
�п�Ժ��ѧ�о������Ƶľ���������������������������ں˴Ź�����Ӱ��ҽҩ���й㷺����;���䲿������������ͼ��ʾ��
 ��������������
��������������
�����й���������ȷ����(����)��
A�����������������ɷ�ɢ��ˮ�У�����FeCl3��Һ�з�ɢ�ʵ�ֱ���൱
B�������������������д��ԣ�����Ϊҩ�������������Ƽ���
C���ڷ�Ӧ���л����������ÿɴٽ��Ȼ���ˮ��
D����Ӧ���Ļ�ѧ����ʽ��6FeOOH��CO=2Fe3O4��3H2O��CO2
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��7��ˮ��Һ�е�����ƽ����ϰ���������棩 ���ͣ�ѡ����
�����йص������Һ��˵����ȷ����(����)��
A��������ˮ�еμ�ŨH2SO4��KW����
B��CaCO3������ϡ���ᣬҲ�����ڴ���
C����Na2Sϡ��Һ�У�c(H��)��c(OH��)��2c(H2S)��c(HS��)
D��NaCl��Һ��CH3COONH4��Һ�������ԣ�����Һ��ˮ�ĵ���̶���ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��3��������Ҫ�Ļ�ѧ��Ӧ��ϰ���������棩 ���ͣ������
ij�������Թ�ҵ��ˮ�к���һ������Na����Al3���� Fe3����Cu2����Cl�����ó�������ͼ��ʾ�Ĺ�������ͼ�����ó���������������ᡢ���ҵ�����еķ���м���ӷ�ˮ�����������Ȼ�������������NaCl����ͽ���ͭ�������˺ܺõ���ᾭ��Ч�档

����д���пհף�
(1)ͼ���Լ�1��________���Լ�2��________��
(2)����1�Ͳ���2���õ��IJ���������________��
(3)����1��Ӧ�����ӷ���ʽΪ_______________________________________��
(4)����3��Ӧ�����ӷ���ʽΪ_______________________________________��
(5)�ӽ�ԼҩƷ�ͻ������濼�ǣ�����5��������Ӧ�����ӷ���ʽӦΪ__________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��2����ѧ���ü�����ϰ���������棩 ���ͣ�ѡ����
�ù�����Ʒ����һ�����ʵ���Ũ�ȵ���Һ���辭���������ܽ⡢ת����Һ�����ݵȲ���������ͼʾ��Ӧ�IJ����淶����(����)��
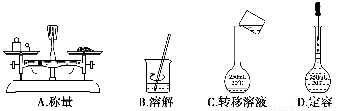
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��1�����ʵ�������ʺͷ�����ϰ���������棩 ���ͣ������
���෨��ѧϰ��ѧ����Ҫ��������������ʶ����ʱ�ɲ��ö��ַ�������±��������
������������
AFeSO4��NO2��MnO2��NaClO��Cu3P��Na2O2H2SO3
BCH3COOH��HOOC��COOH��HClO��H2SHF
C���ֽⷴӦ����ⷴӦ�����ȷ�Ӧ�����ӷ�Ӧ��ɫ��Ӧ
D(NH4)2SO4��NH4Cl��NH4NO3��NH3��H2ONH4HCO3
(1)A�з������_____________________��H2SO3________(����������������)�ɹ�Ϊ���ࡣ
(2)B�з������_________________________��HF________(����������������)�ɹ�Ϊ���ࡣ
(3)C�з������__________________����ɫ��Ӧ����________�仯��
(4)D�з������__________________��NH4HCO3________(����������������)�ɹ�Ϊ���ࡣ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��15��ʵ�黯ѧ��ϰ���������棩 ���ͣ�ʵ����
������������(FeSO4��7H2O)��ҽҩ������Ѫ����Ϊ�ⶨ��Ѫ������Ԫ�صĺ�����ij��ѧ��ȤС�����������ʵ�鷽����
����һ���ζ�����������KMnO4��Һ�ζ��ⶨ��Ԫ�صĺ�����
��Ӧԭ����5Fe2����MnO4����8H��===5Fe3����Mn2����4H2O
(1)ʵ��ǰ������Ҫ��ȷ����һ�����ʵ���Ũ�ȵ�KMnO4��Һ250 mL������ʱ��Ҫ����������ƽ�����������ձ�����ͷ�ι��⣬����________(����������)��
(2)����ʵ����KMnO4��Һ��Ҫ�ữ�������ữ������________��
A��ϡ���ᡡ��B��Ũ���ᡡ��C��ϡ���ᡡ��D��ϡ����
(3)ijͬѧ��Ƶ����еζ���ʽ�У����������________(�гֲ�����ȥ)(����ĸ���)��

���������������������������£�

(4)�������г���H2O2�����ʹ�õ�������__________________________��
(5)�������Ƿ����ʡ��________��������________________________________
_______________________________________��
(6)��������һϵ�в��������ǣ����ˡ�ϴ�ӡ�________����ȴ��������
(7)����ʵ������ģ���ÿƬ��Ѫ������Ԫ�ص�����________g(�ú�a�Ĵ���ʽ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��14�����ʽṹ��������ϰ���������棩 ���ͣ������
��.���л������У����зǼ��Թ��ۼ������ӻ�������(����)��
A��CaC2 B��N2H4 C��Na2S2 D��NH4NO3
��.ͼA��ʾ��ת����ϵ��(���巴Ӧ������)��a��b��c��d�ֱ�Ϊ���ֶ�����Ԫ�صij������ʣ������Ϊ���ǵĻ����i����ҺΪ�������ᣬa��һ��ͬ��������ľ�����ͼB��ʾ��

�ش��������⣺
(1)ͼB��Ӧ������������________���侧���е�ԭ����Ϊ________����������Ϊ________��
(2)d��Ԫ�ص�ԭ�Ӻ�������Ų�ʽΪ________��
(3)ͼA���ɶ���Ԫ����ɵ������У��е���ߵ���________��ԭ����________________________________________________________________________�������ʵķ��ӹ���Ϊ________������ԭ�ӵ��ӻ��������Ϊ________��
(4)ͼA�е�˫ԭ�ӷ����У��������ķ�����________��
(5)k�ķ���ʽΪ________������ԭ�ӵ��ӻ��������Ϊ________������________����(���������������Ǽ�����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��12����ѧʵ�������ϰ���������棩 ���ͣ�ѡ����
����ʵ���������ɻ�ʵ����ۺ�������(����)��
A��֤��һƿ����ɫ���������������Ƕ�������������ʪ��ĵ⻯��?������ֽ���飬�۲���ֽ��ɫ�ı仯
B�����ȼ���������ϡ�����ٵμ�KSCN��Һ��δ����Ѫ��ɫ�������ȼ�����������
C������ˮ��pH���ò�����պȡ��ˮ����pH��ֽ�ϣ������ɫ��ͱ���ɫ���Ƚ�
D��������������ͭ����Һ���Լ�����ᡢ������������ᡢ����������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ���ѵ��ר��11ʵ������ʹ�ü�����������ϰ���������棩 ���ͣ�ѡ����
����֧�Թ��зֱ����������ͬ����ɫ��Һ�������²�����������ȷ���ǣ���������
ѡ���١����֡����ᡡ��
Aij������ʪ��ĵ��۵⻯����ֽ��ֽ����������һ��������
B�μ���ˮ�ͱ����������ϲ���Һ����ɫԭ��Һ����I��
C�ýྻ��˿պȡ������Һ������ɫ��Ӧ����ʻ�ɫԭ��Һһ����������Һ
D�μ�BaCl2��Һ����������ϡ����İ�ɫ����ԭ��Һһ������Ag��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com