
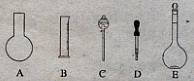
| A����ȴ | B������ | C��ϴ�� | D������ E���ܽ� F��ҡ�ȡ� G����Һ |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
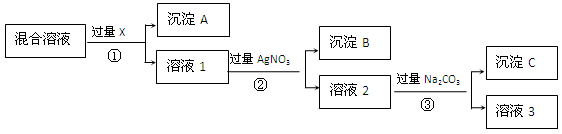
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 __________________________________________________
__________________________________________________ ______________________��
______________________�� 1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______ __���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ______ __mol��L��1��
1)����Һ����ȡ20.00 mL FeSO4��Һ������ƿ�У���0.10 mol��L��1������KMnO4��Һ�����յ㣬��ȥKMnO4��Һ20.00 mL������������MnԪ��ȫ���ʣ�2�ۣ��ζ���Ӧ�����ӷ���ʽΪ______ __���ݴ˿ɲ��FeSO4��Һ�����ʵ���Ũ��Ϊ______ __mol��L��1���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
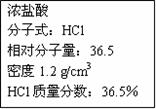  |
| Ӧ��ȡŨ�������/mL | Ӧѡ������ƿ�Ĺ��/mL |
| | |
 �������� ��ˮ��ʹ��Һ����ǡ����̶����С�
�������� ��ˮ��ʹ��Һ����ǡ����̶����С� �ȡ�
�ȡ� ��ת�Ƶķ������Ŀ��
��ת�Ƶķ������Ŀ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ѧ��Ϊ����˫ˮ�ⷴӦ���䷴Ӧ����ʽΪ2Fe3++3SO32-+6H2O=2Fe(OH)3(����)+3H2SO3��
ѧ��Ϊ����˫ˮ�ⷴӦ���䷴Ӧ����ʽΪ2Fe3++3SO32-+6H2O=2Fe(OH)3(����)+3H2SO3�� Ӧѡ�õ��Լ��� ��
Ӧѡ�õ��Լ��� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������������ʳ�üӵ���(��KIO3)�к��е� |
| B���õ�Ƽ����������Ƿ��в������� |
| C���ᴿ�����ʿ����ڵ�������Һ�мӱ���CuSO4��Һ��������������Ȼ��ѳ�����������ˮ�� |
| D��������Һ�м����������ˮ�⣬��ȴ����������Һ��������Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
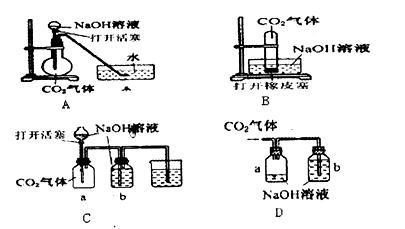
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ȥN2�е�����O2��ͨ�����ȵ�CuO��ĩ���ռ����� |
| B����ȥCO2�е�����HCl��ͨ��Na2CO3��Һ���ռ����� |
C����ȥNa Cl��Һ������CaCl2����������Na2CO3������ Cl��Һ������CaCl2����������Na2CO3������ |
| D����ȥKCl��Һ������MgCl2����������NaOH��Һ������ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com