

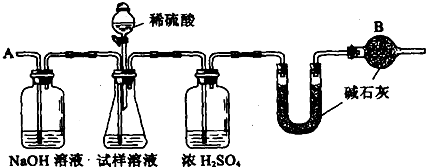
��Ҫʵ�鲽�����£�
�ٰ�ͼ��װ�����������װ�õ�������
�ڽ�a g����������ƿ�У�����������ˮ�ܽ⣬�õ�������Һ
�۳���ʢ�м�ʯ�ҵ�U�ܵ��������õ�b g
�ܴӷ�Һ©������6mol��L��1�����ᣬֱ�����ٲ�������ʱΪֹ
�ݴӵ���A����������һ�����Ŀ���
���ٴγ���ʢ�м�ʯ�ҵ�U�ܵ��������õ�c g
���ظ�����ݺ͢IJ�����ֱ��U�ܵ������������䣬Ϊd g
����պͻش����⣺
��1������������ƽ������Ʒʱ�������ƽ��ָ������ƫת��˵��
��2��װ���и����B��������
��3���������Һ©���е����ỻ��Ũ����ͬ�����ᣬ���ԵĽ�� (��ƫ�ߡ�ƫ�ͻ�)
��4������ݵ�Ŀ����
��5������ߵ�Ŀ����
��6���������д����������������ʽΪ
��7��������������ʵ�鷽���ⶨ�����д�������������������һ�ֲ�ͬ��ʵ�鷽����
�𰸣�
��1����Ʒ�أ�������
��2����ֹ�����е�CO2��ˮ������U����
��3��ƫ��
��4���ѷ�Ӧ������CO2ȫ������U����
��5���жϷ�Ӧ������CO2�Ƿ�ȫ���ų�������U���еļ�ʯ������
��6��![]() ��100��
��100��
��7�����ԡ�
��������1��������ƽ����ʱ�����̷�ҩƷ�����̷����룬��ָ����ƫ��˵�����̽��أ�Ҳ����ҩƷ��������������������2��ͨ������ʵ�鿪ʼ�ͽ���ʱU�ιܵ�����������ȷ������CO2����������U�ιܺ��һ������ܣ����Է�ֹ�����е�CO2��ˮ�����Ƚ���U�ιܣ�ʹʵ������Ϊȷ����3���������ǻӷ����ᣬ���ɵ�CO2�л����HCl�������ܱ�ŨH2SO4��ȥ�������Ա���ʯ�����գ�Ҳ����ʵ��������ʯ�ҵ�������ƫ�����Ľ����ƫ�ߡ���4��CO2���ܶȽϴ����ɵ�CO2���ܻ��������ƿ�У���������������ɵ�CO2ȫ������U�ι��С���5��������γ���U�ιܵĽ���������䣬˵����ƿ�����е�CO2�Ķ�����ʯ�������ˡ�
��6��Na2CO3+H2SO4====Na2SO4+H2O+CO2��
106 g 44 g
m��Na2CO3�� ��d��b�� g
���ԣ�Na2CO3����������Ϊ��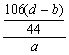 ��100%=
��100%=![]() ��100%
��100%
��7����ʵ��ķ����϶࣬������Գ���һ����������Ʒ������������ϡHNO3ʹCO2ȫ���ų���μ�����AgNO3������ϴ��������������ͨ������Ҳ��ȷ����Ʒ�Ĵ��ȡ���������ֻҪԭ����ȷ���������ж��ǿ��Եġ�


 ��������ϵ�д�
��������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| 106(d-b) |
| 44a |
| 106(d-b) |
| 44a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
- 3 |
- 3 |
| ||
| 106(d-b) |
| 44a |
| 106(d-b) |
| 44a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| 106(d-b) |
| 44 |
| 106(d-b) |
| 44 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| 106(d-b) |
| 44a |
| 106(d-b) |
| 44a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�����������������ѧ��һ��ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
(10��).��֪ij���������к��������Ȼ��ƣ�Ϊ�ⶨ�����д��������������������ͼװ�ý���ʵ�顣

��Ҫ�������£�����գ�
�� ��ͼ��װ�����������װ�õ������Ԣ� ��a g����������ƿ�У�����������ˮ�ܽ⣬�õ�������Һ���� ����ʢ�м�ʯ�ҵ�U�ιܵ�����Ϊb g���� �ӷ�Һ©���е���6mol/L��ϡ���ᣬֱ�����ٲ�������Ϊֹ���� �ӵ���A����������һ�����Ŀ������� �ٴγ���ʢ�м�ʯ�ҵ�U�ιܵ�����Ϊc g���� �ظ��ݺ͢IJ�����ֱ��U�ιܵ������������䣬�������Ϊd g��
�ش��������⣺(��ʯ����һ�ָ����,���ܸ�����������)
(1)װ���и����B��������
(2)�������Һ©���е����ỻ��ͬŨ�ȵ�����,���ԵĽ���� (��ƫ��.ƫ�ͻ�)
(3)����ݵ�Ŀ����
(4)����ߵ�Ŀ����
(5)�������д�������������ļ���ʽΪ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com