
�ڣ�Te��Ϊ��A��Ԫ�أ��ǵ�����¼����²��ϵ���Ҫ�ɷ�֮һ����ҵ�Ͽɴӵ�⾫��ͭ������������ȡ�ڡ�
��1����ͭ�к���Cu������Zn��Ag��Au��TeO2�������������⾫��������������Ҫ����TeO2�������������ʼ������������⾫����ͭʱ�������缫��ӦʽΪ ��
��2��TeO2���������������ˮ��������ǿ���ǿ�����������������ȡ�ڵ�һ�ֹ����������£�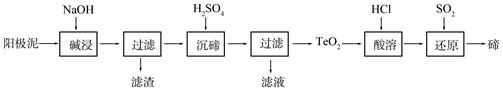
�١������ʱTeO2������Ӧ�Ļ�ѧ����ʽΪ ��
�ڡ����ڡ�ʱ������Һ��pHΪ4.5-5.0������TeO2���������H2SO4��������Һ��ȹ��������ڵij�������ȫ��ԭ���� ����ֹ�ֲ���ȹ���IJ��������� ��
�ۡ����ܡ���SO2ͨ��TeCl4��Һ�н��С���ԭ���õ��ڣ��÷�Ӧ�Ļ�ѧ����ʽ�� ��
��1��Zn-2e-=Zn2+ Cu-2e-=Cu2+ ����4�֣���2�֣�
��2���� TeO2+2NaOH=Na2TeO3+H2O ��3�֣�
��TeO2�����������H2SO4�����ᵼ��TeO2������H2SO4��Ӧ������ʧ����3�֣�
��������H2SO4�������Ͻ��� ��3�֣�
�� TeCl4 + 2SO2 + 4H2O="Te" + 4HCl + 2H2SO4 ��3�֣�
���������������1����⾫����ͭʱ����ͭ�е�Cu������Zn����������������Ӧ��Zn��Cu���ã���ʧ���ӣ����������缫��ӦʽΪZn-2e-=Zn2+ Cu-2e-=Cu2+
��2����TeO2��������������������Ʒ����������������������Ƶķ�Ӧ����ѧ����ʽΪTeO2+2NaOH=Na2TeO3+H2O��
����ΪTeO2�����������H2SO4�����ᵼ��TeO2������H2SO4��Ӧ������ʧ����ֹ�ֲ���ȹ���IJ��������ǻ�������H2SO4�������Ͻ��裻
��SO2��ԭTeCl4ΪTe������������Ϊ���ᣬ��ѧ����ʽΪTeCl4+2SO2+4H2O=Te+4HCl+2H2SO4
���㣺�����ͭ�����ĵ�ⷴӦʽ����д���Ը��������Ļ�ѧ����ʽ���жϼ���д��ʵ��������ж�


 �����������Ů��ͯ������ϵ�д�
�����������Ů��ͯ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��12�֣���ҵ��CH3OH��NaClO3Ϊԭ����������������ȡClO2��ͬʱ����CO2���壬��֪�÷�Ӧ��Ϊ�������У���һ��Ϊ2ClO3- + 2Cl- + 4H+ = 2ClO2��+ Cl2��+ 2H2O��
��1��д���ڶ�����Ӧ�����ӷ���ʽ ��
��2����ҵ����ʱ���ڷ�Ӧ���м�����Cl-���������� ��
��3�������лᷢ������ӦClO3- + Cl- + H+ - Cl2��+ H2O��δ��ƽ��������÷�Ӧ��Ļ��������Cl2���������Ϊ3/73������ʼͶ��ʱCH3OH��NaClO3�����ʵ���֮��Ϊ ��
��д��������̣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(10��)�����ݲ�ͬ������������Ʒ�٢ڢۣ��ס��ҡ�����λͬѧ��ȡһ����Ʒ�ֱ��뺬��0.8molHNO3��ϡ���ᷴӦ,��Ӧ�������������ʣ�࣬������Ļ�ԭ����ֻ��NO���ݴ˻ش��������⣺
��1����ȡ��Ʒ�٣���Ӧ�����Һ����ֻ��Fe3+���ӵ���ʽ���ڣ�����Ʒ�ٵ�����Ϊ g��
��2����ȡ��Ʒ�ڣ���Ӧ�����Һ�мȺ���Fe3+���ֺ���Fe2+���ӣ�����Ʒ�ڵ�����(��Ϊag)��Χ�� ��
��3����ȡ��Ʒ�ۣ������������뵽ϡ�����У������Ͻ��裬��Ӧ��������Һ��Fe3+��Fe2+��NO3�����������е��������ӵ����ʵ����ı仯��������ͼ��ʾ��������߱�ʾ���� ���ʵ����仯�����ߣ������ڴ�ͼ���ٻ������������ӵ����ʵ����ı仯���ߡ�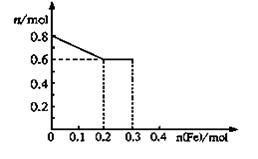
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ⱥ��ʳ��ˮ�ķ�ӦΪ2NaCl+2H2O Cl2��+H2��+2NaOH��
Cl2��+H2��+2NaOH��
27. ����������ʽ�ϱ������ת�Ƶķ������Ŀ��
28. �÷�Ӧ�б�������Ԫ���� ����������֮һΪH2�������Ϸ����ı仯Ϊ ��Ӧ��ѡ�������������ԭ������
29. ������������������������Ҳ������ԭ�������з�Ӧ������ֻ������������ ����д��ţ���
a��Cl2+2KI��2KCl+I2 b��Cl2 + 2NaOH ��NaCl + NaClO + H2O
c��CuCl2 Cl2��+ Cu d��3Cl2 + 2Fe
Cl2��+ Cu d��3Cl2 + 2Fe 2FeCl3
2FeCl3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��15�֣��ں��췢��ʱ���£�N2H4�����������ﳣ��������ƽ�����
��Һ̬�������ȼ��ʱ����Һ̬N2O4��Ϸ���������ԭ��Ӧ����֪ÿ1g�³�ַ�Ӧ��������̬ˮ�ų�����Ϊa KJ����д���÷�Ӧ���Ȼ�ѧ����ʽ ��
��ʵ������N2H4��H2O��NaOH����һ�������ռ�114��116�����ּ�Ϊ��ˮ�¡�
������������в���Ҫ�������� ���������ĸ����
| A���ƾ��� | B����ֱ�������� | C����ƿ | D��ֱ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��X��Y��Z��W���ֶ���������Ԫ�أ�A�ǵؿ��к������Ľ���Ԫ�أ�����������Ԫ����B��ԭ�Ӱ뾶���X��Y��Z��WԪ�������ڱ��е����λ������ͼ��ʾ������ZԪ��ԭ�������������ǵ��Ӳ�����2������ش�����
���⣺ 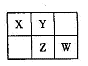
��1��W����������ﻯѧʽ�� ��Z��ԭ�ӽṹʾ��ͼΪ ��
��2��A��B��������������Ӧ��ˮ���ﷴӦ�Ļ�ѧ����ʽΪ ��
��3��AW3�����ھ�ˮ����ԭ���� �����������ӷ���ʽ��ʾ��
��4����ҵ�ϳ�X�ļ���̬�⻯���Ƿ��ȷ�Ӧ�����д�ʩ�м������
��Ӧ���ʣ��������ԭ��ת���ʵ��� ��
a�������¶�
b���������
c����X�ļ���̬�⻯�Pʱ����
d������Ӧ��ϵ��ѹǿ
��5����״���£�2.24L X�ļ���̬�⻯�ﱻ200 mL l mol L��1X������������Ӧ��ˮ������Һ���պ�������Һ������Ũ�ȴӴ�С��˳���ǣ������ӷ��ű�ʾ�� ��
��6��WY2��ɱ��������ͬʱ���ɽ��綾�軯�����������������ȥ��д����WY2���е�9.9�棩������ȥCN�������ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һ��ǿ������������������������ȡ�
(1)O3��KI��Һ��Ӧ���ɵ����ֵ�����________��________(�����ʽ)��
(2)O3��ˮ���ֽ⣬һ�������£�O3��Ũ�ȼ���һ�������ʱ��(t)���±���ʾ����֪��O3����ʼŨ��Ϊ0.0216 mol/L��
��pH�����ܼ���O3�ֽ⣬������O3�ֽ�������õ���________��
����30�桢pH��4.0�����£�O3�ķֽ�����Ϊ________mol/(L��min)��
�۾ݱ��еĵݱ���ɣ��Ʋ�O3�����������·ֽ��������������˳��Ϊ________(����ĸ����)��
a��40�桢pH��3.0������ b��10�桢pH��4.0
c��30�桢pH��7.0
(3)O3���ɳ���������(ԭ����ͼ)���ϡ�����Ƶá�
��ͼ������Ϊ________(�A����B��)����缫��ӦʽΪ_______________________��
����C��ͨ��O2����A���ĵ缫��ӦʽΪ________��
����C����ͨ��O2��D��E���ֱ��ռ���x L��y L����(��״��)����E���ռ���������O3��ռ���������Ϊ________(����O3�ķֽ�)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ڳ����ķǽ������仯�����У�
��1�������ĵ����Խ��ڵ���;�Ե��֮�䣬�����õ� ���ϣ�������ˮ��Һ�׳�Ϊ �����Ʊ��轺��ľ�ķ������ԭ�ϣ��մɡ� ��ˮ��ȹ����β�Ʒ��ʹ�����������ǽ������ϡ�
��2����Ԫ������Ҫ�ġ�����Ԫ�ء���������ͨ��NaOH��Һ�п��Եõ�Ư��Һ����Ӧ�����ӷ���ʽ�� ��Ư��Һ��Ч�ɷֵĻ�ѧʽΪ ��
��3��ˮ�dz����ķǽ���Ԫ�ػ�����밴Ҫ��д������ˮ�μӵķ�Ӧ�Ļ�ѧ����ʽ��
��ˮֻ����������
��NO2����ͨ��ˮ�У�
��ˮ����������������ԭ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������Ǵ�����Ⱦ��֮һ��������������ķ����ж��֡�
��1�����ü������ԭ���������֪��
CH4 (g)��4NO2(g)��4NO(g)��CO2(g)��2H2O(g) ��H ����574 kJ/mol
CH4(g)��4NO(g) �� 2N2(g)��CO2(g)��2H2O(g) ��H ����1160 kJ/mol
��CH4 ��NO2 ��ԭΪN2 ���Ȼ�ѧ����ʽΪ ��
��2������NH3����ԭ�������SCR����)���ü�����ĿǰӦ����㷺���������������ѳ������� ��Ӧ�Ļ�ѧ����ʽΪ��2NH3(g)��NO(g)��NO2(g)  2N2(g)��3H2O(g)����H < 0
2N2(g)��3H2O(g)����H < 0
Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�� ��д��1�����ɣ���
��3������ClO2�������������ת���������£�
NO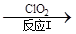 NO2
NO2 N2
N2
��֪��Ӧ��Ļ�ѧ����ʽΪ2NO+ ClO2 + H2O �� NO2 + HNO3 + HCl����Ӧ��Ļ�ѧ����ʽ�� ��������11.2 L N2����״������������ClO2 g ��
��4������CO����ԭ��������Ҳ���Դﵽ������Ⱦ��Ŀ�ġ�
��֪����һ��ʱ�������������ı���������ѧ��Ӧ���ʡ���ͼ�Ƿ�Ӧ2NO(g) + 2CO(g) 2CO2(g)+ N2(g) ��NO��Ũ�����¶�(T)�����������������(S)��ʱ��(t)�ı仯���ߡ��ݴ��жϸ÷�Ӧ�ġ�H 0 (�����������������ȷ����)�����������S1 S2 (�����������������ȷ����)��
2CO2(g)+ N2(g) ��NO��Ũ�����¶�(T)�����������������(S)��ʱ��(t)�ı仯���ߡ��ݴ��жϸ÷�Ӧ�ġ�H 0 (�����������������ȷ����)�����������S1 S2 (�����������������ȷ����)��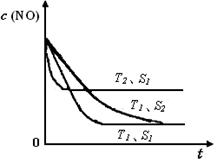
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com