
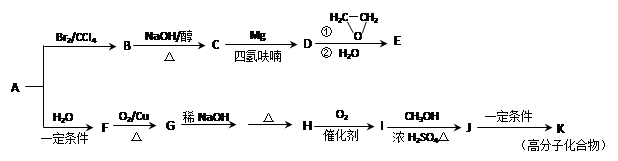
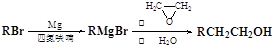
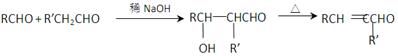
 1,2-��������
1,2-��������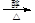 CH2="CHBr" + NaBr + H2O
CH2="CHBr" + NaBr + H2O 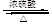
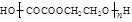 +(2n��1)H2O
+(2n��1)H2O 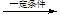
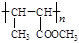
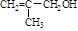
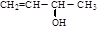
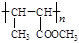 ��
��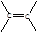 ��B������Ϊ1��2-�������顣
��B������Ϊ1��2-�������顣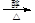 CH2="CHBr" + NaBr + H2O��BrCH2CH2Br����������ˮ��Һ����ˮ�ⷴӦ����HOCH2CH2OH�������Ҷ����ܷ������۷�Ӧ����Ӧ����ʽΪnHOOCCOOH+nHOCH2CH2OH
CH2="CHBr" + NaBr + H2O��BrCH2CH2Br����������ˮ��Һ����ˮ�ⷴӦ����HOCH2CH2OH�������Ҷ����ܷ������۷�Ӧ����Ӧ����ʽΪnHOOCCOOH+nHOCH2CH2OH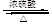
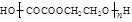 +(2n��1)H2O��
+(2n��1)H2O��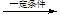
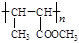 ��
��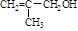 ��
��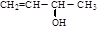 ��
��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
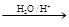 R��CH2CHO + R��OH
R��CH2CHO + R��OH 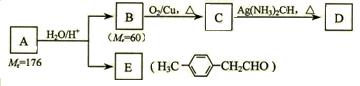
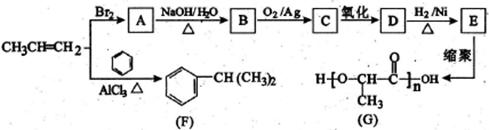
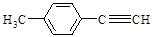 ����һ��·�����£�
����һ��·�����£�
|
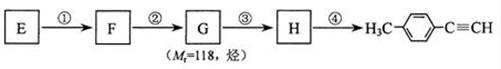 ��4��д��G�Ľṹ��ʽ��____________________________________��
��4��д��G�Ľṹ��ʽ��____________________________________��| ��� | �����Լ�����Ӧ���� | ��Ӧ���� |
| �� | Ũ , �� , �� | |
| �� | Br2��CCl4��Һ | |
| �� | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ����� | �����ʵ��Լ� | �����ʵķ��� |
| �������ӣ� | ��1�� | ��4�� |
| �ƾ���ˮ�� | ��2�� | ��5�� |
| �������������ᣩ | ��3�� | ��6�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 �Ļ�ѧ����ʽΪ
�Ļ�ѧ����ʽΪ ��Ϊͬ���칹��ķ����廯������ �֣�����һ��ͬ���칹��ĺ˴Ź�������������������ԭ�ӵ����շ壬��ͬ���칹��Ľṹ��ʽΪ ��
��Ϊͬ���칹��ķ����廯������ �֣�����һ��ͬ���칹��ĺ˴Ź�������������������ԭ�ӵ����շ壬��ͬ���칹��Ľṹ��ʽΪ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

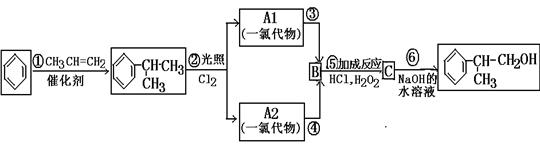
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

 CH3COOC2H5
CH3COOC2H5�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

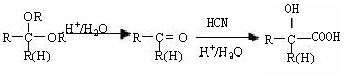
| A��������II �ķ���ʽC5H9O2 |
| B����Ӧ�ܵ�Ŀ���Ƿ�ֹ��CHO�ڵڢݲ������� |
| C����Ӧ������ȥ��Ӧ����Ӧ����������Ӧ |
| D��������D ���Է������۷�Ӧ�õ��߷��ӻ����� |
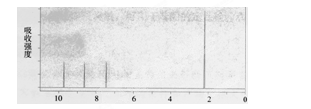
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
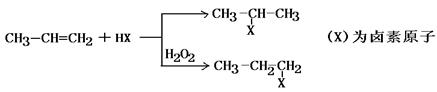
 ��X���ܷ���
��X���ܷ���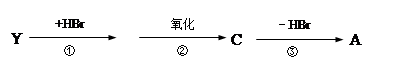
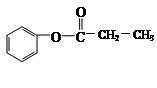 ��__________________________��
��__________________________���鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com