
��16�֣���NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ������������������Һ����μ���1.00mol/L HCl��Һ������HCl��Һ����������ɳ����Ĺ�ϵ��ͼ��ʾ��
�Իش�
��1��A��ij�����Ļ�ѧʽΪ ������ ��
��2��д��A�㵽B�㷢����Ӧ�����ӷ���ʽ ��
��3��ԭ�������MgCl2�������� ��AlCl3�������� ��NaOH�������� ��
��4��C����Һ�����Ϊ mL��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
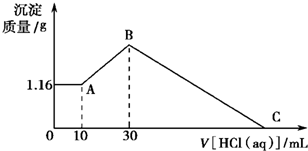 ��NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ�����������õ�����Һ����μ���1mol?L-1 HCl��Һ������HCl��Һ�����V�����ɳ���������m�Ĺ�ϵ��ͼ��ʾ���Իش�?
��NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ�����������õ�����Һ����μ���1mol?L-1 HCl��Һ������HCl��Һ�����V�����ɳ���������m�Ĺ�ϵ��ͼ��ʾ���Իش�?�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
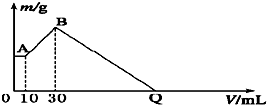 ��NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ����������������Һ����μ���1mol?L-1 HCl��Һ������HCl��Һ����������ɳ�����������ϵ��ͼ��ʾ���Իش�
��NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ����������������Һ����μ���1mol?L-1 HCl��Һ������HCl��Һ����������ɳ�����������ϵ��ͼ��ʾ���Իش��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
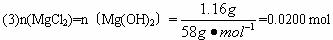
������������⣺
��1��A��ij�����Ļ�ѧʽΪ____________��������______________________��
��2��д��A�㵽B�㷢����Ӧ�����ӷ���ʽ_____________________________��
��3��ԭ�������MgCl2��������________g��AlCl3��������________g��NaOH��������________g��
��4��C��HCl��Һ�����Ϊ________mL��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�����ʡ������һ���¿���ѧ�Ծ� ���ͣ������
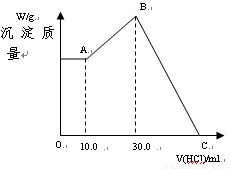
��16�֣���NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ������������������Һ����μ���1.00mol/L HCl��Һ������HCl��Һ����������ɳ����Ĺ�ϵ��ͼ��ʾ��
�Իش�
��1��A��ij�����Ļ�ѧʽΪ ������ ��
��2��д��A�㵽B�㷢����Ӧ�����ӷ���ʽ ��
��3��ԭ�������MgCl2�������� ��AlCl3�������� ��NaOH�������� ��
��4��C����Һ�����Ϊ mL��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com