
����Ŀ��ij�¶�ʱ��BaSO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ����ش��������⣺
��1������˵����ȷ����__________(����ĸ���)��
A������Na2SO4����ʹ��Һ��a��䵽b��
B��ͨ����������ʹ��Һ��d��䵽c��
C��d����BaSO4��������
D��a���Ӧ��Ksp����c���Ӧ��Ksp
��2����100 mL 1 mol��L��1 H2SO4����100 mL��Ba2�� 0.137 g ����Һ�г�ַ�Ӧ���˳���������Һ�в�����Ba2�������ʵ���Ũ��Ϊ____________(������λ��Ч����)��
��3����������100 mL��ˮ��100 mL 0.01 mol��L��1��H2SO4�ֱ�ϴ�ӣ������������ʧ��BaSO4������֮��Ϊ_____________��
���𰸡���1��C ��2��2.02��10��10 mol��L��1 ��3��103��1
��������
�����������1��A�����ᱵ��Һ�д������ܽ�ƽ�⣬a����ƽ�������ϣ�����Na2SO4��������c(SO42-)��ƽ�����ƣ�c(Ba2+)Ӧ���ͣ���A����B��d��ʱ��Һ�����ͣ������ܼ�ˮ��c(SO42-)��c(Ba2+)������B����C��d���ʾQc��Ksp����Һ�����ͣ������г�����������C��ȷ��D��Ksp��һ�������¶Ȳ���Ksp���䣬�������ϵ�����һ��Ksp����ȣ���D���ʴ�ΪC��
��2����ͼ���֪Ksp(BaSO4)=10-5��10-5=10-10����100mL 1molL-1H2SO4��Һ����100mL��Ba2+0.137g����Һ�г�ַ�Ӧ��Ӧ����BaSO4����������ӹ���������n(Ba2+)=0.137g��137g/mol=0.001mol��ʣ�����������ӵ�Ũ��Ϊ��c(SO42-)=![]() =0.495mol/L������ʣ��ı�����Ϊ��c(Ba2+)=
=0.495mol/L������ʣ��ı�����Ϊ��c(Ba2+)=![]() =2.02��10-10mol/L��
=2.02��10-10mol/L��
��3����100mL����ˮϴ�ӳ���ʱ���ܽ��BaSO4�����ʵ���Ϊ0.1L �� c(Ba2+) = 0.1L �� ![]() mol/L= 0 .1L ��
mol/L= 0 .1L �� ![]() mol/L = 10-6mol����100mL 0.01molL-1H2SO4��Һϴ��ʱ����������������˳������ܽ⣬���ܽ��BaSO4�����ʵ���Ϊ0.1L��c(Ba2+)=0.1L��
mol/L = 10-6mol����100mL 0.01molL-1H2SO4��Һϴ��ʱ����������������˳������ܽ⣬���ܽ��BaSO4�����ʵ���Ϊ0.1L��c(Ba2+)=0.1L��![]() mol/L=10-9mol����ͬ�����ʵ�����֮�ȵ������ʵ�����֮�ȣ�������ϴ�ӷ�����BaSO4�����������֮��Ϊ10-6mol��10-9mol=1000��1�����������������ʧ��BaSO4������֮��Ϊ1000��1��
mol/L=10-9mol����ͬ�����ʵ�����֮�ȵ������ʵ�����֮�ȣ�������ϴ�ӷ�����BaSO4�����������֮��Ϊ10-6mol��10-9mol=1000��1�����������������ʧ��BaSO4������֮��Ϊ1000��1��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������������ʡ����������ʵ���Ҫԭ�ϡ�
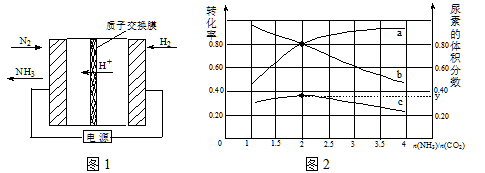
��1���ϳɰ���ӦN2��g��+ 3H2��g��![]() 2NH3��g����һ�����������Է����е�ԭ���� ���绯ѧ���Ǻϳɰ���һ���·�������ԭ����ͼ1��ʾ�������ĵ缫��Ӧʽ�� ��
2NH3��g����һ�����������Է����е�ԭ���� ���绯ѧ���Ǻϳɰ���һ���·�������ԭ����ͼ1��ʾ�������ĵ缫��Ӧʽ�� ��
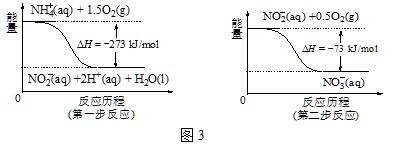
��2����̼��[n��NH3��/n��CO2��]�Ժϳ�����[2NH3��g��+CO2��g��![]() CO��NH2��2��g��+H2O��g��]��Ӱ�죬���º���ʱ���������ʵ���3 mol��NH3��CO2�Բ�ͬ�İ�̼�Ƚ��з�Ӧ�������ͼ2��ʾ��a��b�߷ֱ��ʾCO2��NH3��ת���ʱ仯��c�߱�ʾƽ����ϵ�����ص���������仯��[n��NH3��/ n��CO2��]= ʱ�����ز���������㣬ͼ��y= ����ȷ��0��01����
CO��NH2��2��g��+H2O��g��]��Ӱ�죬���º���ʱ���������ʵ���3 mol��NH3��CO2�Բ�ͬ�İ�̼�Ƚ��з�Ӧ�������ͼ2��ʾ��a��b�߷ֱ��ʾCO2��NH3��ת���ʱ仯��c�߱�ʾƽ����ϵ�����ص���������仯��[n��NH3��/ n��CO2��]= ʱ�����ز���������㣬ͼ��y= ����ȷ��0��01����
��3����ˮ�к���������Ĵ��������ж��֡�
���ô��������������������Եõ�N2H4��ϡ��Һ���÷�Ӧ�Ļ�ѧ����ʽ��_____________��
����NaClO��ҺҲ�ɽ���ˮ�е�NH4+ ת��ΪN2�������������в���N2 0��672 L����״����������Ҫ����0��3 mol��L��1��NaClO��Һ L��
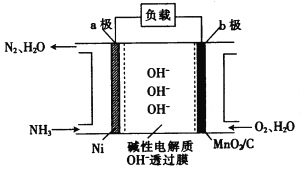
��������������£�NH4+ ����������Ӧ��ת��ΪNO3-��������Ӧ�������仯��ͼ3��ʾ����1 mol NH4+ ��aq��ȫ����������NO3-��aq��ʱ�ų��������� kJ��
����H2����ԭ���ɽ���ˮ��NO3-��Ũ�ȣ��õ��IJ����ܲ������ѭ������Ӧ����Һ��pH ������ߡ��������͡����䡱����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͭп�Ͻ��Ƴɼٽ�Ԫ����ƭ�����¼����з��������в�����������α�ķ�����
A. �ⶨ�ܶ�
B. ����������
C. ����������
D. �۲����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������(N2H4)����һ�ֹ�ҵԭ�ϣ�����һ����Ҫ�Ĺ�ҵ��Ʒ��
(1) �����ʵ����İ��������ֱ�����������������Ӧ������Ϊ������ˮ����ת�Ƶĵ�����֮��Ϊ___________��
(2) ������һ�������¿ɰ���ʽ�ֽ�:3N2H4(g)=N2(g)+4NH3(g)����֪����N-H��N-N��N��N��1 mol�����յ���������Ϊ390.8 kJ��193 kJ��946 kJ��������1 mol N2����Ӧ________(�����ų�������������)___ _______kJ��������
(3)NH3-O2ȼ�ϵ�صĽṹ��ͼ��ʾ��
�� a��Ϊ��ص�________(����������������)����
��������1 mol N2ʱ����·���������ӵ����ʵ���Ϊ_____________��
(4)�������Ը�Ч���ѳ��̵����е�NO�Ӷ����� N2���÷�Ӧ�����������뻹ԭ��������ʵ���֮��Ϊ__________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����һƿ�������Һ�����п��ܺ���NH![]() ��K����Ba2����Al3����Fe3����I����NO
��K����Ba2����Al3����Fe3����I����NO![]() ��CO
��CO![]() ��SO
��SO![]() ��AlO
��AlO![]() ��ȡ����Һ��������ʵ�飺
��ȡ����Һ��������ʵ�飺
����pH��ֽ���飬��Һ��ǿ���ԡ�
��ȡ��Һ��������������CCl4������������ˮ����CCl4����Ϻ�ɫ��
����ȡ��Һ��������μ���NaOH��Һ��A��Һ�����Ա�Ϊ���ԣ�B��Һ����������C������ȫ�ܽ⣻D��������Һ��������ų�����������ʹʪ��ĺ�ɫʯ����ֽ������
��ȡ�����۵õ��ļ�����Һ������Na2CO3��Һ���а�ɫ�������ɡ�
��������ʵ�����ش��������⡣
��1���ɢٿ����ų�________________�Ĵ��ڡ�
��2���ɢڿ���֤��________�Ĵ��ڣ�ͬʱ�ų�__________�Ĵ��ڣ�������_________ ______��
��3���ɢۿ���֤��________�Ĵ��ڣ�д��c��d���漰�Ļ�ѧ����ʽ�������ӷ�Ӧ�������ӷ���ʽ��ʾ��
c________________��d________________��
��4���ɢܿ���֤��____________�Ĵ��ڣ�ͬʱ�ų�____________�Ĵ��ڡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
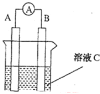
����Ŀ����1����ͼ��ʾ����CΪŨ���ᣬ������ָ�뷢��ƫת��B�缫����ΪFe ,A�缫����ΪCu����B�缫�ĵ缫��ӦʽΪ______________��A�缫�ĵ缫��ӦʽΪ ����Ӧ����һ��ʱ�����ҺC��pH�� (����ߡ������͡��������䡱)��
��2���ҹ��״�����������ˮ�����Ϊ��Դ�����͵ĺ�ˮ��־�ƣ��Ժ�ˮΪ�������Һ���������е�����ʹ����������������������ֻҪ�ѵƷ��뺣ˮ�����ӣ��ͻᷢ��ҫ�۵İ⡣���Դ�ĸ���������________��������ӦΪ___________��������ӦΪ_______________________��
��3�����ε�ؾ��иߵķ���Ч�ʣ�����ܵ����ӣ� ����Li2CO3��Na2CO3�������λ����������ʣ�COΪ����ȼ����������CO2�Ļ����Ϊ������ȼ�����Ƶ���650 ���¹�����ȼ�ϵ�أ�����йص�ط�Ӧʽ��������ӦʽΪ2CO��2CO32��4e��=4CO2��������ӦʽΪ________________________������ܷ�ӦʽΪ_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������Ȼ�ѧ����ʽ�У���ȷ������ ��
A�������ȼ����Ϊ890.3 kJ��mol��1�������ȼ�յ��Ȼ�ѧ����ʽ�ɱ�ʾΪCH4(g)��2O2(g)��CO2(g)��2H2O(g) ��H����890.3 kJ��mol��1
B����101 kPaʱ��2 g H2��ȫȼ������Һ̬ˮ���ų�285.8 kJ����������ȼ�����Ȼ�ѧ����ʽ��ʾΪ
2H2(g)��O2(g)��2H2O(l) ��H����571.6 kJ��mol��1
C��HCl��NaOH��Ӧ���к��Ȧ�H����57.3 kJ��mol����H2SO4��Ca(OH)2��Ӧ���к��Ȧ�H��2��(��57.3)kJ��mol��1
D��500����30 MPa�£���0.5 mol N2(g)��1.5 mol H2(g)�����ܱ������г�ַ�Ӧ����NH3(g)����19.3 kJ�����Ȼ�ѧ����ʽΪN2(g)��3H2(g)![]() 2NH3(g) ��H����38.6 kJ��mol��1
2NH3(g) ��H����38.6 kJ��mol��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����й���Ԫ�����ڱ���˵����������ǣ� ��
A.Ԫ�����ڱ���Ԫ�ذ�ԭ��������С���ж��ɵ�
B.Ԫ��ԭ�ӵĵ��Ӳ����������������ڵ���������
C.Ԫ��ԭ�ӵ������������������������������
D.Ԫ�����ڱ��й���18�У�16����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com