
��8�֣�Li-SOC12��ؿ����������������õ�صĵ缫���Ϸֱ�Ϊ﮺�̼�����Һ��LiAlCl4��SOCl2.��ص��ܷ�Ӧ�ɱ�ʾΪ��4Li+2SOC12  4LiCl +S+SO2��
4LiCl +S+SO2��
��ش��������⣺
(1)��صĸ�������Ϊ_________�������ĵ缫��ӦΪ____________________��
(2) SOCl2�ӷ���ʵ�����г���NaOH��Һ����SOC12�����������ˮ�ε�SOCl2�У���Ӧ�Ļ�ѧ����ʽΪ__________________________________________��
(3)�ô����ص�⺬��0.1 mol/LCuSO4��0.1 mol/LNaCl�Ļ����Һ100 mL�������·��ת����0.02 mole-���ҵ��صĵ缫��Ϊ���Ե缫�����������������ڱ�״���µ������______L�����������Һ��ˮϡ����1L����ʱ��Һ��pH=______________��
��1��Li Li-e-=Li+��ÿ��1�֣�
��2��SOCl2+H2O=2HCl��+SO2��(2��)
��3��0.168 2��ÿ��2�֣�
���������������1��������ص��ܷ�Ӧ֪���÷�Ӧ��Li�Ļ��ϼ���0�����ߵ�+1�ۣ�����������Ӧ������صĸ������缫��ӦʽΪLi-e-=Li+����2��SOCl2��ˮ��Ӧ����SO2��HCl����Ӧ�ķ���ʽΪSOCl2+H2O=2HCl��+SO2������3��0.1 mol/LCuSO4��0.1 mol/LNaCl�Ļ����Һ100 mL����0.01molCuSO4��0.01molNaCl������·��ת����0.02 mole-ʱ��������ӵķŵ�˳��֪�������缫��Ӧ����Ϊ��2Cl--2e-=Cl2����4OH--4e-=2H2O+O2�������õ����غ���������ݼ������������0.005mol����������0.0025mol���������������ɵ������ڱ�״���µ����=��0.005mol+0.0025mol����22.4L/mol=0.168L���������������Ӽ���0.01mol������Һ������������0.01mol�����������Һ��ˮϡ����1L����Һ��������Ũ��Ϊ0.01mol/L����ҺPH=2��
���㣺����ԭ���ԭ���͵��ԭ����Ӧ�á�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ZnƬ��CuƬ��ϡ������ɵ�ԭ���װ���У�����һ��ʱ�������˵����ȷ����
| A��пƬ��������ͭƬ�������ݲ��� | B�����������Ǵ�пƬ����ͭƬ |
| C����Һ����������ʵ���Ũ�ȼ�С | D���������Һ��pH���ֲ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����10�֣����������ӽ���Ĥ�������ӽ���Ĥ��ʯī�缫�������ȼҵ�е�Ĥ����ԭ�����ش��������⡣
��1��д���ȼҵ�е�ⱥ��ʳ��ˮ�����ӷ���ʽ ��
��2�������ý���Ĥ������������ͼ��ܣ����һ�������������Һ������������Һ��������Һ��װ�ã�����������ʵĻ�ѧʽ��
A_________��C_________�� E_________��ĤbΪ ���ӽ���Ĥ�����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ�ס����ǵ绯ѧʵ��װ�á�
��1�����ס������ձ��о�ʢ��NaCl��Һ����
�ټ���ʯī���ϵĵ缫��ӦʽΪ ��
�������ܷ�Ӧ�����ӷ���ʽΪ ��
�۽�ʪ��ĵ���KI��ֽ�������ձ��Ϸ���������ֽ�ȱ�������ɫ��������Ϊ������Cl2���������ɵ�I2 ������Ӧ��Cl2��I2�����ʵ���֮��Ϊ5��1���� ���������ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ ��
��2�����������ձ��о�ʢ������CuSO4��Һ����
�ټ��������ϵĵ缫��ӦʽΪ ��
�������ʼʱ����ʢ��200mLpH=5��CuSO4��Һ(25��)��һ��ʱ�����Һ��pH��Ϊ1����Ҫʹ��Һ�ָ������ǰ��״̬��������Һ�м���������� (д��ѧʽ)��������Ϊ g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(12��)����ѧ������ѧ�뼼����
��ˮռ�����ܴ�ˮ����97��2�������Ѻ�ˮ�����ͻ�����������������ȿ��Խ����ˮ��Դȱ�������⣬�ֿ��Գ�����ú�����Դ��
(1)�༶��������Ŀǰ����ˮ����������Ҫ�������÷�����һ�������½���ˮ�������������������ȴ���øߴ��ȵ�ˮ���ɴ˿��ж϶༶�������� (������仯�� ��ѧ�仯��)��
(2)���ú�ˮɹ�ε�ԭ���� ������ʳ�ξ�����ĸҺ�к���KCl��MgCl2���������롢�ᴿ������ ��
(3)���ȼҵ�����õ�ⱥ��ʳ��ˮ�Ƶ���Ҫ������Ʒ�����ȼҵ�У���Ĥ�����(��ͼ����ʾ)���������ӽ���Ĥ���(��ͼ����ʾ)����ȡ����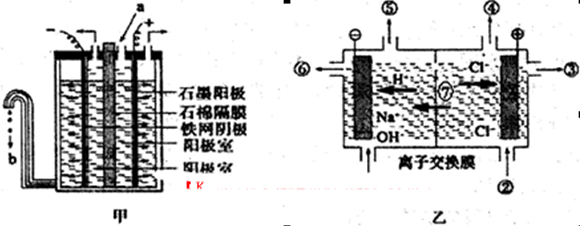
��д�����缫�ķ�Ӧʽ������ ������ ��
��ʯ��Ĥ�������� �����ӽ���Ĥ�����Тޡ��߷ֱ��� �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�Ϊ�������÷Ϸ�����������V2O5��VOSO4�������Բ�������������Ա����������һ�����ӽ��������շ����¹��գ���Ҫ�������£�
���ֺ���������ˮ�е��ܽ������£�
| ���� | VOSO4 | V2O5 | NH4VO3 | ��VO2��2SO4 |
| �ܽ��� | ���� | ���� | ���� | ���� |

 VO2+��H2O��V3+����س��ʱ�����ĵ缫��ӦʽΪ ��
VO2+��H2O��V3+����س��ʱ�����ĵ缫��ӦʽΪ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��10�֣�����ȼ�ϵ�ص�ʵ��װ����ͼ��ʾ���ش������йظ�ʵ������⡣
��1��Ҫʹ��������ܷ�����ʵ��ʱ�IJ���˳���ǣ���_______�����һ��ʱ���__________��
��2����ʵ���ȫ��������������Ҫת����ʽ�� ��
��3��д���������ֲ���ʱ�ĵ缫��Ӧʽ����ע���缫���ƣ�
�ٰ��¿���S1���Ͽ�����S2��һ��ʱ��� C1Ϊ �����缫��Ӧ�� ��
�ڶϿ�����S1��Ѹ�ٰ��¿���S2������ʾ����ʱC1������Һ�Լ��ԣ�C2������Һ�����ԣ�C2Ϊ______���� �缫��Ӧ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��9�֣���������װ�������ԭ��ص���_____________ ������ţ�
��������ͼ����ʾ������ԭ��أ���ش��������⣨�����ǣ����ûش𣩡�
��1������������_______________________
��2�������ĵ缫��ӦʽΪ________________________
��3����ع���ʱ�ܷ�Ӧ�����ӷ���ʽ��____________________________
��4�������ӵ��ƶ�����___________________________________
��5�������е��ӵ�����________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������������������ʴ��ÿ����ʴ����ʧ�ĸ���ռ���������������ķ�֮һ����ش�����ڸ�ʴ�����������е��й����⡣
(1)�����ĸ�װ�ÿɷ�ֹ��������ʴ________________��

(2)��ʵ�������У����������ı����ͭ��ֹ������ʴ��װ��ʾ��ͼ��ͼ����ش�
��A�缫��Ӧ�Ľ�����________(дԪ������)��B�缫�ĵ缫��Ӧʽ��________________________��
�ڶƲ������ͭ���ȶ�п�������ױ���ʴ�����Ҫ˵��ԭ��
____________________________________________________
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com