
���¶ȸ���40��ʱ��ͭ��ƽ���ܽ��������ŷ�Ӧ�¶����߶��½�������Ҫԭ���� ��
�����ᴿ���CuSO4��Һ�м���һ������Na2SO3��NaCl��Һ�����ȣ�����CuCl�������Ʊ�CuCl�����ӷ���ʽ�� ��
28. (9��)����һ�仰������������ֵ��ǰ��ΪM������ΪN��M��N�Ĺ�ϵ��A��B��C��D��ѡ��
A. M>N B.M<N C.M=N D. ���Ƚ�
����ͬ�¶��£�1L 1mol/L ��NH4Cl��Һ�е�NH4��������2 L 0.5mol��L��1NH4Cl��Һ��NH4���ĸ����� ��
����ͬ�¶��£�pHֵΪ12���ռ���Һ��ˮ�ĵ���Ⱥ�pHֵΪ12��CH3COONa��Һ��ˮ�ĵ���ȣ� ��
����������ʱ�ı���ʯ��ˮ��һ�����µ�50�棻��һ�ݼ�������CaO���ָ������£�����Һ�е�c(Ca2+)�� ��
�ȳ��������ݵ�Ũ�ȵĴ�����Һ�����ڶ��������¶ȣ�����Һ��c(HCO3��)�� ��
�ɽ�pHֵΪ2�Ĵ�������ᶼϡ����ͬ��������ϡ��Һ��pHֵ�� ��
�ʳ�����0.1mol/L��CH3COOH��0.1mol/LCH3COONa�������Ϻ���Һ��c(Na+)��c(CH3COO��)�� ��
��ͬ�¶��£�0.1mol/LFeCl3��Һ��Fe3+ˮ��ٷ�����0.01mol��L��1FeCl3��Һ��Fe3+ ��ˮ��ٷ��ʣ� ��
��������ijǿ���ijǿ����Һ�������Ϻ���Һ��pHֵΪ7��ԭ����Һ��ԭ����Һ�����ʵ���Ũ�ȣ� ��
��PHֵ��ͬ�Ĵ�������ᣬ�ֱ�������ˮϡ����ԭ����M����N����ϡ�ͺ�����Һ��PHֵ��Ȼ��ͬ�� ��M��N�Ĺ�ϵ�ǣ� ��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ���� |
| �� |
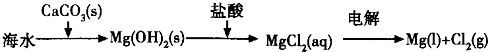
| ||
| ||
| ||
| ||
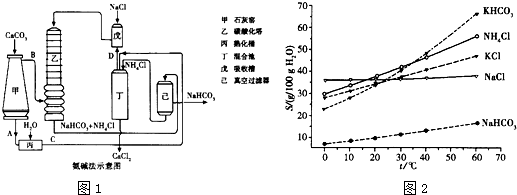
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| ͭƽ���ܽ����� ����10-3mol?L-1?min-1�� |
7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| ͭƽ���ܽ����� ����10-3 mol?L-1?min-1�� | 7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| 17Cv |
| 200�� |
| 17Cv |
| 200�� |
| ʵ������ | ʵ�鷽�� | ʵ������ |
| ��֤������ | ȡ�����⻯�ص�����Һ��ϡ�������Թ��У��������������Һ������ȡ��������������Һ���Թ��У��������������Һ���� | ��Һ����ɫ ��Һ����ɫ ������ ��������ɫ��������Һ����� ��������ɫ��������Һ����� �� |
| ��֤�Ȳ��ȶ��� | ȡ��������������Һ���Թ��У� ������ ������ �ô����ǵ�ľ������ �ô����ǵ�ľ������ �� |
�������ݣ������ǵ�ľ����ȼ�� |
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| ͭƽ���ܽ����� ����10-3mol?L-1?min-1�� |
7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
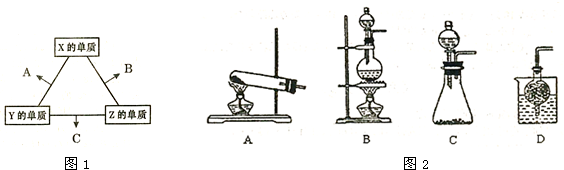
| �¶ȣ��棩 | 20 | 30 | 40 | 50 | 60 | 70 | 80 |
| ͭ��ƽ���ܽ����� ����10-3mol-L-1?min-1�� |
7.34 | 8.01 | 9.25 | 7.98 | 7.24 | 6.73 | 5.76 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com