
��16�֣�����Ǧ��PbSO4���㷺Ӧ��������Ǧ���ء���ɫ���ϵȡ����÷�Ǧ��PbS��ֱ���Ʊ�����Ǧ��ĩ���������£�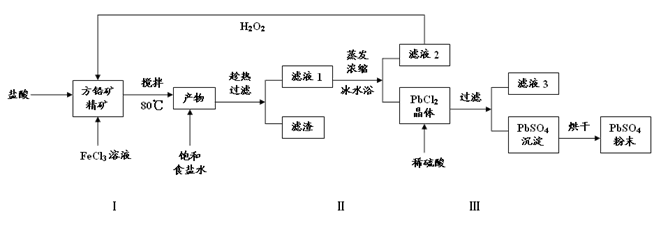
��֪��������PbCl2��s��+2Cl��(aq) PbCl4-(aq) ��H��0
PbCl4-(aq) ��H��0
������Ksp(PbSO4)=1.08��10-8, Ksp(PbCl2)=1.6��10-5
������Fe3����Pb2��������������ʽ��ȫ����ʱ����Һ��PHֵ�ֱ�Ϊ3.2��7.04
��1�������������PbCl2��S�����ӷ���ʽ �������������һ��Ŀ����Ϊ�˿���PHֵ��0.5��1.0��ԭ���� ��
��2���û�ѧƽ���ƶ���ԭ�����Ͳ������ʹ�ñ�ˮԡ��ԭ�� ��
��3��д��PbCl2����ת��ΪPbSO4���������ӷ���ʽ ��
��4���������ӷ���ʽ������Һ2����H2O2��ѭ�����õ�ԭ�� ����Һ3�� ��
��5��Ǧ���صĵ��Һ�����ᣬ���������缫�ϳ����� PbSO4�ֱ�ת��ΪPbO2��Pb�����ʱ�����ĵ缫��ӦʽΪ ��
��16�֣�
��1��PbS+2Fe3��+2Cl��= PbCl2+2Fe2��+S��3�֣�������Fe3����Pb2����ˮ�⣨2 �֣�
��2���ñ�ˮԡʹ��ӦPbCl2��s��+2Cl��(aq) PbCl4��(aq)�����ƶ���ʹPbCl4 ������ת��ΪPbCl2�������������3�֣�
PbCl4��(aq)�����ƶ���ʹPbCl4 ������ת��ΪPbCl2�������������3�֣�
��3��PbCl2��s��+SO42-(aq) PbSO4 (s) +2Cl��(aq) ��2�֣�
PbSO4 (s) +2Cl��(aq) ��2�֣�
��4��2Fe2��+ H2O2+2H��=2Fe3��+2H2O��2�֣������ᣨ2�֣�
��5��PbSO4+2e��="Pb+" SO42����2�֣�
���������������1�������������PbCl2��S��˵����������ᡢ�Ȼ�����PbS����������ԭ��Ӧ��SԪ�ػ��ϼ����ߣ�����Ԫ�صĻ��ϼ۽��ͣ����ӷ���ʽΪPbS+2Fe3��+2Cl��= PbCl2+2Fe2��+S��Fe3����Pb2��������������ʽ��ȫ����ʱ����Һ��PHֵ�ֱ�Ϊ3.2��7.04�����Լ����������һ��Ŀ����Ϊ�˿���PHֵ��0.5��1.0��ԭ��������Fe3����Pb2����ˮ�⣻
��2�����뱥��ʳ��ˮ�����PbCl2��s��+2Cl��(aq) PbCl4-(aq) ��H��0���ڱ�ˮԡ�У��¶Ƚϵͣ�ʹƽ�������ƶ�������PbCl2�����������
PbCl4-(aq) ��H��0���ڱ�ˮԡ�У��¶Ƚϵͣ�ʹƽ�������ƶ�������PbCl2�����������
��3��Ksp(PbSO4)=1.08��10-8< Ksp(PbCl2)=1.6��10-5�����ݳ�����ת����PbCl2����ת��ΪPbSO4��������ΪPbCl2�м���ϡ�����ƻ����Ȼ�Ǧ���ܽ�ƽ�⣬ʹ�ܽ�ƽ�������ƶ���������Ǧ�����ӷ���ʽΪPbCl2��s��+SO42-(aq) PbSO4 (s) +2Cl��(aq)��
PbSO4 (s) +2Cl��(aq)��
��4����Һ2����Ҫ�ɷ����Ȼ���������������ⷴӦ�������Ȼ�������������ʹ�ã����ӷ���ʽΪ2Fe2��+ H2O2+2H��=2Fe3��+2H2O���ɣ�3����֪�Ȼ�Ǧת��Ϊ����Ǧͬʱ�����Ȼ��⣬������Һ3��������Һ��
��5�����ʱ�ǰѵ���ת��Ϊ��ѧ�ܣ��൱�ڵ���װ�ã���������������ԭ��Ӧ��Pb�Ļ��ϼ۽��ͳ�Ϊ����Pb���缫��ӦʽΪPbSO4+2e��="Pb+" SO42��
���㣺��������̵ķ�������ѧƽ���Ӧ�ã��绯ѧԭ����Ӧ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����Һ���ܴ��������һ�����ӻ������
| A��K+ ��Al3+��SO42����NH3��H2O | B��NH4+��H+��NO3����HCO3�� |
C��Na+ ��CH3COO����CO32����OH�� | D��Na+��K+��SO32����Cl2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
������ط�Ӧ�����ӷ���ʽ��д��ȷ����
| A������������������Fe(OH)3+3H+ = Fe3++3H2O |
| B������ͭ��Һ�����ԣ�Cu2+ + 2H2O = Cu(OH)2��+ 2H+ |
C����̼�������Һ�мӹ���ʯ��ˮ�����ȣ�NH4++OH-  NH3��+H2O NH3��+H2O |
| D�����ữ�ĸ��������Һ����˫��ˮ��2MnO4-+6H++5H2O2 = 2Mn2++5O2��+8H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(12��)�ҹ��й����ĺ����ߣ���ˮ�ۺ����ô��п�Ϊ����ˮ���庬��Ϊ65 mg��L��1���Ӻ�ˮ����ȡ��Ĺ�ҵ�������£�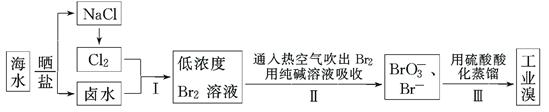
��1�����ϲ�������ѻ������̬���壬������ֽ�֮ת��ɻ���̬���壬��Ŀ���ǣ�
________________________________________________________________________��
��2�������ͨ���ȿ�����ˮ��������Br2�����������________��
A�������� B����ԭ�� C���ӷ��� D����ʴ��
��3���������̢����漰�����ӷ�Ӧ���£��������淽���������ʵ��Ļ�ѧ��������
��4�����������д�������������Ҳ�����ö�������ˮ��Һ���գ�������������������
д�������������ˮ��Һ��Ӧ�����ӷ���ʽ��________________________________��
�ɴ˷�Ӧ��֪�������������⣬�ڹ�ҵ������Ӧ�������Ҫ������ ��
��5��ʵ���ҷ����廹�������ܼ���ȡ�������п������������ȡ������________��
A���Ҵ� B�����Ȼ�̼ C���ѻ����� D����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��6�֣�������з�Ӧ�����ӷ���ʽ��
��1����NaHSO4��Һ����μ���Ba(OH)2��Һ��ʹԭ��Һ�е�SO42��ǡ����ȫ������
��2����������Һ����μ���Ba(OH) ��Һ����Al3+ǡ��ȫ������ʱ��
��Һ����Al3+ǡ��ȫ������ʱ��
��3����Ca(HCO3) 2��Һ�м��������NaOH��Һ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣������ת������Դ���úͻ�����������Ҫ�о����⡣�������������ж��ַ�����
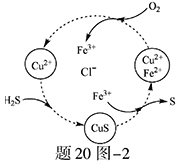
��1�����ռ�����H2S�����Һ���뵽����20ͼ��1��ʾ�ĵ��ص����������е�⡣���������������������·�Ӧ��S2����2e�� S ��n��1��S+ S2��
S ��n��1��S+ S2�� Sn2��
Sn2��
��д�����ʱ�����ĵ缫��Ӧʽ�� ��
�ڵ�������������Һ��ϡ�����ữ�õ����ʣ������ӷ���ʽ��д�� ��
��2����H2S�Ϳ����Ļ������ͨ��FeCl3��FeCl2��CuCl2�Ļ����Һ�з�Ӧ����S��������ת������20ͼ��2��ʾ��
����ͼʾ��ת���У����ϼ۲����Ԫ���� ��
�ڷ�Ӧ�е���1molH2Sת��Ϊ����ʱ��������Һ��Fe3�������ʵ������䣬��Ҫ����O2�����ʵ���Ϊ ��
�����¶�һ���Ͳ�������Һ�������£�����ͨ�������壬����ֽ��衣��ʹ���ɵ������в���CuS���ɲ�ȡ�Ĵ�ʩ�� ��
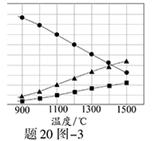
��3��H2S�ڸ����·ֽ�������������H2������Ӧ�ڲ�ͬ�¶��´ﵽƽ��ʱ����������и���ֵ������������20ͼ��3��ʾ��H2S�ڸ����·ֽⷴӦ�Ļ�ѧ����ʽΪ ��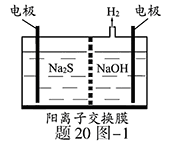
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ɫ��ҺX����Na+��Ag+��Ba2+��Al3+��[Al(OH)4]--�� MnO4����CO32-- ��SO42���е�������������ϣ�ȡ��Һ���������������飺���ѧ���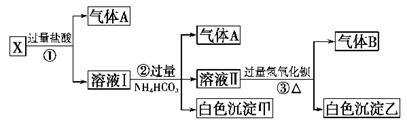
��1������A�ijɷ��ǣ�_________________������B�ijɷ���_____________
��2)X��Һ��һ�����ڵ������ǣ�____________________________
��3)д������ٷ�����Ӧ���������ӷ�Ӧ����ʽ��_________________________
��4��д��������γɰ�ɫ���������ӷ���ʽ��______________________
��5)д����ɫ�����ҵĿ�����ɣ�____________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��2����ѧ�뼼������15�֣�
��Ƴ���ͭ��ˮ�к���CN-��Cr2O72-���ӣ���Ҫ������������ŷš��ó��ⶨ�������̽��з�ˮ�������ش��������⣺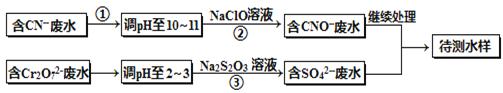
��1������������ˮ��������Ҫʹ�õķ�����_________________��
��2�����з�Ӧ��������ų����÷�Ӧ�����ӷ���ʽΪ______________��
��3��������У�ÿ����0.4mol Cr2O72-ʱת�Ƶ���2.4mol���÷�Ӧ�����ӷ���ʽΪ ��
��4��ȡ��������ˮ�����Թ��У�����NaOH��Һ���۲쵽����ɫ�������ɣ��ټ�Na2S��Һ����ɫ����ת���ɺ�ɫ��������ʹ�û�ѧ��������ֽ��Ͳ����������ԭ�� ��
��5��Ŀǰ��������Cr2O72-��ˮ������������巨���÷������ˮ�м���FeSO4 ��7H2O��Cr2O72-��ԭ��Cr3+������pH��Fe��Crת�����൱�ڣ� ����������,�������ֱ�ʾԪ�ؼ�̬���ij�����
����������,�������ֱ�ʾԪ�ؼ�̬���ij�����
����1mol Cr2O72-�������a mol FeSO4 ? 7H2O�����н�����ȷ����_______��
| A��x ="0.5" ,a ="8" | B��x ="0.5" ,a =" 10" | C��x =" 1.5" ,a =8 | D��x =" 1.5" ,a = 10 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
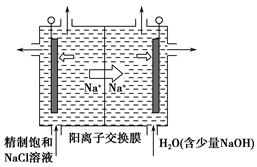
��������(ClO2)Ϊһ�ֻ���ɫ���壬�ǹ����Ϲ��ϵĸ�Ч�����ס����١���ȫ��ɱ����������
(1)��ҵ���Ʊ�ClO2�ķ�Ӧԭ��Ϊ2NaClO3��4HCl=2ClO2����Cl2����2H2O��2NaCl��
��Ũ�����ڷ�Ӧ����ʾ������������________��
| A��ֻ�л�ԭ�� | B����ԭ�Ժ����� |
| C��ֻ�������� | D�������Ժ����� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com