
��10�֣�X��Y��Z��W���������������ת����ϵ������X��W���ʣ�Y��ZΪ������,δ�г���Ӧ��������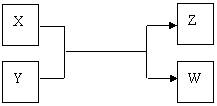
����Z�������г��õĵ�ζƷ��W��������Һ��������
��1�������£�X����ɫ�� ��
��2����ҵ��Z�ж�����;���û�ѧ����ʽ��ʾZ��һ����; ��
��3�����������õ�Z�����˵���أ�����X��Y��Һ��Ӧʱ���Եõ�һ�ֵ����Σ��˷�Ӧ�����ӷ���ʽ�� ��
����X�ǹ�ҵ���������Ľ������ʣ�Z��һ�־��д��Եĺ�ɫ���壬��
��1�� X��Y��Ӧ�Ļ�ѧ����ʽ�� ��
X��Y��Ӧ�Ļ�ѧ����ʽ�� ��
��2����������װ��ֻ����Z + W=" X" + Y��Ӧ���г�װ��δ��������
����ɴ�ʵ���жಽ���������������ǣ�
��a����ȼ�ƾ� �� b���μ����� c���������崿��
�� b���μ����� c���������崿��
�������������Ⱥ�˳���� ������ĸ����
������װ����Ҫ�Ľ��������ǣ������������� ��
��3���� 3.48 gZ����50 mL4 mol/L��ϡHNO3�г�ַ�Ӧ������112 mL��NO����״��������Ӧ�����Һ�еμ�NaOH��Һ�ܲ�������������������࣬������Ҫ����2 mol/L��NaOH��Һ mL (��ȷ��0.1)��
(10��)
��
��1������ɫ (1��)
��2��2NaCl +2 H2O H2��+ Cl2�� + 2 NaOH (2��)
H2��+ Cl2�� + 2 NaOH (2��)
��3��3Cl2 + I��+ 3 H2O ="=" 6 Cl- + IO3��+ 6 H+  (2��)
(2��)
��
��1��3 Fe + 4 H2O��g�� Fe3O4 + 4 H2 (2��)
Fe3O4 + 4 H2 (2��)
��2����b c a (1��)
���ڼס���װ���м�����һ��װ��ˮ��ϴ ��ƿ (1��)
��ƿ (1��)
��3��97.5 (1��) (10��)
��
��1������ɫ (1��)
��2��2NaCl +2 H2O H2��+ Cl2�� + 2 NaOH (2��)
H2��+ Cl2�� + 2 NaOH (2��)
��3��3Cl2 + I��+ 3 H2O ="=" 6 Cl- + IO3��+ 6 H+  (2��)
(2��)
��
��1��3 Fe + 4 H2O��g�� Fe3O4 + 4 H2 (2��)
Fe3O4 + 4 H2 (2��)
��2����b c a (1��)
���ڼס���װ���м�����һ��װ��ˮ��ϴ ��ƿ (1��)
��ƿ (1��)
��3��97.5 (1��)
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 X��Y��Z��W���ֻ�������ɳ���Ԫ����ɣ�����X��������Ԫ�أ�Y��Z��������Ԫ�أ�X��Y��Z����ɫ��Ӧ��Ϊ��ɫ��WΪ��ɫ��ζ���壮�����ֻ����������ͼת����ϵ�����ַ�Ӧ����P��Ӧ��������ȥ������ش�
X��Y��Z��W���ֻ�������ɳ���Ԫ����ɣ�����X��������Ԫ�أ�Y��Z��������Ԫ�أ�X��Y��Z����ɫ��Ӧ��Ϊ��ɫ��WΪ��ɫ��ζ���壮�����ֻ����������ͼת����ϵ�����ַ�Ӧ����P��Ӧ��������ȥ������ش�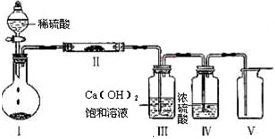
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com