
2012��3��21 ���ǵڶ�ʮ�조����ˮ�ա�������ˮ��Դ����������
(1)ClO2��Cl2(��ԭ���ﶼΪCl��)�������г��õ��������������ĵ����ʵ�������������ʱ��ClO2������Ч����Cl2��________����
(2)ij��ɫ��ˮ�п��ܺ���Fe3����Al3����Mg2����Na����NO3����CO32����SO42�������еļ��֣�Ϊ������ɷ֣��ֱ�ȡ��ˮ��Ʒ100 mL������������ʵ�飬��������й���������ͼ��ʾ��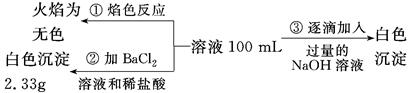
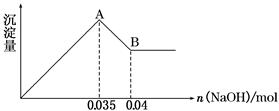
�������ͼ�ش��������⣺
��ʵ����������1.0 mol/L��NaOH��Һ100 mL�������������˲�������������ƽ����Ͳ��ҩ�ס��ձ�����ͷ�ιܣ���ȱ�ٵ�����Ϊ_________________________________________��
��ʵ����г�������A��B��������������Ӧ�����ӷ���ʽΪ____________________��
����ȷ��NO3���Ƿ���ڣ�________(����ڡ����������ڡ���ȷ����)�������ڣ��Լ���c(NO3��)��________(�������ڣ����ʲ�������)��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪һ������������������ȼ�գ����û������100 mL 3��00 mol/L��NaOH��Һ���ܶ�Ϊ1��12 g/mL��ǡ����ȫ���գ������Һ�к���NaClO�����ʵ���Ϊ0��0500 mol��
��1��ԭNaOH��Һ����������Ϊ ��
��2��������Һ��Cl�������ʵ���Ϊ mol��
��3�����������Ͳμӷ�Ӧ�����������ʵ���֮��n(Cl2)��n(H2)�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)��ͬ�¡�ͬѹ�£�ʵ����CO��N2��O2��������Ļ��������ܶ���H2��14.5��������O2����������Ϊ ��������CO��N2�����ʵ���֮��Ϊ1��1��������������Ԫ�ص���������Ϊ ��
(2)��ͬ�����£�ijCl2��O2�������100 mLǡ����150 mL H2��������HCl��H2O������������Cl2��O2�������Ϊ ����������ƽ����Է�������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ͭ����Ͻ�����������ʹ�õĽ������ϡ�
(1)����ͭ��ȡ�������ַ�ʽ�ѻ�(����)

(2)��1��Cu2O������(�ṹ����ͼ��ʾ)��Cuԭ����λ��Ϊ__________��
(3)��ѧ��ͨ��X�����Ʋ���мȺ�����λ�����ֺ����������ṹʾ��ͼ�ɼ�ʾ���£�
�ٵ����Ļ�ѧʽ����������ʽ��ʾΪ____________��
�ڵ�����SO42���Ŀռ乹��Ϊ________��H2O��Oԭ�ӵ��ӻ�����Ϊ________��
��ij��ȤС���ȡ2.500 g�������壬������ʹ��ʧˮ����ȷ�ⶨ��ͬ�¶���ʣ�������������õ���ͼ��ʾ��ʵ����ʾ��ͼ������˵����ȷ����(����)
| A������ӳ�������105 ��Ĺ�����ֻ��������� |
| B�������������γ���λ����4��ˮ����ͬʱʧȥ |
| C��120 ��ʱ��ʣ�����Ļ�ѧʽ��CuSO4��H2O |
| D������������ʧˮʱ���˷�����������С��ͬ�������е�ˮ���ӿ��Է�Ϊ3�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����Ĺ�ҵ�Ʊ���һ����Ҫ�Ļ����������̣��������������л����������Ⱦ����Ҫ�����������п��ǵ���ɫ���ա�
Iβ�������պ��ۺ����á�
�Թ�ҵ�������β������ˮ��ʯ��ʯ����̿��̼����狀�KCIΪԭ�Ͽ��Ժϳ��ơ�����ء�������淋����ʡ��ϳ�·�����£�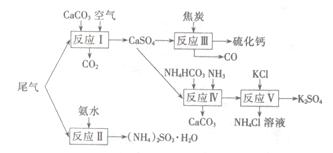
��1����ӦIII���������뻹ԭ�������ʵ���֮��Ϊ ��
��2����Ӧ���Ļ�ѧ����ʽΪ ��
��3����ӦV��25�桢40%���Ҷ�����Һ�н��У��÷�Ӧ��˳�����е�ԭ��Ϊ ��
������Ļ������á�
SO2�Ĵ�������ʹ�õĴ���ΪV2O5��ʵ�������У�������ʹ��һ��ʱ��Ậ��V2O5��VOSO4��SiO2�ȣ�����VOSO4��������ˮ������V2O5������Ҫ�������£�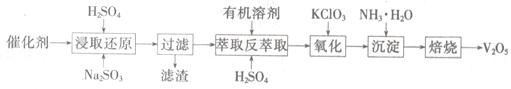
��4��������ȡʹ�õ�������������һ������ʱ������____ ��������
��5����ȡ��ԭ���̵IJ���֮һ��VOSO4����Ӧ�Ļ�ѧ����ʽΪ ��
�������̵Ļ�ѧ����ʽΪKClO3+6VOSO4+3H2SO4= 2(VO)2(SO4)3+KCl+3H2O�������������Լ�Na2SO3��KC1O3�����ʵ���֮��Ϊ12��7����ô�����V2O5��VOSO4�����ʵ���֮��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ϩ������Ļ�����干a mol����b mol O2������һ�ܱ������У���ȼ���ַ�Ӧ����ϩ������ȫ�������꣬�õ�CO��CO2�Ļ�������45 g H2O������
��1����a��1ʱ����ϩ����������ʵ���֮��n��C2H4����n��C2H6���� ��
��2����a��1���ҷ�Ӧ��CO��CO2�Ļ����������ʵ���Ϊ��ӦǰO2��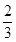 ʱ��b�� ���õ���CO��CO2�����ʵ���֮��n��CO����n��CO2���� ��
ʱ��b�� ���õ���CO��CO2�����ʵ���֮��n��CO����n��CO2���� ��
��3��a��ȡֵ��Χ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Դ����Ľ�һ��ͻ���������Ȼ�ѧѭ��������о��ܵ��������ҵ�������������о����֣�����������������(MnFe2O4)Ҳ���������Ȼ�ѧѭ���ֽ�ˮ���⣬MnFe2O4���Ʊ��������£�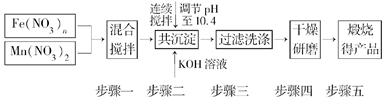
(1)ԭ��Fe(NO3)n��n��________��Ͷ��ԭ��Fe(NO3)n��Mn(NO3)2�����ʵ���֮��ӦΪ________��
(2)������С��������衱��Ŀ����__________________________________________
��������ϴ�Ӹɾ��ı���________________________________________________
(3)����MnFe2O4�Ȼ�ѧѭ������ķ�Ӧ�ɱ�ʾΪ��
MnFe2O4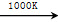 MnFe2O4��x��O2����
MnFe2O4��x��O2����
MnFe2O4��x��xH2O MnFe2O4��xH2��
MnFe2O4��xH2��
�������������������Ӧ���ش��������⣺
����MnFe2O4��x��x��0.8����MnFe2O4��x��Fe2��ռȫ����Ԫ�صİٷ���Ϊ________��
�ڸ��Ȼ�ѧѭ�����ⷨ���ŵ���_____________________��________________________ (�����㼴��)��
���Ȼ�ѧѭ�����������в���֮������һ���Ľ����о�������___________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��.ʵ��������1mol/L Na2CO3��Һ250ml��
��1����Ҫ����Na2CO3 g����2������Һ�е���������ĿΪ ����
��3����Ҫ���ʵ���Ũ��Ϊ5mol/L ��Na2CO3��Һ ml��
��4��������Һ������ϡ���ᷴӦ�������������ڱ�״���µ����Ϊ L��
��5�����Ƹ���Һ�IJ���˳����(����ĸ��ʾ,���ظ�ʹ��) ��
| A������ | B��ϴ�� | C������ | D���ܽ� E��ҡ�� F��ת�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��11.2g��Ͷ��200mLijŨ�ȵ������У���������ǡ����ȫ��Ӧ����
��1������������HCl�����ʵ���Ũ��
��2����Ӧ�����ɵ�H2�ڱ�״���µ����
��3���ڷ�Ӧ�����Һ��ͨ��Cl2��д����������Ӧ�Ļ�ѧ����ʽ����˫���ű������ת�Ƶķ������Ŀ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com