
��ͼ�У�A�������ķ���װ�ã�B��C�Ǿ��������װ�ã�D��װ��˿������Ӧ��E�ĵײ�����ɫ����ۼ���F�����ն��������װ�á�
��1������װ������һ��������ָ���� ��������ĸ��ʾ����
��2��ͨ��B��Ϊ�˳�ȥ ����B��Ӧ���� ��ͨ��C��Ϊ�˳�ȥ ��
��3��д��D��F�з�����Ӧ�Ļ�ѧ����ʽ �� ��
��4�����A�в�������3.36L����״����������㣺
��д��A�з�����Ӧ�Ļ�ѧ����ʽ�������A�еĵ���ת����� ��
������MnO2�����ʵ��� ���۱�������HCl�����ʵ��� ��
��1��B
��2��HCl ����ʳ��ˮ ˮ����(ˮ) ����1�֣�
��3��2Fe+3Cl2 2FeCl3 Cl2+2NaOH=NaCl+NaClO+H2O ����2�֣�
2FeCl3 Cl2+2NaOH=NaCl+NaClO+H2O ����2�֣�
��4������2�֣�
�ٱ����ת�Ƶ����� ˫���Ŷ����ԡ�
��0.15mol ��0.3mol
���������������1��B�н����ܺͳ�����Ӧ�Ե�����2���Ʊ��������к���HCl��ˮ���������ʣ�Ӧ���ñ���ʳ��ˮ��ȥHCl������Ũ�����ȥˮ������
3mol��0.15mol ��
���㣺����������Ʊ��������й����⡣


 ��ѧ��ʦ����ϵ�д�
��ѧ��ʦ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ҵ�ϳ�����������ʢװ��Ũ���ᡣΪ�о����ʲ�������Ũ����ķ�Ӧ��ijѧϰС�����������̽�����
��̽��һ��
��1������ȥ�������������������̼�ظ֣�������Ũ�����У�10���Ӻ���������ͭ��Һ�У�Ƭ�̺�ȡ���۲죬�������������Ա仯����ԭ����__________________��
��2����ȡ����6.0 g����15.0 mLŨ�����У����ȣ���ַ�Ӧ���ռ�������Y��
��ͬѧȡ336 mL����״��������Yͨ��������ˮ�У�������Ӧ��SO2+Br2+2H2O=2HBr+H2SO4
Ȼ���������BaCl2��Һ�����ʵ�������ø������2.33 g���ɴ���֪����Y��SO2���������Ϊ______��
��̽������
��������ʵ����SO2��������Ľ������ͬѧ��Ϊ����Y�л����ܺ���H2��Q���塣Ϊ�����������̽��ʵ��װ�ã�ͼ�мг�����ʡ�ԣ���
��3��װ��B���Լ���������_________________________________________��
��4����Ϊ����Y�л�����Q��������______________________________�����û�ѧ����ʽ��ʾ����
��5��Ϊȷ��Q�Ĵ��ڣ�����װ��������M��______��ѡ����ţ���
a.A֮ǰ b.A��B�� c.B��C�� d.C��D��
��6���������Y�к���H2��Ԥ��ʵ������Ӧ��____________________________��
��7����Ҫ�ⶨ���������Y��H2�ĺ�������״����Լ��28 mL H2���������ò���H2����ķ����⣬�ɷ�ѡ�����������ķ����������жϲ�˵������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ҵ�ϳ�����������ʢװ��Ũ���ᡣij��ȤС���ͬѧ���֣���һ������������Ũ�������ʱ���۲쵽������ȫ�ܽ⣬�������������塣ʵ�������������Լ��� 0.01 mol/L ����KMnO4��Һ��0.10 mol/L KI��Һ��������ˮ��������Һ������ˮ������Э������̽��������Һ������ijɷ֡�
��������롿
��������Һ�еĽ������ӿ��ܺ���Fe2����Fe3���е�һ�ֻ����֣�
�����������п϶����� ���塣
��ʵ��̽����
| | ʵ����� | Ԥ������ | �� �� |
| ��֤����� | ����٣�ȡ����0.01 mol/L ����KMnO4��Һ������������Һ�� | | |
| ����ڣ� | | ����Fe3�� | |
| ��֤����� | ����������ͨ������װ�� | | �������ֻ��������� |


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ��ѧ��ȤС�������ͼʾʵ��װ�ã�ͼ��ʡ���˼г����������ⶨij��̼�Ͻ�������������������̽������Ũ����ķ�Ӧ��
��1��m����̼�Ͻ��м������Ũ���ᣬδ��ȼ�ƾ���ǰ��A��B��������������ԭ���Ǣٳ�����̼��Ũ�����Ӧ���� ��
��2��д������ʱA��̼��Ũ���ᷢ����Ӧ�Ļ�ѧ����ʽ ��
��3��B�е������ǣ� ��
C�������ǣ� ��
��4����A�в����ݳ�����ʱ��ֹͣ���ȣ�����E�����أ�E����bg������̼�Ͻ���������������Ϊ (д��m��b�ı���ʽ)��
�ɷ�Ӧһ��ʱ����õι���ȡA�е���Һ���뵽����ˮ����Ϊ�����������������������ӵijɷ����������ֿ���:
A��ֻ����Fe3+��B��ֻ����Fe2+��C�� ��
��֤C��ʵ�鷽����
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijͬѧ�������װ���Ʊ����ռ��������Ȼ�����Ӳ�ʲ�����E��װ��ϸ��˿����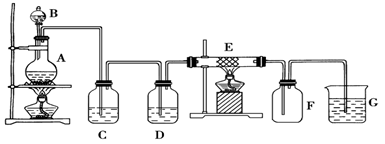
�Իش�
��1������װ��A�������Եķ�����
��2��װ��A�з�Ӧ�Ļ�ѧ����ʽΪ
��3��װ��C�������ǣ� ________________��װ��D�е��Լ��ǣ�____ _______��
��4���ɼ���װ��E�����ɵ������������ӵķ����������� ��
��5����������ͨ��ʯ����Һ�У��۲�������ǣ� ��
��6��װ�� G�з�����Ӧ�����ӷ���ʽΪ��________________ _ ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijѧУ��ѧѧϰС��Ϊ̽���������������ʣ�����ͼ��ʾװ�ý���ʵ�飬��ش���������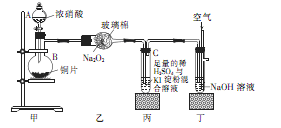
��1��װ�ü���ʢ��Ũ���������A�������� ������B�з�����Ӧ�Ļ�ѧ����ʽΪ
��2�������������еĿհ�
��3��ȡ��װ�ñ��е��Թ�C�������еμ�������Na2SO3��Һ����Һ��ɫ��ȥ���ù����з�����Ӧ�����ӷ���ʽΪ ����Ӧ�����Һ����Ҫ����SO32����SO42����I���������ӣ�����д����SO32����SO42����I����ʵ�鱨�档
��ѡ�Լ���2mol��L��1HCl��1mol��L��1H2SO4��1mol��L��1BaCl2��1mol��L��1Ba(NO3)2��CCl4�����Ʊ�����ˮ�����Ʊ�����ˮ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�� (1)ij��ѧ��ȤС����������װ����ѡȡ��Ҫ��װ����ȡ (NH4)2SO4��Һ�����ӵ�˳���ýӿ������ĸ��ʾ���ǣ�a 
(2)��װ��C������Һ����뿪�IJ���������__ _______��
װ��D�������� ��
��4�֣�Ϊ����Ȼ�淋ľ��ü�ֵ���ҹ���ѧ�����������������þ�ȷֽ��Ȼ���ư������õ���ʽ�Ȼ�þ(MgOHCl)�Ĺ��ա�ijͬѧ���ݸ�ԭ����Ƶ�ʵ��װ����ͼ��
��ش��������⣺
(1) װ��A�з�����Ӧ���ɼ�ʽ�Ȼ�þ�Ļ�ѧ����ʽΪ_________ ____
װ��B�м�ʯ�ҵ�������_____ __
(2) ��Ӧ�����г���ͨ��N2�����������㣺һ��ʹ��Ӧ�����İ�����ȫ��������ϡ���������գ�����_______________ ______
(3) װ��C���Թ��з�Ӧ�����ӷ���ʽΪ ______________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���ڴ�ŵ��������ƿ��ܻᱻ�����е�����������ij��ѧ��ȤС��ͨ��ʵ�����ⶨ���������Լ��������ij̶ȣ��������ͼʵ�飬��ش���������⣺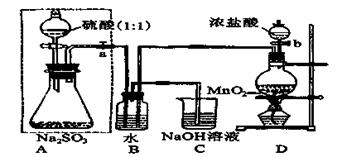
(1)Dװ���з�Ӧ�Ļ�ѧ����ʽΪ ��
Bװ���з�Ӧ�����ӷ���Ϊ ��
(2)����ag Na2SO3��Ʒ������ƿ�У���Bװ�÷�Ӧ�����Һ�м���������BaCl2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ�����ð�ɫ����bg��ԭ��Ʒ��Na2SO3��������Ϊ��Ϊ�� ��
(3)Ϊ��֤ʵ��ⶨ��ȷ�ԣ�A�е�����ʲôʱ��μ� ��
Cװ����NaOH��Һ�������� _________________��
(4)���������Լ���������ˮ����ϡ���ᡢ��ϡ���ᡢ��BaCl2��Һ����Ba(NO3)2��Һ�������ѡ������Լ������һ�ֲ�ͬ��ʵ�鷽�����ⶨ��������ˮ�������Ʊ������ij̶ȣ���ʹ���Լ���˳��Ϊ�� ��(���Լ����)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о�С��̽����
I ��ͭƬ��Ũ����ķ�Ӧ���г�װ�ú�A�м���װ������,�������Ѽ��飩
II�� SO2�� Fe( NO3)3��Һ�ķ�Ӧ[1.0 mol/L�� Fe(NO3)3��Һ�� pH��1]��ش������й����⣺
̽��I
(l)ijѧ����������ʵ��:ȡ12.8gͭƬ��20 mL 18 mol?L-1��Ũ�����������ƿ�й���,ֱ����Ӧ���,�������ƿ�л���ͭƬʣ�࣬ͬʱ������ѧ��֪ʶͬѧ����Ϊ���н϶������ʣ�ࡣ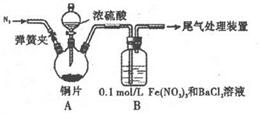
��װ��A�з�Ӧ�Ļ�ѧ����ʽ��_______
�ڸ�ͬѧ���������Ũ�ȵ�ʵ�鷽���Dzⶨ��������������䷽���ж���,�������з����в����е���______ (����ĸ����
| A�������������建��ͨ��Ԥ�ȳ�����ʢ�м�ʯ�ҵĸ����,������Ӧ���ٴγ��F |
| B�������������建��ͨ�������{�������Һ,�ټ�������BaCl2��Һ�����ˡ�ϴ�ӡ������������ |
| C������ˮ���ⶨ�������������(����ɱ�״���� |
| D�����ű���NaHSO3��Һ�ķ����ⶨ�������������(����ɱ�״���� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com