
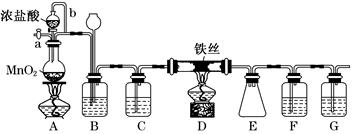
 Mn2����Cl2����2H2O��(2)Ũ�����ŨH2SO4��(3)ƽ��ѹǿ(��ʹŨ����˳������)����ȥCl2�е�HCl���壬�ж�ʵ������е����Ƿ�Ʒ������(4)�ž�ʵ��װ���еĿ���
Mn2����Cl2����2H2O��(2)Ũ�����ŨH2SO4��(3)ƽ��ѹǿ(��ʹŨ����˳������)����ȥCl2�е�HCl���壬�ж�ʵ������е����Ƿ�Ʒ������(4)�ž�ʵ��װ���еĿ���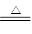 Mn2����Cl2����2H2O��(2)Fװ�����ڷ�ֹ����ˮ�⣬����F��ʢ��Ũ���ᡣ(3)����b��������ƽ��ѹǿ��ʹŨ����˳�����£�װ��B�������dz�ȥCl2�е�HCl���壬ͬʱ�ж�ʵ������е����Ƿ�Ʒ������(4)װ���еĿ�����Ӱ���Ʒ�Ĵ��ȣ����Ե�ȼD���ľƾ���ǰ�����ž�ʵ��װ���еĿ�����(5)�����ж����ڲ�жװ��ǰ���뱻��ȫ���ա�(6)�������Ը��������Һ�����������ӡ�
Mn2����Cl2����2H2O��(2)Fװ�����ڷ�ֹ����ˮ�⣬����F��ʢ��Ũ���ᡣ(3)����b��������ƽ��ѹǿ��ʹŨ����˳�����£�װ��B�������dz�ȥCl2�е�HCl���壬ͬʱ�ж�ʵ������е����Ƿ�Ʒ������(4)װ���еĿ�����Ӱ���Ʒ�Ĵ��ȣ����Ե�ȼD���ľƾ���ǰ�����ž�ʵ��װ���еĿ�����(5)�����ж����ڲ�жװ��ǰ���뱻��ȫ���ա�(6)�������Ը��������Һ�����������ӡ�

 Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д� �㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
�㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
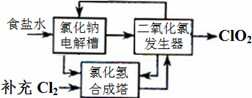
 2ClO2��+2Na2SO4��H2O
2ClO2��+2Na2SO4��H2O H++OH-�� ________________�������ӷ���ʽ��ʾ��.
H++OH-�� ________________�������ӷ���ʽ��ʾ��.�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
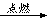 C
C�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
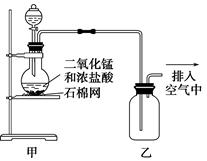
| A����װ��ͼ�����ٴ����������Դ��� |
| B����ƿ�е�MnO2�ɻ���KMnO4 |
| C�����Һ���һʢ�б���ʳ��ˮ���ձ��ɽ���β������ |
| D���ڼ���ƿ�ĵ��ܿڴ���һƬʪ��ĵ���KI��ֽ����֤���Ƿ��������ݳ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��MnO2+4HCl(Ũ) MnCl2+2H2O+Cl2�� MnCl2+2H2O+Cl2�� |
B��2HCl+Ca(OH)2 CaCl2+2H2O CaCl2+2H2O |
C��5Cl2+I2+6H2O 10HCl+2HIO3 10HCl+2HIO3 |
D��2KClO3 2KCl+3O2�� 2KCl+3O2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
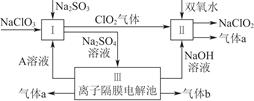
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��


| A����ͼ��:���MnO2����,�����ȫ�������� |
| B����ͼ��:ʪ�����ɫ������ɫ,֤��Cl2��Ư���� |
| C����ͼ��:������ɫ�Ĺ���,֤��Cl2��ǿ������ |
| D����ͼ��:���պ���Ͳ�����������С,˵��������Cl2�����˼ӳɷ�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Ư���Ǵ����Ư��Һ�ǻ���� |
| B��Ư�۵���Ч�ɷ���Ca(ClO)2 |
| C����ҵ�Ͻ�����ͨ�����ʯ��ˮ��ȡƯ�� |
| D��Ư��Һ����Ч�ɷ���Na2O2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
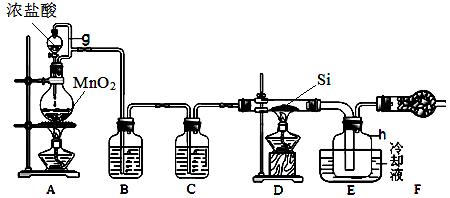
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com