
 2NH3��g����H=-92��0KJ��mol-1����1mol N2��3mol H2����һ�ܱ������У����ֺ��º�ѹ���ڴ�������ʱ���з�Ӧ���ﵽƽ��ʱ�����N2��ת����Ϊ20%��������ͬ�����£���ʼʱ�ڸ������г���2mol NH3����Ӧ�ﵽƽ��ʱ�������仯��
2NH3��g����H=-92��0KJ��mol-1����1mol N2��3mol H2����һ�ܱ������У����ֺ��º�ѹ���ڴ�������ʱ���з�Ӧ���ﵽƽ��ʱ�����N2��ת����Ϊ20%��������ͬ�����£���ʼʱ�ڸ������г���2mol NH3����Ӧ�ﵽƽ��ʱ�������仯��| A������18��4KJ���� | B���ų�73��6KJ���� |
| C���ų�18��4KJ���� | D������73��6KJ���� |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����67.7 kJ��mol��1 | B����43.5 kJ��mol��1 |
| C����43.5 kJ��mol��1 | D����67.7 kJ��mol��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 2NH3�����ֱ���t��ʱ�ⶨ����NH3�������������ͼ���ң�
2NH3�����ֱ���t��ʱ�ⶨ����NH3�������������ͼ���ң�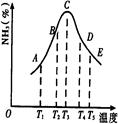
| A�������¶� | B�������¶� | C������ѹǿ | D����Сѹǿ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
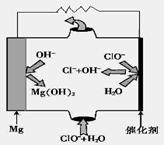
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
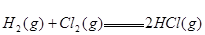 ��
��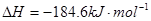 ����Ӧ
����Ӧ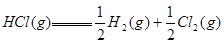 �ġ�HΪ���� ��
�ġ�HΪ���� ��| A��+184 KJ��mol | B��-92.3 KJ��mol | C��-369 KJ��mol�� | D��+92.3 KJ��mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 O2(g)===H2O(1) ��H=��285.6kJ/mol
O2(g)===H2O(1) ��H=��285.6kJ/mol�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com