
| A����ȥ�Ҵ���ˮ����������ʯ�ң������ռ������ |
| B��������۵�ˮ������������������ˮ��Һ��ֱ�Ӽ�������Cu(OH)2��Һ��Ȼ����ȣ��۲��Ƿ��к�ɫ�������� |
| C����ȥ���������е�����ӱ���̼������Һ���������Һ����ˮ�� |
| D��������Һ�����ƣ��ڽྻ���Թ��м�2% AgNO3��Һ1��2 mL����μ���2%ϡ��ˮ���ߵα���������ǡ���ܽ�ʱΪֹ |


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| �������� | ������g�� | |
| ��ƿ + NaOH��Һ + ���� | ��1�� | 188.30 |
| ��2�� | 188.28 | |
| ��3�� | 188.25 | |
| ��4�� | 188.24 | |
| ��5�� | 188.24 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
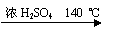 H2O + CH3CH2��O��CH2CH3 (����)
H2O + CH3CH2��O��CH2CH3 (����)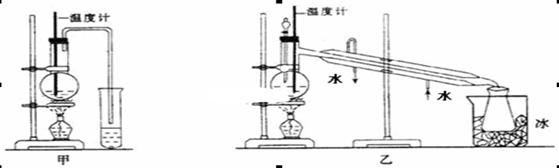
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| ���� | ʽ�� | ��״ | �ܶ�/g/cm3 | �е�/�� | �ܽ�� | |
| �� �� | 93 | ��ɫ��״Һ�壬 ���л�ԭ�� | 1.02 | 184.4 | ����ˮ | �������Ҵ������ѵ� |
| �� �� | 60 | ��ɫҺ�� | 1.05 | 118.1 | ������ˮ | �������Ҵ������� |
| �������� | 135 | ��ɫ���� | 1.22 | 304 | ������ˮ��������ˮ | �������Ҵ������� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
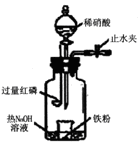
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
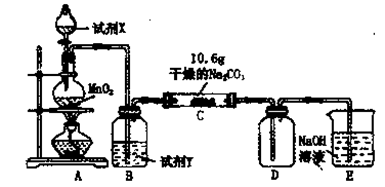
 ��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ��
��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ȡ�ķ����ɽ����ͺ�ú�ͽ��з��� |
| B���ӹ���ϡ�������ˣ��ɳ�ȥ����ͭ���е�����þ�ۺ����� |
| C�����ܽ⡢���˵ķ����ɷ�������غ��Ȼ��ƹ������� |
| D������������������ͨ�����ȵ�����ͭ���Գ�ȥ���е����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
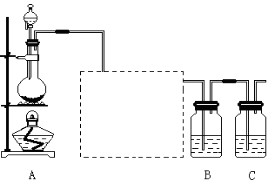
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
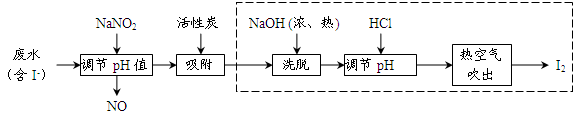
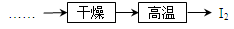
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com