
�������ȣ�ClO2����һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ��59�棬�е�Ϊ11.0�棬������ˮ����ҵ�����Գ�ʪ��KClO3�Ͳ��ᣨH2C2O4����60��ʱ��Ӧ�Ƶá�ijѧ��������ͼ��ʾװ��ģ�ҵ��ȡ���ռ�ClO2��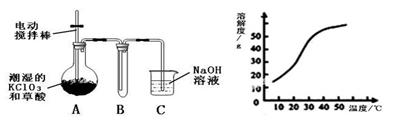
��1��A���������¶ȿ���װ�ã����ƾ��ơ��¶ȼ��⣬����Ҫ�IJ��������� ��
��2����Ӧ����װ��C�пɵ�NaClO2��Һ����֪���¶ȵ���38��ʱNaClO2������Һ������������NaClO2��3H2O�����¶ȸ���38��ʱ����������NaClO2����������ͼ��ʾ��NaClO2���ܽ�����ߣ��벹���NaClO2��Һ���Ƶ�NaClO2����IJ������裺 �� �����ᾧ���� ���� ϴ�ӣ��� ���
��3��ClO2�ܲ��ȶ������������ƣ���ˮ���յõ�ClO2��Һ��Ϊ�ⶨ������Һ��ClO2��Ũ�ȣ�����������ʵ�飺�� ȷ��ȡClO2��ҺV1mL���뵽��ƿ�У�����������ˮϡ�ͣ�����������pH��2.0���� ����������KI���壬����Ƭ�̡���ʱ������Ӧ�����ӷ���ʽΪ�� ���� �������ָʾ������c mol/L Na2S2O3��Һ�ζ������յ�ʱ����Na2S2O3��ҺV2 mL����ԭClO2��Һ��Ũ��Ϊ mol��L���ú���ĸ�Ĵ���ʽ��ʾ��������֪2 Na2S2O3+I2= Na2S4O6+2NaI��
����Na+��Ba2+��Cu2+��SO42����Cl�� ����γɵ�����ǿ�������Һ���ֱ�װ����ͼװ��
�еļס��ҡ��������ձ��н��е�⣬�缫��Ϊʯī�缫��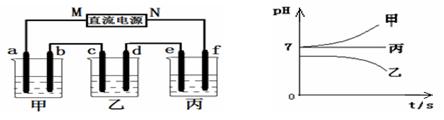
��ͨ��Դ������һ��ʱ��������c�缫�������ӡ������¸��ձ�����ҺpH����ʱ��t�Ĺ�ϵ������ͼ�������������ܽ������Ӱ�죩���ݴ˻ش��������⣺
��1��д�����ձ��з�����Ӧ�Ļ�ѧ����ʽ ��
��2���缫f�Ϸ����ĵ缫��ӦΪ ��
��3��������һ��ʱ�������ձ���c�缫����������8g��Ҫʹ���ձ�����Һ�ָ���ԭ����״̬��Ӧ���еIJ����� ��
I�� (1) �ձ� (2��) ��2�� ���ȹ��� ��2 �֣�
��3��2ClO2 + 8H+ + 10I��="==2" Cl��+ 5I2 + 4H2O ��3 �֣� 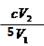 ��2 �֣�
��2 �֣�
��1��2CuSO4 + 2H2O  2Cu + O2�� + 2H2SO4��2�֣�
2Cu + O2�� + 2H2SO4��2�֣�
��2��4OH����4e��=2H2O + O2����2�֣�
��3������ձ��м���2.25gˮ��3�֣�
���������������1������Ҫ������������Ϣ���������ᵽ�Ʊ������������ɳ�ʪ��KClO3�Ͳ��ᣨH2C2O4����60��ʱ��Ӧ�Ƶõģ���˰�ʾ�÷�Ӧ��Ҫ���Ʒ�Ӧ�¶ȡ����Ƹ÷�Ӧ�¶���60�棬����ѡ��60�����ˮԡ�������ˮԡ���¶ȿ���װ�ó��ƾ��ơ��¶ȼ��⣬����Ҫ�IJ�������Ӧ������װˮԡ�����õĴ��ձ���
��2��������˵��NaClO2��Һ���õ�2�о��壬������38��ʱNaClO2������Һ������������NaClO2��3H2O������38��ʱ����������NaClO2 �����Ҫ�õ��ᾧˮ��NaClO2 ���壬Ҫ��֤�ڹ��˵�ʱ���¶ȸ���38�棬��˲��ó��ȹ��ˡ�
��3���ữ��ClO2��Һ�м���KI�������Һ������˵���еⵥ�����ɣ����Է�����������ԭ��Ӧ���ӷ���ʽΪ2ClO2 + 8H+ + 10I��="==2" Cl��+ 5I2 + 4H2O��
������֪��Ӧ����ʽ��ϵ�ã�n(ClO2)="2/5" n(I2) �� n(I2)="1/2" n(Na2S2O3)�����n(ClO2)="1/5" n(Na2S2O3)������c (ClO2 )= 1/5��c��V2 ��V1 = mol��L
mol��L
�ӳ����¸��ձ�����ҺpH����ʱ��t�Ĺ�ϵͼ��֪���ס������ձ���ʼpHֵ����7���ס����ĵ������Һ��ǿ��ǿ���Σ����ձ���ʼpHֵС��7���������ҺΪǿ�������Ρ����ձ��ĵ缫C�������أ�����ƶ�����ԭ��Ϊ�н���Cu�������õ缫�����������ɴ˿��Եó���ԴMΪ������NΪ������a��c��eΪ������b��d��fΪ������
��1�����ձ��ĵ缫C�������أ�����ΪCu2+�ŵ磻��ҺpHֵ���ͣ�˵����Һ������OH-�ŵ磬ʹ��Һ�����������ࣻ���Ը��ݷŵ���������Ʋ�ó����еĵ������ҺΪCuSO4��Һ����˵�����Һ�Ļ�ѧ����ʽΪ2CuSO4 + 2H2O  2Cu + O2�� + 2H2SO4 ��
2Cu + O2�� + 2H2SO4 ��
��2�����ձ��еĵ������ǿ��ǿ���Σ����ŵ��Ľ��У����е���ҺpHֵ���䣬������eΪH+�ŵ磬����f��OH-�ŵ磬���ʵ���ڵ��ˮ���ɴ˿��Ʋ�õ������Һ��Na2SO4�����f�缫��ӦʽΪ4OH����4e��=2H2O + O2����
��3�����ձ���c�缫����������8g����������0.125mol�ĵ���Cu����ת�Ƶ���0.25mol����˱��ձ��е������0.125mol��ˮ��Ҫʹ��Һ�ָ���Ӧ�ü���0.125mol��ˮ����2.25gˮ��
���㣺���⿼�����ʵ��������ʵ��������绯ѧ����֪ʶ��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һ����ı��أ���ˮ���̺�80����Ԫ�ء��ȼҵ���Ʊ�����þ��ԭ�϶������ں�ˮ��
�����ȼҵ�У�����ʯ��Ĥ���������ʳ��ˮ����ͼ����ʾ����
��1��д�������ķ�Ӧʽ�� ��
��2��ͼ����������b�� ��Һ��
��3��ʯ��Ĥ�������� ��
�����ſƼ��ķ�չ�����ղ��ϸ��£����Ч�ʺͲ�Ʒ���ȵõ���ߡ�20����80����𣬸�Ĥ�����������ӽ���Ĥ��⼼��ȡ����
��1�����ӽ���Ĥ���ۣ���ͼ����ʾ���Тޡ��߷ֱ��� �� ��
��2����֪һ�����ӵĵ�����1.602��10��19C��������Ĥ���۵�ⱥ��ʳ��ˮ������·��ͨ��1.929��105 C�ĵ���ʱ������NaOH g��
����ͼ�ǹ�ҵ������þ�����̡�
��1��д�����з�Ӧ�Ļ�ѧ����ʽ��
�ٳ����أ� �ڵ� �⣺
��2����������������ѭ��ʹ�õ������� ��
��3�����������Ȼ�þ�Ľᾧˮ����ʹ֮��ˮת��Ϊ��ˮ�Ȼ�þ��ע����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ȤС��������ͼ��ʾװ�ý���ʵ�顣
��1���Ͽ�K2���պ�K1��U�ι��ڳ��缫���������⣬���ɹ۲쵽�������� �������缫��ӦʽΪ ������·��ת��0.001mol����ʱ���Ҳ�ע�����������ռ������� mL������Ϊ��״������
��2���Ͽ�K2���պ�K1һ��ʱ�䣬��ע�����г���һ����������Ͽ�K1���պ�K2����ʱװ���ڻ�ѧ��ת��Ϊ���ܡ�ʵ������ת���Ļ�ѧ����ʽΪ ��
��3������ʵ��װ�ò�©��������2����ע����������ȫ���μӷ�Ӧ��U�ι��ڵ���Һ������ ����ܡ����ܡ����ָ�ԭ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪Ǧ���صĹ���ԭ��ΪPb��PbO2��2H2SO4 2PbSO4��2H2O��������ͼװ�ý��е��(���Һ����)����õ� Ǧ������ת��0.4 mol����ʱ���缫����������11.2 g����ش� �������⡣
2PbSO4��2H2O��������ͼװ�ý��е��(���Һ����)����õ� Ǧ������ת��0.4 mol����ʱ���缫����������11.2 g����ش� �������⡣
(1)A��Ǧ���ص� ����Ǧ����������ӦʽΪ ���ŵ�����е��Һ���ܶ� (���С�����������䡱)��
(2)Ag�缫�ĵ缫��Ӧʽ�� ���õ缫�ĵ缫���ﹲ g��
(3)Cu�缫�ĵ缫��Ӧʽ�� ��CuSO4��Һ��Ũ�� (���С�����������䡱)
(4)��ͼ��ʾ�����й�����ij����(������x)��ʱ��ı仯���ߣ���x��ʾ ��
a����U�ι��в�������������
b����U�ι������������ļ�����
c����U�������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijͬѧ����ԭ���ԭ����ʵ��ʱ,������ʵ�鲽��:
���õ��߽����������Ƶ����˷ֱ��봿����пƬ��ͭƬ������(��ͼ1);
�ڰ�һ�鴿����пƬ����ʢ��ϡ������ձ���;
�۰�һ�鴿����ͭƬ����ʢ��ϡ������ձ���;
���õ��߰�пƬ��ͭƬ����������,��ƽ�еز���ʢ��ϡ������ձ���(��ͼ2)��
�ش���������:
(1)ʵ�鲽�����Ӧ�۲쵽���������� ��
(2)ʵ�鲽�����Ӧ�۲쵽���������� ��
(3)ʵ�鲽�����Ӧ�۲쵽���������� ��
(4)ʵ�鲽�����Ӧ�۲쵽���������� ��
(5)ͨ��ʵ�鲽��ܸ�ͬѧͷ��������һ������(�����),�ò���������
(6)Ϊ��֤ʵ�ò���,��ͬѧ������˵ڢݲ�ʵ��,���Ҫ�����ڢݲ�ʵ���װ��ʾ��ͼ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�ϵ�ⱥ��ʳ��ˮ����ȡ���ֻ���ԭ�ϣ����в���ԭ�Ͽ������Ʊ��ྦྷ�衣
(1)��ͼ�����ӽ���Ĥ����ⱥ��ʳ��ˮʾ��ͼ����������������������________��NaOH��Һ�ij���Ϊ________(����ĸ)�����Ʊ���ʳ��ˮ�Ľ���Ϊ________(����ĸ)����������Ӧʹ�õ�Һ����________��
(2)�ྦྷ����Ҫ����SiHCl3��ԭ�����������丱����SiCl4���ۺ������ܵ��㷺��ע��
��SiCl4���������̿��(����ά��Ҫԭ����ͬ)������Ϊ������SiCl4��H2��O2��Ӧ�����������֣���ѧ����ʽΪ___________________________________��
��SiCl4��ת��ΪSiHCl3��ѭ��ʹ�ã�һ�������£���20 L�����ܱ������еķ�Ӧ��
3SiCl4(g)��2H2(g)��Si(s) 4SiHCl3(g)
4SiHCl3(g)
��ƽ���H2��SiHCl3���ʵ���Ũ�ȷֱ�Ϊ0.140 mol/L��0.020 mol/L����H2ȫ����Դ�����ӽ���Ĥ���ĵ���������������Ĵ�NaCl������Ϊ________kg��
(3)������Ĥ���۵�ⱥ��ʳ��ˮ������ȡ�����ƣ�ͬʱ�������������Ƶ�������213.0 kg������������________m3(��״��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��A��B��λѧ����������ԭ��ط�Ӧ��֤�����Ļ��˳��̽��������й����ʣ��ֱ��������ͼ��ʾ��ԭ��أ�������������⣺
(1)�ٸ������ϣ�
A��________��B��________��
�ڵ缫��Ӧʽ��
A�أ�������________��������________
B�أ�������________��������________
(2)B���ܷ�Ӧ�����ӷ���ʽΪ_________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ѧ������ȤС���ö��Ե缫��ⱥ��ʳ��ˮ��������Ca2�� ��Mg2������ϵ��̽����װ����ͼ��ʾ��
��1�����ʱ����ͬѧ���ֵ缫a������Һ���ֻ��ǣ��������ӷ���ʽ��ʾԭ��________________________________________________________________________��
��2��һ��ʱ�������ΪC����Һ���ܳ��ֵ�������________________________���������ӷ���ʽ��ʾԭ��______________________________________��
��3��ʵ���������ͬѧ��A�е�������ȴ����뵽H2S��Һ�з��������ݳ��֣������뵽ϡ������ȴû���κ��������û�ѧ����ʽ�ͼ�Ҫ�����ֽ���ԭ��________________________________________________________________________________________________________________________________________________��
��4�����ŷ�Ӧ�Ľ��У���ȤС���ͬѧ�Ƕ��ر�ע�D����Һ��ɫ����ȥ�����Ƕ���Һ��ɫ��ȥ����Ҫԭ����������¼��裬������ɼ������
����һ��B���ݳ���������ˮ��Ӧ���ɵ�������ǿ�����ԣ�ʹ��ɫ����ȥ��
�������___________________________________________________��
��5���������ʵ����֤��������һ��д��ʵ�鲽�輰���ۣ�________________________________________________________________________________________________________________________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com