
(��)����ѡ����
����˵������ȷ����________��
A����ϩ��������8���Ҽ���1���м�
B����SiO2�����У�1��Siԭ�Ӻ�2��Oԭ���γ�2�����ۼ�
C��NF3�ķе��NH3�ķе�͵ö࣬����ΪNH3���Ӽ��������NF3ֻ�з��»���
D��NCl3��BC13�����У�����ԭ�Ӷ����� sp3�ӻ�
E��SO3��CO32����Ϊ�ȵ����壬SO3�Ǽ��Է���
(��)������ʹ�ý�������ʷ�����У�������ͭ��������֮�����ֽ����㷺Ӧ�õĽ�������ѧ��Ԥ������(Ti)��������Ϊ��δ�����͵Ľ��������Իش��������⣺
(1)TiԪ����Ԫ�����ڱ��е�λ���ǵ�________���ڵ�________�壻���̬ԭ�ӵĵ����Ų�ʽΪ________��
(2)��Ti�Ļ������У����Գ��֣�2����3����4���ֻ��ϼۣ������ԣ�4�۵�Ti��Ϊ�ȶ���ƫ���ᱵ�����ȶ��Ժã���糣���ߣ���С�ͱ�ѹ������Ͳ���������ж���Ӧ�ã�ƫ���ᱵ�����о����Ľṹʾ��ͼ��ͼ��ʾ�����Ļ�ѧʽ��________������Ti4+������λ��Ϊ________��Ba2+������λ��Ϊ________��

(3)�����µ�TiCl4���д̼��Գ�ζ����ɫ��Һ�壬�۵㣭23.2�棬�е�136.2�棬����TiCl4��________���壮
(4)��֪Ti3+���γ���λ��Ϊ6��������ռ乹��Ϊ�������壬����ͼ1��ʾ������ͨ����������ͼ2��ʾ�ķ�������ʾ��ռ乹��(����A��ʾ���壬M��ʾ����ԭ��)����λ������[Co(NH3)4Cl2]�Ŀռ乹��ҲΪ�������ͣ�����________��ͬ���칹�壮

(��)(1)��֪����������ӵĿռ�ṹ����ͼ��ʾ����������ԭ�Ӳ�ȡ________�ӻ���(2)R��1��36��Ԫ����δ�ɶԵ���������ԭ�ӣ�R3+����Һ�д�������ת����ϵ��

R3+![]() R(OH)3
R(OH)3![]() [R(OH)4]��
[R(OH)4]��
�ٻ�̬Rԭ�ӵļ۵����Ų�ʽΪ________��
��[R(OH)4]���д��ڵĻ�ѧ����________��
A�����Ӽ�
B�����Լ�
C���Ǽ��Լ�
D�����
 ��������ϵ�д�
��������ϵ�д� ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
��I������ѡ���⣨6�֣�
��ѧ���ѧ����������ᡢ����������أ�������������ȷ����__________��
A�������Ҵ�����(����������һ�������Ҵ�)���������ܽ��ͻ�����β�����к������ŷ�
B����ҵ����ʯ�����úȼ�պ��γɵ��������������������Ƶ�ʯ��
C��Ϊ����Ч�ķ�չ�����Դ�����õ��ˮ�ķ��������Ʊ�H2
D������ͣ������װ�������ʩ���ɽ�����β����CO��NOx��Ӧ����������
E�����û�ѧ��Ӧԭ������ƺ������µ�ҩ��
��II����14�֣�
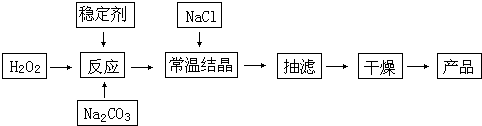
��嫵ĺ�����һ�������Դ���⣬�̲��ŷ��ĵĿ�����DZ���Ļ�ѧ��Դ����ͼ�Ǻ�ˮ�ӹ���ʾ��ͼ��������ͼ�ش����⡣

��1����ˮ��������ͨ�����õ��Ʊ���ˮ�ķ����� ��д�����֣���
��2����ͼ�Ǵ�Ũ����ˮ����ȡ�������ͼ��д����ͼ�Т٢ڵĻ�ѧʽ���� ���� ���������з����Ļ�ѧ��Ӧ����ʽΪ ��

��3���Ʊ�����þ��ͨ��������ڵ�MgC12��������MgO����ԭ���� ��
��4��ʳ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ��ȼҵ����ͨ����ⱥ��ʳ��ˮ���Ʊ�NaOH��H2��C12����ˮ�еõ��Ĵ�������������һЩ���ʣ��������һЩ��ѧ�Լ���ʹ���ʳ��������������ˮ������������ӽ���������ԭ���� �����ʳ��ˮ�����ӽ���Ĥ�����н��У����ӽ���Ĥ�������� ��
��5���ྦྷ����Ҫ����SiHCl3��ԭ�����������丱����SiCl4��ת��ΪSiHCl3��ѭ��ʹ�á�һ�������£���20L�����ܱ������еķ�Ӧ��3SiCl4��g��+2 H2��g��+Si��g�� 4SiHCl3��g������ƽ���H2��SiHCl3���ʵ���Ũ�ȷֱ�Ϊ0.140mol/L��0.020mol/L����H2ȫ����Դ���ȼҵ�������������Ĵ�NaCl������Ϊ kg��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ�꺣��ʡ�����и����߿����в��Եػ�ѧ�Ծ� ���ͣ������
��20�֣�
��I������ѡ������6�֣�
����˵������ȷ���� ��
A��SO2��O3��PO43����C1O4����Ϊ�ȵ�����
B���������ʧȥһ��H+���γ�CH3������̼ԭ�ӵ��ӻ����ͷ����˸ı�
C��Tiԭ�ӵĺ�������Ų�ʽΪ[Ar]3d34s2
D��CS2��H2O��C2H2����ֱ���ͷ���
E��CH4��BCl3��CO2���Ǻ��м��Լ��ķǼ��Է���
��II����14�֣�
������������һ����Ȼ�粻���ڵ��˹��ϳɳ�Ӳ���ϣ�Ӳ�Ƚ����ڽ��ʯ���dz�Ӳ�������������Ҫ�ɾ�֮һ����ش��������⣺
��1���ڵڶ����ڣ�ԭ�ӵĵ�һ������һ����˵������������� ���Ƚ�����ԭ�ӵĵ�һ�����ܣ�Be B��N O���>����<��������ԭ���� ��
��2����ͼΪ����������ľ���������Ļ�ѧʽΪ ���þ�����Bԭ�����Nԭ�ӵ� ��϶�����þ����ı߳�Ϊa cm����ô�þ�����ܶ�Ϊ g/cm3
��ֻҪ���г���ʽ����
[��Դ:ZXXK]
��3������������ľ���ṹ����ʯ�Ľṹ���ƣ������۵�Ƚ��ʯ�ĵͣ��Է�����ԭ��
��
��4�������������������������ڸ��¸�ѹ���Ʊ��������������ֳơ���ʯī�����ṹ������������ʯī���ƣ�������������Nԭ�ӵ��ӻ��������Ϊ �������Ϸ��ķ����л��������������ƽ��ṹʾ��ͼ���á��𡱴���Nԭ�ӣ��á�����Bԭ�ӣ�ÿ��ԭ�Ӳ�����7������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꺣��ʡ������У������ѧ���� ���ͣ������
���л���ѧ������
18-1����6�֣�����Ϊ����ѡ���⣬ȫѡ�Ե�6�֣�©ѡ���������֣���ѡ���ѡΪ0�֣�
���й����л����˵���У���ȷ���ǣ� ��
A�������Ǹ߷��ӻ������ˮ������ܷ���������Ӧ
B������ά��һ�����Ǻϳɸ߷��Ӳ���
C�������µ�������Ʊ���ɫ����������������Cu��OH��2������Ӧ
D����ij����Ļ�������Cl2��Ϲ��գ���������״Һ�����ɣ�˵���������϶����м���
E��  ����������ԭ�Ӳ����ܹ�ƽ��
����������ԭ�Ӳ����ܹ�ƽ��
18-2����14�֣� ��֪��һ��̼ԭ�������������ǻ�ʱ����������ת����

�������ͼ����

��1��A�ĺ˴Ź��������г��� ��壻E�к��еĹ����ŵ������� ��
��2����Ӧ�۵Ļ�ѧ����ʽΪ ��
��3����֪B����Է�������Ϊ162������ȫȼ�յIJ�����CO 2��H2O�����ʵ���֮��Ϊ n��CO2�� �Un ��H2O����2�U1����B�ķ���ʽΪ ��
��4��F�Ǹ߷��ӹ���������е���Ҫԭ�ϡ�F���������ص㣺�����ڷ����廯������ܸ�FeCl3��Һ������ɫ��Ӧ�����ܷ����Ӿ۷�Ӧ���ܱ����ϵ�һ�ȴ���ֻ�����֡�F��һ�������·����Ӿ۷�Ӧ�Ļ�ѧ����ʽΪ ��
��5��������G��F��ͬ���칹�壬���ڷ����廯����ܷ���������Ӧ��G�ж��ֿ��ܵĽṹ����д�����ǵĽṹ��ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꺣��ʡ�����и�����ѧ�ڸ߿����У�һ����ѧ���� ���ͣ������
��20�֣�
��I������ѡ���⣨6�֣�
���ڻ������������������У���ȷ���� ��
A��������ͨ������ԭ����ʯ��ʯ��ʯӢ������ڲ�����¯�з������ӵ�������ѧ�仯
B����ҵ�Ϻϳɰ�û�в��ø����ѹǿ�Ǵ��豸�Ͷ���Ҫ���濼�ǵ�
C�����ȷ�ֻ������������ʱӲ�ȵ�Ӳˮ
D����ҵ�ϲ��õ�������Ȼ�������������
E���øߴ��Ƚ�̿��ʯӢɰ��Ӧ���Ƶô��ȴﵽ9��9�ĵ��������
��II����14�֣�
��ҵ����ȡ����淋�����ͼ���£���ش��������⣺?

��1��1909�껯ѧ�ҹ�����ʵ�����״κϳ��˰���2007�껯ѧ�Ҹ���¡����ض��ڹ����о���֤ʵ�������뵪���ڹ����������ϳɰ��ķ�Ӧ���̣�ʾ�����£�

 �ֱ��ʾN2��H2��NH3��ͼ�ݱ�ʾ���ɵ�NH3�뿪�������棬ͼ�ں�ͼ�۵ĺ���ֱ���
��
��
�ֱ��ʾN2��H2��NH3��ͼ�ݱ�ʾ���ɵ�NH3�뿪�������棬ͼ�ں�ͼ�۵ĺ���ֱ���
��
��
��2���ںϳɰ����豸(�ϳ���)�У������Ƚ�������Ŀ���� ��
�ںϳ��������������ͨ�������Ŀ���� ��
��3����������Ĺ����г������һЩ��������������������ַ���������
��Һ���շ���NO+NO2+2NaOH===2NaNO2+H2O
NH3��ԭ����8NH3+6NO2 7N2+12H2O��NOҲ�����Ƶķ�Ӧ��
7N2+12H2O��NOҲ�����Ƶķ�Ӧ��
�����ɫ��ѧ�ĽǶȷ������ַ��������ӣ�
��4��ij���ʳ���NH3�Ʊ�NH4NO3����֪����NH3��NO�IJ�����96%��NO��HNO3�IJ�����92%������HNO3����ȥ��NH3������ռ�ܺ�NH3����(������������ģ����� %��
��5���������һ�ֳ��õĵ��ʣ��������ʹ�øû���ʱ����Ӧע���������������±���
|
|
ע������ |
���� |
|
�� |
|
|
|
�� |
|
|
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com