
��֪��2CO(g)+O2(g)=2CO2(g) ��H=-566 kJ/mol

Na2O2(s)+CO2(g)=Na2CO3(s)+ ![]() ��H=-226 kJ/mol
��H=-226 kJ/mol
���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ���ǣ� ��
A��CO��ȼ����Ϊ283 kJ
B����ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������
C��2Na2O2(s)+2CO2(s)=2Na2CO3(s)+O2(g) ��H��-452 kJ/mo
D��CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪��2CO(g)+O2(g)=2CO2(g)�������� �� ������H=-566 kJ/mol
![]() Na2O2(s)+CO2(g)=Na2CO3(s)+
Na2O2(s)+CO2(g)=Na2CO3(s)+ ![]() ������������������ ��H=-226 kJ/mol
������������������ ��H=-226 kJ/mol
![]() ���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ����
���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ����

![]() A.CO��ȼ����Ϊ283 kJ
A.CO��ȼ����Ϊ283 kJ
![]() B.��ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
B.��ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
![]() C.2Na2O2(s)+2CO2(s)=2Na2CO3(s)+O2(g)������ ��H��-452 kJ/mol
C.2Na2O2(s)+2CO2(s)=2Na2CO3(s)+O2(g)������ ��H��-452 kJ/mol
![]() D.CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023
D.CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪��2CO(g)+O2(g)=2CO2(g) ��H=-566 kJ/mol

![]() Na2O2(s)+CO2(g)=Na2CO3(s)+
Na2O2(s)+CO2(g)=Na2CO3(s)+ ![]() ��H=-226 kJ/mol
��H=-226 kJ/mol
![]() ���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ���ǣ� ��
���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ���ǣ� ��
![]() A��CO��ȼ����Ϊ283 kJ
A��CO��ȼ����Ϊ283 kJ
![]() B����ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
B����ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
![]() C��2Na2O2(s)+2CO2(s)=2Na2CO3(s)+O2(g) ��H��-452 kJ/mol
C��2Na2O2(s)+2CO2(s)=2Na2CO3(s)+O2(g) ��H��-452 kJ/mol
![]() D��CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023
D��CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꼪��ʡ������һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��֪��2CO(g)��O2(g)===2CO2(g) ��H����566 kJ/mol
Na2O2(s)��CO2(g)===Na2CO3(s)��1/2O2(g) ��H����226 kJ/mol
���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ����
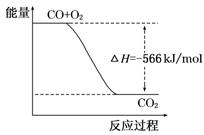
A��CO��ȼ����Ϊ283 kJ
B����ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
C��2Na2O2(s)��2CO2(s)===2Na2CO2(s)��O2(g)����H>��452 kJ/mol
D��CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ6.02��1023
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�갲����ѧ�߶�������ϰ��ѧ����һ�� ���ͣ�ѡ����
��֪��2CO(g)+O2(g)=2CO2(g) ��H=-566 kJ/mol; Na2O2(s)+CO(g)=Na2CO3(s) ��H=-226 kJ/mol ���������Ȼ�ѧ����ʽ�жϣ�����˵����ȷ����

A.CO��ȼ����Ϊ283 kJ
B.��ͼ�ɱ�ʾ��CO����CO2�ķ�Ӧ���̺�������ϵ
C.2Na2O2(s)+2CO(s)=2Na2CO3(s) ��H��-452 kJ/mol
D.CO(g)��Na2O2(s)��Ӧ�ų�509 kJ����ʱ������ת����Ϊ 6.02��1023
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com