
��12�֣���ˮ��Դ�����þ��й���ǰ������ˮ����Ҫ���ӵĺ������£�
| �ɷ� | ����/(mg L��1) | �ɷ� | ����/(mg L��1) |
| Cl�� | 18980 | Ca2+ | 400 |
| Na+ | 10560 | HCO3�� | 142 |
| SO42�� | 2560 | Br�� | 64 |
| Mg2+ | 1272 |
|
|
��1����������������ˮʾ��ͼ��ͼ��ʾ�����������������ӽ���Ĥ������������������ͨ����
��������Ҫ�缫��Ӧʽ�� ��
����������������������ɫ��������ɷ��� ��CaCO3������CaCO3�����ӷ���ʽ�� ��
�۵�ˮ�ij���Ϊ ���a������b����c������
��2�����ú�ˮ������ȡ���þ����ȡ�������£�
����ȡ��Ĺ����У�����2��Br����Br2ת����Ŀ���� ���������з�����Ӧ����
�ӷ���ʽ�� ����ƽ���ƶ�ԭ������ͨ������Ŀ���� ��
�ڴ�MgCl2��Һ�еõ�MgCl2��6H2O�������Ҫ������ �����ˡ�ϴ�ӡ����
�������������̣�����10 m3��ˮ�е���Ԫ��ת��Ϊ��ҵ�壬������Ҫ��״����Cl2�����Ϊ L������Cl2�ܽ⣩��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| �� |
| ||
| 2800�� |
| �� |
| ||
| ��ԭ |
| HCl |
| ||
| 714�� |
| HCl |
| ||
| ��ԭ |
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �� |
| ��� |
| 2800�� |
| �� |
| C |
| ��ԭ |
| HCl |
| ||
| 714�� |
| HCl |
| ���ý��� |
| ��ԭ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
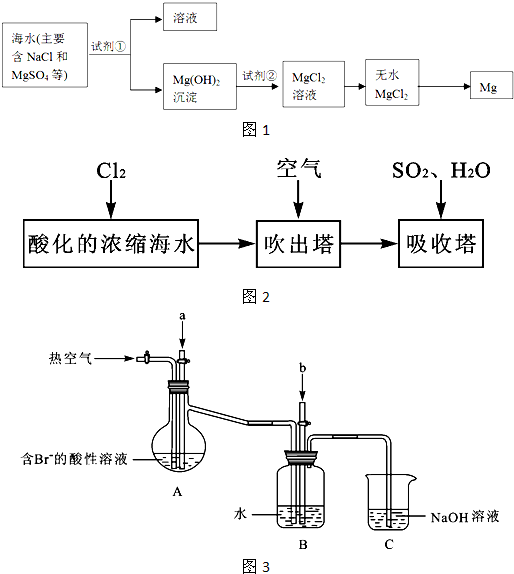
����Լռ����������71%����ˮ��ѧ��Դ�����þ��зdz�������ǰ����
��1����ˮɹ�οɻ�ô��Σ���ʵ�����д��ξ����ܽ⡢ �� ���Ƶþ��Ρ�
��2��þ����Ͻ���һ����;�ܹ�Ľ������ϣ�Ŀǰ������60%��þ�ǴӺ�ˮ����ȡ�ģ�����Ҫ�������£�
��Ϊ��ʹMgSO4ת��ΪMg(OH) 2���Լ��ٿ���ѡ�� ��ҪʹMgSO4��ȫת��Ϊ�����������Լ��ٵ���Ӧ ��
���Լ��ڿ���ѡ�� ��
���Դӽ�Լ��Դ����߽���þ�Ĵ��ȷ������������˵�ұþ������ ��
��3���弰�仯������;ʮ�ֹ㷺���ҹ����ڴ�����չ��ˮ������о��Ϳ�����������ҵ��Ũ����ˮΪԭ����ȡ��IJ��ֹ������£�
ij����С����ʵ����ģ�����������������װ�ý���ʵ�飨��������Ʒ���ѱ��������г�װ������ȥ����
��Aװ����ͨ��a�����Ŀ���ǣ������ӷ���ʽ��ʾ�� ��
��Aװ����ͨ��a����һ��ʱ���ֹͣͨ�룬��ͨ�ȿ�����ͨ���ȿ�����Ŀ����
��
�۷�Ӧ�����У�Bװ������SO42-���ɡ�����SO42-�ķ����� ��
��Cװ�õ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�걱���а�һ��ѧ��һ�ڶ�ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
����Լռ����������71%����ˮ��ѧ��Դ�����þ��зdz�������ǰ����
��1����ˮɹ�οɻ�ô��Σ���ʵ�����д��ξ����ܽ⡢ �� ���Ƶþ��Ρ�
��2��þ����Ͻ���һ����;�ܹ�Ľ������ϣ�Ŀǰ������60%��þ�ǴӺ�ˮ����ȡ�ģ�����Ҫ�������£�
��Ϊ��ʹMgSO4ת��ΪMg(OH) 2���Լ��ٿ���ѡ�� ��ҪʹMgSO4��ȫת��Ϊ�����������Լ��ٵ���Ӧ ��
���Լ��ڿ���ѡ�� ��
���Դӽ�Լ��Դ����߽���þ�Ĵ��ȷ������������˵�ұþ������ ��
��3���弰�仯������;ʮ�ֹ㷺���ҹ����ڴ�����չ��ˮ������о��Ϳ�����������ҵ��Ũ����ˮΪԭ����ȡ��IJ��ֹ������£�
ij����С����ʵ����ģ�����������������װ�ý���ʵ�飨��������Ʒ���ѱ��������г�װ������ȥ����
��Aװ����ͨ��a�����Ŀ���ǣ������ӷ���ʽ��ʾ�� ��
��Aװ����ͨ��a����һ��ʱ���ֹͣͨ�룬��ͨ�ȿ�����ͨ���ȿ�����Ŀ����
��
�۷�Ӧ�����У�Bװ������SO42-���ɡ�����SO42-�ķ����� ��
��Cװ�õ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�걱���и�һ�ڶ�ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
����Լռ����������71%����ˮ��ѧ��Դ�����þ��зdz�������ǰ����
��1����ˮɹ�οɻ�ô��Σ���ʵ�����д��ξ����ܽ⡢ �� ���Ƶþ��Ρ�
��2��þ����Ͻ���һ����;�ܹ�Ľ������ϣ�Ŀǰ������60%��þ�ǴӺ�ˮ����ȡ�ģ�����Ҫ�������£�

��Ϊ��ʹMgSO4ת��ΪMg(OH) 2���Լ��ٿ���ѡ�� ��ҪʹMgSO4��ȫת��Ϊ�����������Լ��ٵ���Ӧ ��
���Լ��ڿ���ѡ�� ��
���Դӽ�Լ��Դ����߽���þ�Ĵ��ȷ������������˵�ұþ������ ��

��3���弰�仯������;ʮ�ֹ㷺���ҹ����ڴ�����չ��ˮ������о��Ϳ�����������ҵ��Ũ����ˮΪԭ����ȡ��IJ��ֹ������£�

ij����С����ʵ����ģ�����������������װ�ý���ʵ�飨��������Ʒ���ѱ��������г�װ������ȥ����

��Aװ����ͨ��a�����Ŀ���ǣ������ӷ���ʽ��ʾ�� ��
��Aװ����ͨ��a����һ��ʱ���ֹͣͨ�룬��ͨ�ȿ�����ͨ���ȿ�����Ŀ����
��
�۷�Ӧ�����У�Bװ������SO42-���ɡ�����SO42-�ķ����� ��
��Cװ�õ������� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com