
ij�����ĵ�������к���ͭ�����Ƚ��������Ϊʵ����Դ�Ļ������ò���Ч��ֹ������Ⱦ��������¹������̣�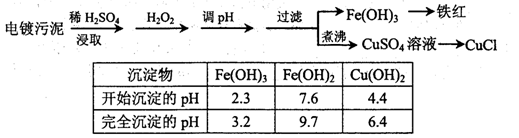
��1����������H2O2��Ŀ���� ����pH�����м�����Լ������ ���ѧʽ����ʵ���ҽ��й��˲������õ��IJ��������� ��
��2�����CuSO4��Һ��ԭ���� ����CuSO4��Һ�м���һ������NaCl��Na2SO3���������ɰ�ɫ��CuCl������д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3��CuCl��Ʒ��CuCl��������������96��50%Ϊ���Һϸ������ȡ���Ʊ���CuCl��Ʒ0��2500g����һ������0��5mol��L-1FeCl3��Һ�У�����Ʒ��ȫ�ܽ��ˮ20mL����0.1000mol��L-1��Ce��SO4��2��Һ�ζ��������յ�ʱ����Ce��SO4��2��Һ24��60mL���йصĻ�ѧ��ӦΪ��
Fe3����CuCl��Fe2����Cu2����Cl����Ce4����Fe2����Fe3����Ce3����ͨ������˵����CuCl��Ʒ ������ϡ������ϡ������ұ���
��4��25��ʱ��KSP [Fe��OH��3]= 4.0��10-38��Fe3+����ˮ�ⷴӦFe3����3H2O Fe(OH)3��3H�����÷�Ӧ��ƽ�ⳣ��Ϊ ��
Fe(OH)3��3H�����÷�Ӧ��ƽ�ⳣ��Ϊ ��
��14�֣���1����Fe2+������Fe3�������ڵ���pHֵ��Cu2+���루2�֣���
CuO��Cu(OH)2��CuCO3�ȣ�2�֣���©�����ձ�����������2�֣�
��2��������Һ�е�H2O2������Ӱ����һ��CuCl�����ɣ�2�֣�
2CuSO4��2NaCl��Na2SO3��H2O��2CuCl����2Na2SO4��H2SO4��2�֣�
��3�����ϣ�2�֣� ��4��2.5��10��5��2�֣�
���������������1����������к���ͭ�����Ƚ���������������Ҫ���ܽ�ͭ�����Ƚ��������˫��ˮ��ǿ�����ԣ���������ԭ�Ե����ʣ�Fe2+���л�ԭ�ԣ���������H2O2��Fe2+�ܱ�˫��ˮ����ΪFe3�������ڵ���pHֵ��Cu2+���룻�ڵ���pH�����м�����Լ���������ȥ��Һ�е��ᣬ�Ҳ��������µ����ʣ�����Ҫ������Լ�������CuO��Cu(OH)2��CuCO3�����ݱ������ݿ�֪������Һ��pHֵ4.4ʱ��ͭ���ӿ�ʼ���ֳ���������Һ��pHֵΪ3.2ʱ�����������ӳ�����ȫ��ͭ����δ��������������Ҫʹ���������Ӻ�ͭ���ӷ��룬��Һ��pHӦ����3.2��pH��4.4.���ɵ�����������������ͨ������ʵ�ַ��룬���˲��õ�������������̨��©�����ձ����������ȣ��������ڲ���������©�����ձ�����������
��2����Ӧ�й��������ǹ����ģ�����������������ԣ��������ȥ��Ӱ��CuCl�����ɡ�˫��ˮ�����ֽ⣬�������CuSO4��Һ��ԭ���dz�����Һ�е�H2O2������Ӱ����һ��CuCl�����ɣ���ΪCuSO4�У�2�۵�ͭ�ܰ�Na2SO3�У�4�۵��������ɣ�6�۵�����2�۵�ͭ����ԭ���ɣ�1�۵�ͭ����������CuCl���������Ը÷�Ӧ�Ļ�ѧ����ʽΪ2CuSO4��2NaCl��Na2SO3��H2O��2CuCl����2Na2SO4��H2SO4��
��3������Ʒ��CuCl������Ϊx���йصĻ�ѧ��ӦΪFe3����CuCl��Fe2����Cu2����Cl����Ce4����Fe2����Fe3����Ce3�������ɻ�ѧ��Ӧ����ʽ��֪��CuCl������Fe2+������Ce4+
1 1
n(CuCl) 24.60��10-3L��0.1000 mol/L
��� n(CuCl)��24.60��10-3L��0.1000 mol/L��2.46��10-3mol
���Ը���ƷCuCl������Ϊ2.46��10-3mol��99.5g/mol��0.2448g
������Ʒ��CuCl������������ ��100%��97.91%��96.50%�����Ը���Ʒ��CuCl�������������ϱ���
��100%��97.91%��96.50%�����Ը���Ʒ��CuCl�������������ϱ���
��4��KSP [Fe(OH)3]��c(Fe3+)��c3(OH-)��4.0��10-38��c(H+)��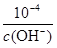 ����˷�ӦFe3����3 H2O
����˷�ӦFe3����3 H2O Fe(OH)3��3H����ƽ�ⳣ��K��
Fe(OH)3��3H����ƽ�ⳣ��K��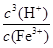 ��
��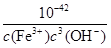 ��2.5��10-5��
��2.5��10-5��
���㣺�����Լ���ѡ��Ӧ�����Ŀ�֪��������ѡ��������ԭ��Ӧ�͵���ƽ�ⳣ���ļ����Լ��йط���ʽ����д��


 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ӹ�������A�������Է������¿�ͼ��ʾ��һϵ�б仯��

��1����ʵ�����г��÷�Ӧ����ȡ����C����Ҫ�ռ�����C����ѡ����ͼװ���е� ������ĸ��
A B C
��2�������ڵ������� ���ڲ���������ʹ�õIJ��������������� ��
��3��д�����з�Ӧ�ķ���ʽ��
��Ӧ�ٵĻ�ѧ����ʽ
��Ӧ�۵Ļ�ѧ����ʽ
��4����ʵ�����У�Ҫ��ø��﴿���Ļ���ɫ����F�� ���Խ���ͨ����ͼ�е�װ�ã�����ƿI��ʢ�ŵ�
�� ��ƿII��ʢ�ŵ��� ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ѡ���������ʷ���ͼ��鷽��������ѡ�����ں����ϡ�
| A����ȡ�� | B�������� | C���ᾧ�� | D����Һ��E������F��������G������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ˮ�Ǿ����Դ���⡣��ͼ������Ӻ�ˮ��Դ��ȡijЩ��Ҫ����ԭ�ϵ�����ʾ��ͼ��

�ش��������⣺
��1������A��_________________(��ʵ�������������)���ú��ַ���֪����ˮɹ�εĹ�������Һ��ʳ�κ���������ߣ�________
a�������Ȼ��ƺ��� b���ⶨ��Һ�ܶ� c���۲��Ƿ��г�������
��2������B����������Լ��е�һ�֣�����ʵ���______��ѡ���ţ���
a������������Һ b������ʯ��ˮ c��ʯ���� d��̼������Һ
��3�����±��ͨ��Cl2��������Ӧ�����ӷ���ʽ��____________________________������C��_______________________________��
��4����ͼ�����߿������̵���Ҫ������_______________________����֮Ŀ����ͬ����������ͼ�л��м�������ָ������һ��________________________��
��5����MgCl2ת��ΪMgʱ��õĸ���Ʒ��_____________���û�ѧ����ʽ��ʾ�����ʵ�ij����;______________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������������A��B��C��D��E�����ǵ������ӿ�����Na����NH4+��Cu2����Ba2����Al3����Ag����Fe3���������ӿ�����Cl����NO3-��SO42-��CO32-����֪��
�������ξ�����ˮ��ˮ��Һ��Ϊ��ɫ��
��D����ɫ��Ӧ�ʻ�ɫ��
��A����Һ�����ԣ�B��C��E����Һ�����ԣ�D����Һ�ʼ��ԡ�
�������������ε���Һ�зֱ����Ba(NO3)2��Һ��ֻ��A��C����Һ������������
�������������ε���Һ�У��ֱ���백ˮ��E��C����Һ�����ɳ����������Ӱ�ˮ��C�г�����ʧ��
�ް�A����Һ�ֱ���뵽B��C��E����Һ�У��������ɲ�����ϡ����ij�����
��ش��������⣺
��1���������У�һ��û�е��������� ��������������ͬ�������εĻ�ѧʽ�� ��
��2��D�Ļ�ѧʽΪ ��D��Һ�Լ��Ե�ԭ����(�����ӷ���ʽ��ʾ) ��
��3��E�Ͱ�ˮ��Ӧ�����ӷ���ʽ�� ��
��4�����ʵ�����B�������������ӣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
����������������Ŀǰ����ˮ���塱������Ҫ����֮һ���乤���������£�
��1��Ŀǰ���Ӻ�ˮ����ȡ����Լռ������������� ��
��2����������������ữ�����Cl2��������,��ԭ���� ��
��3������ܵ����ӷ���ʽ��
��4���������������У�������¶�Ҫ������80?90�档�¶ȹ�����Ͷ�����������,������ ��
��5���������������������õ�Һ������ˮ�Ļ������������ǵ�����ܶ����ϴ���ص���з��롣���������������� �����논Һ��ӷ������� (��Ͽڡ����¿ڡ�)�ų���
��6����ֱ���ú���ĺ�ˮ��������õ�Һ�壬��Ҫ����������������SO2���ա��Ȼ�����ԭ���� ��
��7��ijͬѧ��ÿ�±����ĺ���Ϊ0.8g��L-1����֪�����?�����干��ʧ��25%������ߺͲ�����ֹ���ʧ��������������10%��������10 m3�����Ŀ�±���ɵõ�Һ�� mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
(4��)����(����������)
| ��� | ���� | �����Լ� |
| ��1�� | CO2��HCl�� | |
| ��2�� | Fe2O3��Al2O3�� | |
| ��3�� | NaCl��Һ��MgCl2�� | |
| ��4�� | NO���壨NO2�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��12�֣��ߴ�������[��ѧʽ��Sr(NO3)2]���������źŵơ���ѧ�����ȡ�
�Ź�ҵ���������г���������ơ����ᱵ�����ʣ���������ƿ�����Ũ���ᣬ�������ȡ����ᱵ������Ũ���ᡣ���������Ϣ�����������ᴿ�����ȵ�����ʵ�鲽�裺
��ȡ�����ʵ���������Ʒ�� �����衣
�� ��
�۽���������ˮ�У����Թ�������ʹBa2+���������ú�����£�N2H4�����������ỹԭ������pH=7��8�����ˡ�
�ܽ���Һ���������pH=2��3�� �����ˣ�ϴ�ӡ�
�ݽ��õ���Sr(NO3)2��2H2O������100 �������¸���õ��ߴ������ȡ�
��Sr(NO3)2�����ֽ⣬����Sr(NO2)2��O2����500 ��ʱSr(NO2)2��һ���ֽ�����SrO���������ȡһ��������Sr(NO2)2��Sr(NO3)2��Ʒ����������ȫ�ֽ⣬�õ�5.20 g SrO�����5.08 g������塣�������Ʒ��Sr(NO3)2������������д��������̣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����е�Ħ����һ����CaCO3��Al(OH)3��SiO2��ɡ�ijС��Լ���������Ħ�����ɷּ��京������̽����
��1������������Ϣ���Ʋ�Ħ����Ӧ�߱���������
A��������ˮ B��������ˮ C�������ϴ� D��������С
��2����֪A�������Ħ������Al(OH)3��ȡ����������Ʒ����ˮ��ֽ��衢���ˣ�
���������м������NaOH��Һ��Al(OH)3��NaOH��Һ��Ӧ�����ӷ���ʽ____ __��
������������Һ��ͨ�����CO2���ټ������ϡ���ᣬ�۲쵽�������� ��
��3����֪B��������Ħ�������ܺ���һ���������ʣ����������Ħ������������ʱ�������в��������ʡ�
�� ��BƷ������Ħ�����ijɷ�����������裺
����1��ֻ��SiO2 ����2��������SiO2��CaCO3
����3��������________________��
�� ����Ʒ�����BƷ������Ħ�����ijɷֽ�����֤����д�±�����ѡ�Լ���ϡ���ᡢϡ���ᡢNaOH��Һ������ʯ��ˮ
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ����������Ʒ����ˮ�ɷֽ��衢���ˣ�����ҺA�ͳ���B�� | |
| ����2�� | |
| ����3�� | |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com