
| ��� | c��NaOH��/mol?L-1 | ʱ��/min | |||||||
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | ||
| 1 | 0.5 | 10.0 | 9.0 | 8.0 | 7.5 | 7.0 | 6.5 | 6.5 | 6.5 |
| 2 | 1.0 | 10.0 | 8.5 | 7.0 | 6.0 | 5.0 | 4.5 | 4.5 | 4.5 |
| 3 | 2.0 | 10.0 | 8.0 | 6.0 | 4.5 | 3.0 | 2.0 | 1.5 | 1.5 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ��� | C��NaOH��/mol?L-1 | ʱ��/min | ||||||||
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | ||
| 1 | 0.5 | 10.0 | 9.0 | 8.0 | 7.5 | 7.0 | 6.5 | 6.5 | 6.5 | 6.5 |
| 2 | 1.0 | 10.0 | 8.5 | 7.0 | 6.0 | 5.0 | 4.5 | 4.5 | 4.5 | 4.5 |
| 3 | 2.0 | 10.0 | 8.0 | 6.0 | 4.5 | 3.0 | 2.0 | 1.5 | 1.5 | 1.5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�





�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
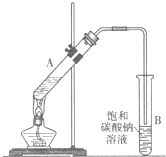 ������������Ҫ�Ļ���ԭ�ϣ�ʵ���Һϳ�����������װ����ͼ��ʾ��
������������Ҫ�Ļ���ԭ�ϣ�ʵ���Һϳ�����������װ����ͼ��ʾ��| ���� | �Ҵ� | �������� | ���� | |
| �е�/�� | 118 | 78.3 | 77.1 | 34.5 |
| �ܽ��� | ������ˮ | ��������ˮ | �����ѻ��� | ����ˮ |
| Ũ���� |
| ��140 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ�人�и������µ��в��������ۺϻ�ѧ�Ծ��������棩 ���ͣ�ʵ����
������������Ҫ�Ļ���ԭ�ϡ�ʵ���Һϳ�����������װ������ͼ��ʾ��

�й����ݼ�����Ӧ:

����Ӧ��
C2H5OH+C2H5OH C2H5OC2H5+H2O
C2H5OC2H5+H2O
��ش��������⣺
��1���ڴ��Թ�A�����ӵ��Լ���6 mL�Ҵ���4 mL�����4 mLŨ����,�������Լ�������˳������Ϊ _______��_______ ��_______
��2���Թ�B�е��ܽӽ�Һ��δ����Һ���µ������� _______
��3���ֶ��Թ�B�����������ֲ�Ʒ�����ᴿ���������£�
�ٽ��Թ�B�л��Һ������,ת�� _______(����������)���з���;
�����������ϲ�Һ���м�����ˮ�����ƣ������������ˮ�����Ƶ�Ŀ���ǣ�
�۽���������������Һ���������������ƿ�У�������������ռ�_______0C���ҵ�Һ�弴�ô���������������
��4������ɫ��ѧ�ĽǶȷ�����ʹ��Ũ������������������֮����Ҫ��____________________
��5������ʱ����һ��ƺʹ���ʹ��������ɿڣ����÷���ʵ������Ļ�ѧ����ʽ����: ___ _______________
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com