
�����֣�ʳ���еĿ�����������軯�أ��仯ѧʽΪK4[Fe(CN)6]��3H2O��42.2 g K4[Fe(CN)6]��3H2O��Ʒ������ˮ���̵���������(��Ʒ�������¶ȵı仯����)����ͼ��ʾ��
�Իش��������⣺
(1)��ȷ��150 ��ʱ�������ʵĻ�ѧʽ_____________��
(2)��������֪����Ȼ�����軯���������Ժܵͣ�����ˮ��Һ���ᷴӦ�ų��������軯��(HCN)���壻�����軯�ؼ�����һ���¶�ʱ�ֽܷ�����軯��(KCN)���ݴ��жϣ����ʳƷʱӦע�������Ϊ_____________________________________________________��
(3)��Fe2����Fe3���Ĵ������£���ʵ��2SO2��O2��2H2O��2H2SO4��ת������֪����SO2�ķ���ͨ�뺬Fe2����Fe3������Һ��ʱ������һ����Ӧ�����ӷ���ʽΪ4Fe2����O2��4H����4Fe3����2H2O������һ����Ӧ�����ӷ���ʽΪ__________________________��
(4)��֪Fe(OH)3���ܶȻ�����Ksp��1.1��10��36������ʱ��FeCl3��Һ�еμ�NaOH��Һ������ҺpHΪ3ʱ��ͨ������˵��Fe3���Ƿ������ȫ_____________��(��ʾ����ij����Ũ��С��10��5 mol��L��1ʱ������Ϊ�����ӳ�����ȫ)
(ÿ�գ��֣���ͬ�����8��)(1)K4[Fe(CN)6]
(2)���������������һ�������������¶Ȳ�����400 ��
(3)2Fe3����SO2��2H2O��2Fe2����SO42-��4H����
(4)c(Fe3��)��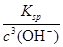 ��1.1��10��3 mol��L��1��1��10��5 mol��L��1����Fe3��û�г�����ȫ
��1.1��10��3 mol��L��1��1��10��5 mol��L��1����Fe3��û�г�����ȫ
���������������1��42.2 g K4[Fe(CN)6]��3H2O���ʵ�����42.2g��422g/mol��0.1mol�����нᾧˮ�����ʵ�����0.3mol��������0.3mol��18g/mol��5.4g������ͼ���֪����Ӧ���е�150��ʱ������ٵ�������42.2g��36.8g��5.4g����˵����ʱ���ٵ��������ǽᾧˮ����������˹���Ļ�ѧʽΪK4[Fe(CN)6]��
��2�����������軯��ˮ��Һ���ᷴӦ�ų��������軯��(HCN)���壬�������軯�ؼ�����һ���¶�ʱ�ֽܷ�����軯��(KCN)��������ʹ��ʱӦ��ע����������������һ�������������¶Ȳ�����400 �档
��3��������Fe2����Fe3���Ĵ������£���ʵ��2SO2��O2��2H2O��2H2SO4��ת��������ݷ���ʽ4Fe2����O2��4H����4Fe3����2H2O��֪��ǰ��ȥ�����õ���һ����Ӧ�����ӷ���ʽΪ2Fe3����SO2��2H2O��2Fe2����SO42-��4H����
��4������ҺpHΪ3ʱ������Һ��OH��Ũ����10��11mol/L�����ʱ��Һ��c(Fe3��)��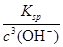 ��1.1��10��3 mol��L��1��1��10��5 mol��L��1����Fe3��û�г�����ȫ��
��1.1��10��3 mol��L��1��1��10��5 mol��L��1����Fe3��û�г�����ȫ��
���㣺�������ʻ�ѧʽȷ���ļ��㡢��������Ӧ�á�������ԭ��Ӧ����ʽ���ж��Լ��ܶȻ������ļ���



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D��E��F��G�������ʼ������ͼ��ʾ��ת����ϵ������A��B��D��G����ͬ��Ԫ�ء�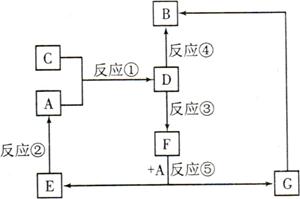
��֪��
AΪ�������ʣ�BΪ���ɫ���壬EΪ�ܶ���С�����壬GΪdz��ɫ����Һ��
D��ˮ��ҺΪ��ɫ��Һ��������������Һ��Ӧ���ɲ�����ϡ����İ�ɫ������
��ˮ��Һ��D�ܽ�ij����������ΪF��F�Ǻ�������Ԫ�صĻ����
��ش��������⣺
��1������C���ʵ�Ԫ�������ڱ��е�λ���� ���ڶ���������Ԫ���У���Ԫ����������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� (��Ԫ�ط��ű�ʾ)��
��2��D��ˮ��Һ�� �ԣ��������ӷ���ʽ����ԭ�� ��
��3��������Ӧ�������û���Ӧ���� (�����)��
��4����Ӧ��(��D��ij������������ΪF)�����ӷ���ʽ�� ��
��5��������C��������ʵ�顣��֪������Ӧ�����У�ÿ����0.1mol KI��ת�Ƶĵ�����ԼΪ3.612��1023�����밴��Ҫ����գ�
| ʵ�鲽�� | ʵ������ | д���ӷ���ʽ |
| ����������ͨ�����KI��Һ | ��Һ������ ɫ | |
| ����ͨ������ | ��Һ�����ɫ | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֡�
| ������ | CO32����SiO32����AlO2����Cl�� |
| ������ | Al3����Cu2����Mg2����NH4+��Na�� |
 Sn(OH)2
Sn(OH)2 Sn2����2OH����
Sn2����2OH�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʳ�κ�ҵ���ξ��й㷺����;����֪��ҵ������Ҫ�ɷ�ΪNaNO2����ۿ���ʳ�β�����ζ��NaNO2�������Ժͻ�ԭ�ԣ�����ֽ�ų�NO2��
��1�������Լ��ɼ���ҵ���κ�ʳ�ε���________��
a��H2O��������������b�������ữ�������� c������
��2��NaNO2������ᷴӦ����������ΪNO�������ӷ���ʽΪ______________��
��3��ij������Һ�к���2%��5%��NaNO2��ֱ���ŷŻ����ˮ��Ⱦ�����������������е�ijһ�־���ʹNaNO2�еĵ�ת��Ϊ�Կ�������Ⱦ�����壬��������________��
a��NaCl������b��NH4Cl������c��ŨH2SO4
��4���ȼҵͨ����ⱥ��ʳ��ˮ�������Ӧ�IJ����д�����ʳ��ˮ�����ӷ���ʽ______________________________________��
���ڱ�״��������3.36 L H2������Һ�����ɵ�OH�������ʵ���Ϊ____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)���ڵ��۵⻯����Һ�У��μ������������Ƽ�����Һ�������ῴ����Һ����ɫ��������Ϊ________�����ӷ���ʽ__________________________��
���ڵ�͵����γɵ���ɫ��Һ�У��μ��������Ƽ�����Һ��������ɫ����ʧ��������Ϊ________________________�����ӷ���ʽ��_______________________��
�۶ԱȢٺ͢�ʵ�����õĽ������I2��ClO����SO42������������ǿ������˳������Ϊ________________________________________��
(2)������Ƭ��ͭƬ�����ʵ��֤��������ʵ����д����Ӧ�Ļ�ѧ����ʽ��
��Ũ����������Ա�ϡ����ǿ��___________________________��
���Ȼ�����Һ��Fe3���������Ա�����ͭ��Һ�е�Cu2��ǿ��__________________________________________��
�����Ļ�ԭ�Ա�ͭǿ��_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������仯�����ڹ�ũҵ���������������ж�������Ҫ��Ӧ�á�
(1)Ca(ClO)2��NaClO��NaClO2�Ⱥ��Ȼ����ﶼ�dz��õ���������Ư��,����Ϊ���Ƕ�����������������,��д����ҵ����������NaOH��Һ��Ӧ����������NaClO�����ӷ���ʽ��������������������������������
(2)�ػ�ɫǿ�̼�������Cl2OΪ���ʹ��ϸ�Ч��ȫ���������֮һ,ʵ���ҿ��ó�ʪ��Cl2��Na2CO3��Ӧ��ȡ����Cl2O��NaHCO3�����ӷ���ʽ������������������������������������
(3)��ɫ����ClO2��������ˮɱ��������ˮ������
��KClO3��SO2��ǿ������Һ�з�Ӧ���Ƶ�ClO2,�˷�Ӧ�����ӷ���ʽΪ����������������������������
��ClO2��Ũ���������Cl2,ÿ����1 mol Cl2ת�Ƶ��ӵ����ʵ���Ϊ����������
��ClO2�ɽ���ˮ�е�Mn2+ת��ΪMnO2����ȥ,������ԭΪCl-,�÷�Ӧ�������������뻹ԭ�������ʵ���֮��Ϊ����������
(4)��Cl2����ijЩ�����л���ʱ�����������HCl�����÷�Ӧ4HCl+O2 2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�
2Cl2+2H2O,��ʵ���ȵ�ѭ�����á�
��֪:��������Ӧ��,4 mol HCl�������ų�115.6 kJ��������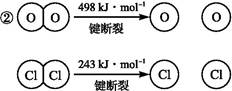
��Ͽ�1 mol H��O����Ͽ�1 mol H��Cl�������������ԼΪ�������� kJ,H2O��H��O����HCl��H��Cl��(�ǿ��������)����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D�������ʾ�Ϊ����������ɵĿ����Ի����������������ʵ����ӣ����Ӳ����ظ���ϣ��У�
| ������ | Na+��Al3+��Ba2+��NH4+ |
| ������ | Cl����OH����CO32����SO42�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������������������ǿ�������������ɷ�����H1N1���С�
��1����̼������һ���ж�����;��������ϵ��̬Ư������ѧʽ�ɱ�ʾΪNa2CO3��3H2O2��������Na2CO3��H2O2��˫�����ʡ�
��H2O2��ʱ����Ϊ��ҵ��Һ���������������ɿ�ҵ��Һ�е��軯��(��NaCN)�������·�Ӧʵ�֣�NaCN��H2O2��H2O=A��NH3������������A�Ļ�ѧʽ______________
��ijǿ���Է�Ӧ��ϵ�У���Ӧ��������ﹲ�������ʣ�
O2��MnO4-��H2O��Mn2����H2O2��H������֪�÷�Ӧ��H2O2ֻ���������¹��̣�H2O2�� O2��
д���÷�Ӧ�����ӷ���ʽ��_______________________________________________��
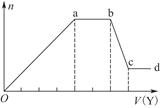
��2��ij��Ȼ��Ļ�ѧʽ�ɱ�ʾΪ:aNa2CO3��bNaHCO3��2H2O��ȡm g��Ȼ������ˮ�����Һ��������Һ����μ���1 mol/L�����ᣬ��״���²�����CO2������������������֮��Ĺ�ϵijͬѧ��������ͼ��ʾ��A��B���ߣ��Իش��������⣺
��_______������ȷ����Ȼ��Ļ�ѧʽΪ___________��
�ڼ���������CO2�������(��״��)�����ֵΪ _____________mL��
��3�� ����������ȱλ����пZnFe2Oy��������NOx��Ⱦ��ʹNOxת��ΪN2��ͬʱZnFe2Oyת��ΪZnFe2O4����2 mol ZnFe2Oy������NO2������0.5 mol N2����y��_______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
KMnO4��һ����Ҫ����������
��1����������������KMnO4�������Ի���ǿ�������ữKMnO4��Һ������Լ��� ��
a������ b������ c������
�ڽ�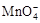 ����C2O42?�����ӷ���ʽ����������
����C2O42?�����ӷ���ʽ����������
��MnO4? +��C2O42?+�� ==��Mn2+ +��CO2��+�� ��
��2��ijͬѧΪ��̽��KMnO4��Һ��Na2C2O4�������ƣ���Һ�ķ�Ӧ���̣���������ʵ�飺
������100 mL 0.0400 mol��L-1��Na2C2O4��Һ�����õ�������ƽ��ҩ�ס��ձ�����Ͳ���������������⣬�������õ��IJ��������� ��
�ڽ�KMnO4��Һ��ε���һ�����������Na2C2O4��Һ�У��¶���ͬ��������������¼���������£�
| ����KMnO4��Һ�Ĵ��� | KMnO4��Һ��ɫ��ȥ�����ʱ�� |
| �ȵ����1�� | 60 s |
| ��ɫ���ٵ����2�� | 15 s |
| ��ɫ���ٵ����3�� | 3 s |
| ��ɫ���ٵ����4�� | 1 s |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com