2O
Na2CO3 + CO2��+ H2O
��2����ͬ�¶��£�Na2CO3��NaHCO3������ˮ ��B��D
��3���٢� ��A��C
��4��Ca2+ + CO32- = CaCO3�� Ca2+ + 2HCO3- ="==" CaCO3��+ CO2��+ H2O
���������������1�� Na2CO3�ȶ������Ȳ��ֽ⣬��NaHCO3���ȶ������ȷֽ⡣��Ӧ�ķ���ʽΪ2NaHCO3  Na2CO3 + CO2��+ H2O����2������ͬ���¶��£�����ͬ������ˮ�ڼ������������Na2CO3��NaHCO3���������Na2CO3������ȫ�ܽ⣬��NaHCO3������ʣ��˵������ͬ���¶���Na2CO3��NaHCO3�������ܽ���ˮ�С���ʢ��Na2CO3���ձ��г��ֵ������Ǽ�ˮ�ܽ�ų�����ʹ��Һ���¶����ߣ��μӷ�̪��Һ����Һ��Ϊ��ɫ��˵����Һ�Լ��ԡ�������ȷѡ��ΪB D����3�������Թ��о�����������в���������죬���Ԣ�ķ�Ӧ�̶ȸ�Ϊ���ҡ������ķ�ӦΪ��NaHCO3+HCl=NaCl+H2O+ CO2��n(NaHCO3) ��n(HCl)=1:1�� n(NaHCO3)=1/84mol=0.012mol<n(HCl)=0.003mol,���Էų�����������������������㡣Na2CO3+ 2HCl=2NaCl+H2O+ CO2��n(Na2CO3) ��n(HCl)=1:2��n(Na2CO3)=" 1/106mol=" 0.009mol>2n(HCl)�����Էų������������������������㡣��Ϊ̼���������ᷴӦ�ֲ����У���һ������̼�����ƺ��Ȼ��ƣ�û�����壬�ڶ����ŷų����壬������Na2CO3��NaHCO3�ֱ���������������ȣ����Էų���������NaHCO3�ࡣ�����ȷѡ��ΪA C����4��Na2CO3��Һ��CaCl2��Һ��Ӧ�����ӷ���ʽΪ��Ca2+ + CO32- ="=" CaCO3����2 mL NaHCO3��Һ�еμ�1 mL 0.5 mol��L-1CaCl2��Һ��������ɫ���������塣�������£�NaHCO3��Һ��CaCl2��Һ��Ӧ�����ӷ���ʽΪCa2+ + 2HCO3- ="==" CaCO3��+ CO2��+ H2O��
Na2CO3 + CO2��+ H2O����2������ͬ���¶��£�����ͬ������ˮ�ڼ������������Na2CO3��NaHCO3���������Na2CO3������ȫ�ܽ⣬��NaHCO3������ʣ��˵������ͬ���¶���Na2CO3��NaHCO3�������ܽ���ˮ�С���ʢ��Na2CO3���ձ��г��ֵ������Ǽ�ˮ�ܽ�ų�����ʹ��Һ���¶����ߣ��μӷ�̪��Һ����Һ��Ϊ��ɫ��˵����Һ�Լ��ԡ�������ȷѡ��ΪB D����3�������Թ��о�����������в���������죬���Ԣ�ķ�Ӧ�̶ȸ�Ϊ���ҡ������ķ�ӦΪ��NaHCO3+HCl=NaCl+H2O+ CO2��n(NaHCO3) ��n(HCl)=1:1�� n(NaHCO3)=1/84mol=0.012mol<n(HCl)=0.003mol,���Էų�����������������������㡣Na2CO3+ 2HCl=2NaCl+H2O+ CO2��n(Na2CO3) ��n(HCl)=1:2��n(Na2CO3)=" 1/106mol=" 0.009mol>2n(HCl)�����Էų������������������������㡣��Ϊ̼���������ᷴӦ�ֲ����У���һ������̼�����ƺ��Ȼ��ƣ�û�����壬�ڶ����ŷų����壬������Na2CO3��NaHCO3�ֱ���������������ȣ����Էų���������NaHCO3�ࡣ�����ȷѡ��ΪA C����4��Na2CO3��Һ��CaCl2��Һ��Ӧ�����ӷ���ʽΪ��Ca2+ + CO32- ="=" CaCO3����2 mL NaHCO3��Һ�еμ�1 mL 0.5 mol��L-1CaCl2��Һ��������ɫ���������塣�������£�NaHCO3��Һ��CaCl2��Һ��Ӧ�����ӷ���ʽΪCa2+ + 2HCO3- ="==" CaCO3��+ CO2��+ H2O��
���㣺����Na2CO3��NaHCO3�����ʵ�֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��п�����Ҫ�ɷ���̼��п��������������Fe2O3��FeO��CuO�����ʡ���ͼ������п��Ϊԭ����ȡ��������п��һ�ֹ������̣������̻����Եõ����ָ���Ʒ��������ͭ�����졣
�����±����ݣ��ش����⣺
| ���� | ��ʼ����ʱ��pH | ��ȫ����ʱ��pH |
| Fe2�� | 6.3 | 9.7 |
| Fe3�� | 1.5 | 3.2 |
| Zn2�� | 6.2 | 8.0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ڽ������ӵ�ij�ֺ�������ҩƤ�ɴ���ʯ��ˮ�ࡢ���������ƶ��ɡ�
��1��Al��ԭ�ӽṹʾ��ͼΪ____________��Al��NaOH��Һ��Ӧ�����ӷ���ʽΪ____________��
��2��30Si��ԭ�ӵ�������Ϊ________________________��
��3��Al3+��Yn-�ĵ�������ͬ��Y�������Ԫ�ص��⻯���ˮ��Һ�������ԣ�������⻯���зе���͵���____________________________________��
��4�����ӹ����У�ҩƤ�ڸ����²�����������ʹ�����������������壬��������____________��
��5���������������36.0 g������Fe2O3��Al2O3��SiO2������������ϡ���ᣬ����õ�11.0 g���壻��Һ�м������NaOH��Һ������õ�21.4 g���壻���������Al2O3����������Ϊ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��20 mL ijŨ�ȵ�AlCl3��Һ�е���2 mol��L��1��NaOH��Һʱ�����õij������������NaOH��Һ�����֮��Ĺ�ϵ��ͼ��ʾ��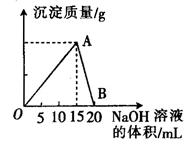
��1��ͼ��A���ʾ��������______________��
��2������������______________g��
��3��B���ʾ��������______________��
��4������AlCl3��Һ�����ʵ���Ũ����______________��
��5�������ó�����Ϊ0.39��ʱ����ȥNaOH��Һ�������_____ mL ��_______ mL��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ����ij����������Cu2O��Al2O3��Fe2O3��SiO2����ȡͭ�IJ����������£�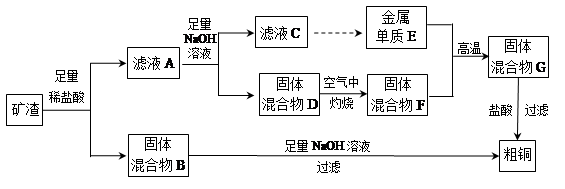
��֪��Cu2O+2H+�TCu+Cu2++H2O
��1����������B������������Һ��Ӧ�Ļ�ѧ����ʽΪ ��
��2����ҺA����Ԫ�صĴ�����ʽֻ��ΪFe2+��������
���漰�����ӷ���ʽΪ ��
��������дCu2O������ķ�Ӧ����������ҺA��Fe2+���Լ�Ϊ �����Լ����ƣ���
��3������ҺC�е���Ԫ���Գ�����ʽ��������ѡ�õ�����Լ�Ϊ __ ������ţ���
| A������������Һ | B��������Һ | C����ˮ | D��������̼ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ���Ѱ۲����ķ�Һ�к��д���FeSO4��H2SO4������Fe2(SO4)3��TiOSO4����������ⷨ������Ѫ�������������������������£�
��֪��TiOSO4������ˮ����ˮ�е���ΪTiO2+��SO42-����ش��������⣺
��1��д��TiOSO4ˮ����������H4TiO4�����ӷ���ʽ ��������м���������м��Ŀ���� ��
��2����ҵ����H4TiO4���Ƶ��Ѱ�TiO2��TiO2ֱ�ӵ�ԭ�������ŷ��������� ��һ�ֽ��Ƚ��ķ����������Ϊ���ڵ�CaCl2��ԭ����ͼ��ʾ�������ĵ缫��ӦΪ_______________��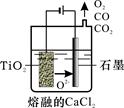
��3������ڵ����ӷ���ʽ�� �����ø���Ʒ��Ҫ ��__________���ѧʽ����
��4������ܵĽᾧ�����б������һ������նȣ�ԭ���� ��
��5�����������ϩ�����в���ϳɣ�
�����ϳ�·�ߵ��ܲ���Ϊ60%��������̼��������Ӧת��Ϊ������������IJ���Ϊ90%��������468 kg�����������壨M��234 g/mol����Ҫ��״���µ���ϩ m3��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ͭ������ϡ���ᣬ��������������Һ���ֽ�һ������ͭƬ���뵽100 mLϡ������������Ļ����Һ�У�ͭƬ��ȫ�ܽ⣨�������ε�ˮ�⼰��Һ����ı仯����
��1��д��ͭ�ܽ������������Һ�����ӷ���ʽ_______________________________________��
��2����ͭ��ȫ�ܽ�ʱ����Һ�е�Fe3����Cu2����H���������ӵ����ʵ���Ũ����ȣ��Ҳ����Һ��pH��1�����ܽ�ͭ��������_________g����Һ�е�c(SO42��)��__________mol/L��
��3����������ͼ��ʾ��װ���з�����1���еķ�Ӧ����X���� ���������������缫��Ӧʽ ��Y���IJ����� ���缫��Ӧʽ ��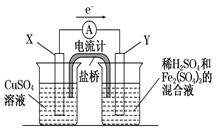
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���к�FeCl2���ʵ��Ȼ�ͭ���壨CuCl2��2H2O����Ϊ��ȡ������CuCl2��2H2O�����Ƚ����Ƴ�ˮ��Һ��Ȼ����ͼ��ʾ��������ᴿ��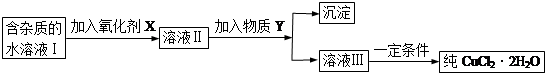

��֪H2O2��KMnO4��NaClO��K2Cr2O7������ǿ�����ԣ�Ҫ����Һ�е�Cu2����Fe2����Fe3������Ϊ�����������Һ��pH�ֱ�Ϊ6.4��6.4��3.7��
��ش��������� [��1��~��2��С������] ��
��1����ʵ�����ʺϵ�������X��__________
A��K2Cr2O7 B��NaClO C��H2O2 D��KMnO4
��2������Y��������___________
A��CuO B��CuCl2 C��Cu��OH��2 D��CuCO3
��3����YΪCu(OH)2��д����ȥFe3�������ӷ���ʽ��
��4��������������Ŀ����_______________________________________��
��5������ܲ���ֱ�������ᾧ�õ�CuCl2��2H2O��__________����ܡ����ܡ������粻�ܣ�Ӧ��β����������ܣ��˿ղ��_______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼʵ��װ��������֤ijЩ���ʵ����ʡ����Թ�A��װ�������Ĺ���NaHCO3��DΪ�̶������ӲֽƬ���Իش��������⣺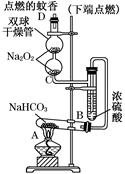
��1����A�Թ��ڷ�����Ӧ�Ļ�ѧ����ʽ��___________________��
��2��Bװ�õ�������__________________________________��
��3����˫�������ڷ�����Ӧ�Ļ�ѧ����ʽΪ_______________��
��4��˫�������ڹ۲쵽��ʵ��������__________________________
____________________________________________________________________��
����ʵ������˵��________________________________________________________��
��5������������ڵ�Na2O2����Na2O����˫�������ڹ۲쵽��ʵ��������_________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com