
ijУ��ѧ��ȤС��Ϊ��̽����������������������ͭ���ʣ���ijŨ�����ᷴӦ�����������̽�����
̽��һ
��1����������Ͷ��ijŨ�������У�ijͬѧ�۲��ʵ������ʱ���֣���Ӧ����һ��ʱ���Ӧ��ʼ�ӿ졣�������Ӧ�ӿ�Ŀ���ԭ���_____________��_____________��
��2������ȡ������10 g����ijŨ�������У���ַ�Ӧ��õ���ҺX���ռ�������Y��Ϊ��̽����ҺX����Ԫ�صļ�̬����ͬѧ�������ʵ�飺
��ҩƷ��������0.1mol/L KSCN��Һ��0.1mol/L KI��Һ��0.2mol/L���Ը��������Һ����ˮ�ȣ��Թܺ͵ιܡ�
������Ƽ�ʵ�飬̽�����������Ƿ���ȷ����д����ʵ�鱨�棺
| ʵ�鲽�� | ���� | ���� | ���ӷ���ʽ |
| ��һ�� | ȡ2��3 mL��Һװ���Թܣ����Թ��м��뼸��KSCN��Һ�� | | |
| �ڶ��� | | ����Һ��ɫ��ȥ������Һ����Fe2+���������Ա仯����Fe3+�� | |

��1���ٷ�Ӧ���ȣ�һ��ʱ�����Һ�¶����ߣ���Ӧ���ʼӿ죻
���������к�����ͭ����Ӧʱ�γ�Fe-Cu ��ԭ��أ��ӿ췴Ӧ����
��2��
��3����֤NO2�Ĵ��ڲ���ȥNO2���� ���� ��������� ���ܵ����ӷ���ʽ ��1�� ����Һ���ɫ������Һ����Fe3+������������Fe3+ Fe3++3SCN��=Fe(SCN)3 ��2�� ��ȡ������Һװ���Թܣ����Թ���μӼ������Ը��������Һ 5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O
��4������ȷ��������NO2��ˮ��ӦҲ������NO���ʲ���ȷ������Y�к�NO��
��5��1��3
���������������1���ٽ�������Ͷ��ijŨ�������У����ַ�Ӧ����һ��ʱ���Ӧ��ʼ�ӿ죬�ɴ�2���������һ���¶�ԭ��������γ�ԭ��أ��ʴ�Ϊ����Ӧ���ȣ�һ��ʱ�����Һ�¶����ߣ���Ӧ���ʼӿ죻���������к�����ͭ����Ӧʱ�γ�Fe-Cu��ԭ��أ��ӿ췴Ӧ���ʣ�
��2������ѡ������������������������ӣ���������������ʱ����Һ���ɫ���������Ӿ��л�ԭ�ԣ����Ա������������Ϊ���������ӣ����¸��������ɫ���ʴ�Ϊ������ ���� ��������� ���ܵ����ӷ���ʽ ��1�� ����Һ���ɫ������Һ����Fe3+������������Fe3+ Fe3++3SCN-=Fe��SCN��3 ��2�� ��ȡ������Һװ���Թܣ����Թ���μӼ������Ը��������Һ 5Fe2++MnO4-+8H+=5Fe3++Mn2++4H2O
��3��װ������NO2��Na2S��Һ��Ӧ���ɿ��������������ᣬ��������Na2S��Һ��Ӧ����S�������ʿ���֤NO2�Ĵ��ڲ���ȥNO2���ʴ�Ϊ����֤NO2�Ĵ��ڲ���ȥNO2��
��4������NO2��ˮ��ӦҲ������NO���ʲ���ȷ������Y�к�NO���������Һͱ�װ�ü��һ������NO2�Ƿ����װ�ã��ʴ�Ϊ������ȷ��������NO2��ˮ��ӦҲ������NO���ʲ���ȷ������Y�к�NO��
��5����5NO2-��2 MnO4-����2n��NO2-��=7.5��10-3 mol����NO��NO2�����ʵ����ֱ�Ϊx��y mol�����У�x+y=10��10-3 mol��2x+1/2��y-x��=7.5��10-3����ã�x=2.5��10-3 mol��y=7.5��10-3 mol������Y��NO��NO2�������Ϊ1��3���ʴ�Ϊ��1��3��
���㣺̽����ѧ��Ӧ�������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����������һ�ֳ���ʳƷ���Ӽ���Ϊ���ijʳƷ���������κ���(ͨ����1 kg��Ʒ�к�SO2��������)��ij�о�С���������������ʵ�����̣�
(1)����A����Ҫ�ɷ���________��Ϊ��ֹ���ʱ�������У�����������ƿ�м���________��ͨ��N2��Ŀ����________��
(2)д�������ڢٲ���Ӧ�����ӷ���ʽ��________________________��
(3)�����ڢڲ��ζ�ǰ���ζ�������NaOH����Һ��ϴ�������������__________________________________________________________��
(4)�����������ϡ���ᴦ����Ʒ�����ҷ���ʵ��ⶨ�Ľ��________(�ƫ�ߡ���ƫ�͡�����Ӱ�족)��
(5)��ȡ��Ʒw g�����ҷ����������0.010 00 mol��L��1I2��ҺV mL����1 kg��Ʒ�к�SO2��������________g(�ú�w��V�Ĵ���ʽ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��
��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2?3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 g NaOH��������ˮ������Һ�����Ϊ1L��
��1��160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ������ ��������Ҫ�������Һ����������������Ҫ��һ�������������������� ��������������������˵������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ� b��ϡ��ClO2�Է�ֹ��ը��
c����NaClO3������ClO2
��3���������ڷ�Ӧ�����ӷ���ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH�Ƿ�����ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6������Һ�еõ�NaClO2?3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������Ũ�� c������ d������ϴ�� e����ȴ�ᾧ
Ҫ�õ�������NaClO2?3H2O���������еIJ����������� ���� ����������ƣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��1��ij��ȤС������������װ�úͷ����ⶨ������SO2����������Ϊ���еIJ����� ������ţ�
| ��� | A | B | C |
| װ�� |  |  |  |
| ԭ�� | ͨ��V���������ⶨ���ɳ��������� | ��KMnO4��Һ�պ���ɫʱ���ⶨͨ����������V | ͨ��V���������ⶨU�����ӵ����� |

| ʵ�鲽�� | Ԥ������ͽ��� |
| | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ʾ��þ�뱥��̼��������Һ��Ӧ������������Ͱ�ɫ�����ijͬѧͨ������ʵ��̽����Ӧԭ������֤���
ʵ��I����ɰֽ��ȥþ����������Ĥ���������ʢ�������з�̪�ı���̼��������Һ���ձ��У�Ѹ�ٷ�Ӧ�������������ݺͰ�ɫ�������Һ��dz��ɫ���
��1����ͬѧ�Է�Ӧ�в����İ�ɫ�������������²²⣺
�²�1����ɫ���������Ϊ����������
�²�2����ɫ���������ΪMgCO3
�²�3����ɫ���������Ϊ��ʽ̼��þ[yMg(OH)2?xMgCO3]
��2��Ϊ��ȷ������������¶���ʵ�飺
| ʵ����� | ʵ �� | ʵ������ | �� �� |
| ʵ��� | ��ʵ��I���ռ����������ȼ | ����ȼ�գ� ����ʵ���ɫ | ����ɷ�Ϊ �� �� |
| ʵ��� | ��ʵ��I�еİ�ɫ�������˳���ϴ�ӣ�ȡ������������ �� | �� | ��ɫ�������к���MgCO3 |
| ʵ��� | ȡʵ����е���Һ�������м����� ���� ���� ϡ��Һ | ������ɫ��������Һ��ɫ��dz | ��Һ�д���CO32- ���� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ʵ�������Ҵ���Ũ���Ṳ����ȡ��ϩ�������¶ȹ��߶����������Ķ������������������ʵ����ȷ�������������������ϩ�Ͷ�������
��1����װ�ÿ�ʢ�ŵ��Լ��ǣ�I ��II ��III ��IV ���������й��Լ����������ո��ڣ���
| A��Ʒ����Һ | B������������Һ | C��Ũ���� | D�����Ը��������Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijС��ͬѧ��һ��Ũ��NaHCO3��Һ���뵽CuSO4��Һ�з��������˳�������ͬѧ��Ϊ������CuCO3����ͬѧ��Ϊ������CuCO3��Cu(OH)2�Ļ������ʵ��ⶨ������CuCO3������������
��1�����ռ�ͬѧ�Ĺ۵㣬������Ӧ�����ӷ���ʽΪ ��
��2����ͬѧ��Ϊ��Cu(OH)2���ɵ����������� (�����ӷ���ʽ��ʾ)��
��3����ͬѧ������ͼ��ʾװ�ý��вⶨ
����װ����������μ��������
���о����������ǰ���뽫��������Һ�з��벢�����������������Ϊ ��ϴ�ӣ����
��װ��E�м�ʯ�ҵ������� ��
��ʵ������������²������裺
a���ر�K1��K3����K2��K4����ַ�Ӧ
b����K1��K4���ر�K2��K3��ͨ���������
c����K1��K3���ر�K2��K4��ͨ�����������
��ȷ��˳���� (��ѡ����ţ���ͬ)��
��δ���в��� ��ʹ�������ƫ�ͣ�
����������Ʒ������Ϊm g��װ��D������������n g���������CuCO3����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
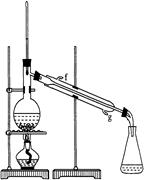
ij��ѧС���������������������װ�ã���ͼ�����Ի������Ʊ�����ϩ��
��֪��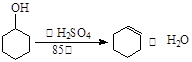
| | �ܶ� ��g/cm3�� | �۵� ���棩 | �е� ���棩 | �ܽ��� |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | ��103 | 83 | ������ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�Ϸ���ijУ��ѧ��ȤС��Ϊ̽������Ũ���ᷴӦ�����������ͼ��ʾװ�ý���ʵ�顣
(1)ʵ������У��۲쵽B�е�ʵ�������� ��
һ��ʱ��۲쵽��C��������������ð���������ݵijɷ��� ��
(2)�á��ɳ鶯����˿�����桰ֱ��Ͷ����Ƭ�����ŵ��� ����Ӧ��������Ҫ�������Ϳ�ʹװ���в���������ȫ�����գ�Ӧ����ȡ�IJ����� ��
(3)��Ӧһ��ʱ������Ƕ�A����Һ�Ľ��������ӽ�����̽����
��������裺
����1����Һ��ֻ����Fe2+��
����2�� ��
����3����Һ�д���Fe2+��Fe3+��
���������ʵ����֤��������1��д��ʵ�������ʵ�������ۡ�
��ѡ�Լ���KMnO4��Һ��NaOH��Һ������һKI��Һ��KSCN��Һ
| ʵ��������� | ʵ������ | ���� |
| | | |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com