
��Ԫ������Ȼ������Ҫ��������������Ҫ�ɷ�ΪAl2O3��������Fe2O3��FeO��SiO2���С���ҵ�����������Ʊ�����ij�ֻ�����Ĺ����������¡�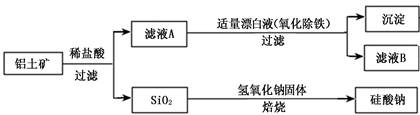
��1������ҺA�м���Ư��Һ��Ŀ��������������������ҺB�����ԡ�
�ٸù������漰ij������ԭ��Ӧ���£�����ɣ�
��Fe2++��ClO��+ =��Fe(OH)3��+��C1��+
�ڼ�����ҺB���Ƿ�����Ԫ�صķ���Ϊ�� ��ע���Լ�������
�۽���ҺB�е���Ԫ���Գ�����ʽ��������ѡ�õ�����Լ�Ϊ ������ţ���
a������������Һ b��������Һ c����ˮ d��������̼
������ҺB�Ʊ��Ȼ��������漰�IJ���Ϊ���ߵμ�Ũ���������Ũ������ȴ�ᾧ�� ����������ƣ���ϴ�ӡ�
��2����֪H2O2��һ�����ᣬ��ǿ������Һ����Ҫ��HO2����ʽ���ڡ�Ŀǰ�о��Ƚ����ŵ�Al��H2O2ȼ�ϵ�أ���ԭ������ͼ��ʾ������ܷ�Ӧ���£�2Al+3HO2��=2AlO2��+OH��+H2O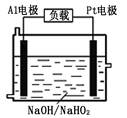
��������ӦʽΪ ��
��Al�缫�ױ�NaOH��Һ��ʴ�����Ǹõ��Ŀǰδ���ƹ�ʹ�õ�
ԭ��֮һ���缫����ʴ�����ӷ���ʽΪ ��
��3���ֲĶ������Է�ֹ�ֲĸ�ʴ���ڶ��������е��Һ���������Σ��ɷ�NaCl��KCl�����ʱ��Ԫ�غ���Ԫ����Ҫ��AlCl4����ʽ���ڣ����������Ȼ�����Һ��ԭ���� ��
��4��SiO2��NaOH�����Ʊ������ƣ��ɲ��õ�װ��Ϊ ������ţ���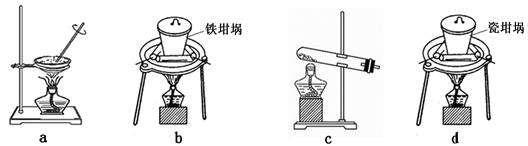
(14��)��1����2Fe2++ClO��+5H2O��2Fe(OH)3��+C1��+4H+��2�֣�
��ȡ������ҺB������KSCN��Һ��������죬�ټ�����ˮ����Ȼ����죬˵����ҺB�в�����Ԫ�ء�
�����������𰸾��÷֣���2�֣� �� c��1�֣� �ܹ��ˣ�1�֣�
��2����HO2��+2e��+H2O��3OH����2�֣� ��2Al+2OH��+2H2O��2AlO2��+3H2����2�֣�
��3���Ȼ�����Һ�У�H+�õ�������ǿ��Al3+����Ƴ�����������������2�֣� ��4��b��2�֣�
���������������1���ٸ��ݷ���ʽ��֪����Ԫ�صĻ��ϼ۴ӣ�2�����ߵ���3�ۣ�ʧȥ1�����ӡ���Ԫ�صĻ��ϼ۴ӣ�1�ۣ����͵���1�ۣ��õ�2�����ӣ�����ݵ��ӵĵ�ʧ�غ��֪����ԭ���������������ʵ���֮����2:1������ԭ���غ������غ��֪����Ӧǰ��ˮ�μӣ���Ӧ�������������ɣ������ƽ��ķ���ʽ��2Fe2++ClO��+5H2O��2Fe(OH)3��+C1��+4H+��
�������������ܺ�KSCN��Һ������ɫ��Ӧ�����Լ��������ӡ����������Ӽ��黹ԭ�ԣ��ܱ���ˮ�������������ӣ��ݴ˿��Լ�����ҺB�Ƿ�����Ԫ�ء���ȡ������ҺB������KSCN��Һ��������죬�ټ�����ˮ����Ȼ����죬˵����ҺB�в�����Ԫ�ء�
����ҺB�е���Ԫ����Al3������ʽ���ڣ�Ҫ�Գ�����ʽ��������Ҫ�����Һ������������������������������Բ���ѡ��ǿ����ѡ�����ˮ����ѡc��
����ȴ�ᾧ���о�����������˷���������ʵ�����Ӧ���ǹ��ˡ�
��2����ԭ����и�����ʧȥ���ӣ������õ����ӡ�����ݷ���ʽ2Al+3HO2��=2AlO2��+OH��+H2O��֪����������ʧȥ���ӣ�������HO2���õ����ӣ����������ĵ缫��Ӧʽ��HO2��+2e��+H2O��3OH����
�ڽ������ܱ�����������Һ�ܽ⣬����ƫ�����ƺ���������Ӧ�����ӷ���ʽ��2Al+2OH��+2H2O��2AlO2��+3H2����
��3�����ǻ��õĽ����������˳�����λ�����ǰ�棬���Ȼ�����Һ�У�H+�õ�������ǿ��Al3+����Ƴ�����������������Al3+���ŵ磬�ò�����������
��4��a���������������ڶ���������������ƣ�����ȷ��b�����������������ڶ���������������ƣ���ȷ��c���Թܣ��������ڶ���������������ƣ�����ȷ��d�Ǵ��������������к��ж��������ܺ��������Ʒ�Ӧ�����������������ƣ�����ȷ����ѡb��
���㣺����������ԭ��Ӧ����ƽ����Ԫ�صļ��顢�����������Ʊ�������ʵ��������缫��Ӧʽ����д��������ѡ���


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
һѧϰС��������ͼ��ʾװ�ã���ij������Fe�ķ�ͭм����ͭ�����IJⶨ����̽���������Ʊ�����ͭ��Һ��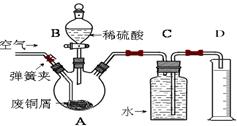
��1����A�м���10g��ͭм��Ʒ���رյ��ɼУ���B������Aע������ϡ�����رա��ٴ�ʱװ��C�в����������� ��
�ڷ�Ӧ���е�A�в��ٲ�������ʱ����C���ռ���448mL���ѻ���ɱ�״�������壬��÷�ͭм��ͭ�������ٷֺ���Ϊ ��
��2��������ʹCu��ȫ�ܽ�IJ����ǣ�
�� ���C��Dװ�ú��ɼ�
��____________________________________________________________________
��3��ΪʹA�й���ӿ��ܽ����ʣ����·������ú������� ��
a.��Aװ�ü��ȣ�b.��A�ڼ�������Fe2O3��c.��A�ڼ�������CuO��d.�������ͨ������e.��A�ڼ�������FeSO4��f.��A�ڼ�������H2O
��4����A����Һ�����ձ��ڣ�����Cu2(OH)2CO3���������pH=4ʱ����Һ����Ԫ�ر���ȫ���������˺ú��ɫ����������ͭ��Һ��
�ٴ˹��̵����ӷ�Ӧ����ʽ�� ��
�ڼ�����Һ����Ԫ���Ƿ���ȫ������õķ�����___________
a.ȡ�����Թܡ��μ�KSCN��Һ
b.ȡ�����Թܡ��μ�����KMnO4��Һ
c.ȡ�����Թܡ�ֽ�ϲ������ ��KSCN��Һ��
��5����ͬѧ��Ϊ�����Բ��ò������巨�����ͭм��ͭ�������ٷֺ��������������������дΪ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�Ȼ�ͭ��һ�ֹ㷺�����������ϡ�ľ�ķ������ȵĻ�����Ʒ��ij�о�С���ô�ͭ��������Fe�������������Ʊ��Ȼ�ͭ���壨CuCl2��2H2O����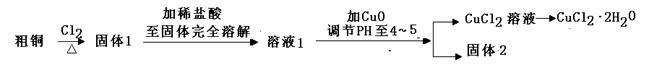
��1��ʵ���Ҳ�������ͼ��ʾ��װ�ã��ɽ���ͭ��Cl2��Ӧת��Ϊ����1�����������ͼг�װ������ȥ����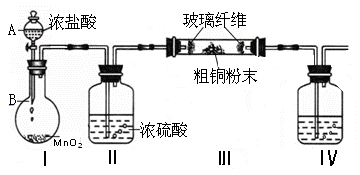
�� ����A�������� ��
�� װ��B�з�����Ӧ�����ӷ���ʽ�� ��
�����Ũ�����˳�����£����ܵ�ԭ����
��װ�â���ʢװ���Լ��� ���������� ��
��2����CuCl2��Һת��ΪCuCl2��2H2O�IJ��������У�������Һ��ɫ����ɫ��Ϊ����ɫ��С��ͬѧ��̽����ԭ��
��֪�����Ȼ�ͭ��Һ��������ת����ϵ��
Cu(H2O)42+(aq) +4Cl��(aq) CuCl42- (aq) +4H2O(l)
CuCl42- (aq) +4H2O(l)
��ɫ ��ɫ
��������Ӧ�Ļ�ѧƽ�ⳣ������ʽ��K= ��
������ʹ��Һ�ɻ�ɫ�����ɫ����д�����ֿɲ��õķ���
a. b. ��
��3����CuCl2��Һ�õ�CuCl2��2H2O�Ĺ�����Ҫ���������Ŀ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ʯ�ǹ�ҵ��������Ҫԭ��֮һ������Ҫ�ɷ�Ϊ����������������в�����Ԫ�غ���Ԫ�أ������ʲ���H2SO4��Ӧ����ij�о���ѧϰС���ij����ʯ������������Ļ�ѧʽ����̽����
������ʯ�к������IJⶨ��
�� ����ͼ��װ���������װ�õ������ԣ�
�� ��5.0g����ʯ����Ӳ�ʲ������У�װ��B��C�е�ҩƷ��Ϊ��ʯ��(���ͼʾ���г�����ʡ��)
�� ����˵����ܿڴ����ϵػ���ͨ��H2����Cװ�ó��ڴ�H2�鴿��ȼA���ƾ���
�� ��ַ�Ӧ�����ƾ��ƣ��ٳ���ͨ����������ȫ��ȴ��
��1��װ��C������Ϊ �������� ��������������
��2����ķ�Ӧ��װ��B����1.35g��������ʯ�����İٷֺ���Ϊ�������� ��
������ʯ�к������IJⶨ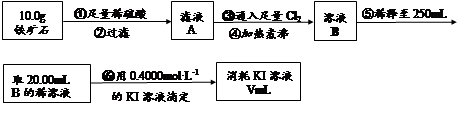
��3����������������������������������������������������������� ��
��4����������õ��IJ����������ձ�������������ͷ�ιܡ������� ���� ��
��5�������йز���IJ�����˵����ȷ���������������������� �������� ��
a����Ϊ��ˮΪ��ɫ�����Եζ������в����ָʾ��
b���ζ������п����õ�����Һ��Ϊָʾ��
c���ζ���������ˮϴ�Ӻ����ֱ��װҺ
d����ƿ����Ҫ�ô���Һ��ϴ
e���ζ������У��۾�ע�ӵζ�����Һ��仯
f���ζ�������30s����Һ���ָ�ԭ������ɫ�ٶ���
��6�����ζ�����������0.4000mol��L?1KI��Һ25.00ml��������ʯ�����İٷֺ���Ϊ�� ����
��7���ɢ���������������ʯ������������Ļ�ѧʽΪ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijʵ��С���CO2ͨ�뱥��Na2CO3��Һ��ȡNaHCO3��װ����ͼ��ʾ���������Ѽ��飬���ּг�װ���ԣ���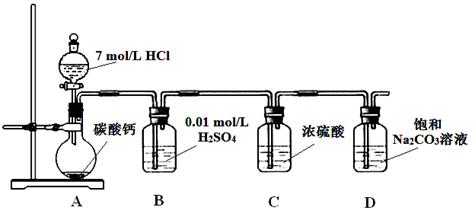
��1��D�в���NaHCO3�Ļ�ѧ����ʽ�� ��
��2�����ϻ�ѧƽ���ƶ�ԭ������B����Һ������ ��
��3����D���д�����ɫ��������ʱ��ֹͣʵ�飬��������ˡ�ϴ�ӡ����ﱸ�á�Ϊȷ������ijɷ֣�ʵ��С����Ʒ������£���ȡһ�������Ĺ��壬���1000 mL��Һ��Ϊ��Һ��������屸�ã���
�� ����1��ȡ��Һ������Ca(OH)2��Һ��ϣ����ְ�ɫ������
ʵ��С������������ԭ�����з�������Ϊ�÷����������������� ��
�� ����2��ȡ��Һ��BaCl2��Һ��ϣ����ְ�ɫ�����������������
ʵ��С����Ϊ�����д���NaHCO3�������ӷ���ʽ�� ��
��С����Ϊ����ȷ���Ƿ����Na2CO3������Ϊ�ý����Ƿ������_____��
�� ����3��ʵ��С���мס���ͬѧ����NaHCO3�IJ��ȶ��Խ�������ʵ�飺
��ͬѧ��ȡ��Һ400 mL����pH�Ʋ���ҺpH����ˮԡ����������200 mL���������IJ����� �����������ɫ�����д���NaHCO3��Ϊ��һ��֤����ɫ�����Ƿ�Ϊ������NaHCO3����ϼ�ͬѧʵ�飬��Ӧ�����ʵ���� ��
��ͬѧ�����������ⶨ�˹�����������¶ȱ仯�����ߣ�����ͼ��ʾ��
a. ����A������õ��Ľ����� ��
b. ����B�����꣬�������������n(NaHCO3:n(Na2CO3))= ��
ͨ������ʵ�飬��С����Ϊ��������Na2CO3��Һ��ͨ�����CO2�Ʊ�NaHCO3��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
SiO2��SO2��CO2����������������ǵĻ�ѧ���ʾ���һ���������ԣ�Mg��Na�Ļ�ѧ����Ҳ����һ�������ԡ�ij��ȤС������ͼ��ʾװ�ý���Mg��SO2��Ӧ��ʵ�顣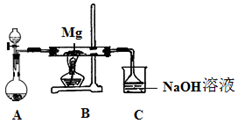
��1��ѡ����ȡSO2�ĺ����Լ� �����ţ���
��ŨHCl ��ŨH2SO4 ��Na2SO3���� ��CaSO3����
��2������װ�û����Ż����Ż��ķ����� ��װ��C��NaOH��Һ�������� ��
��3����ͬѧ�Ʋ�Mg��SO2�ķ�Ӧ��CO2���ƣ���÷�Ӧ����ʽΪ
����ͬѧ���Ʋ��ǣ�2Mg+3SO2 2MgSO3+S����ͬѧ���Ʋ��ǣ�3Mg+SO2
2MgSO3+S����ͬѧ���Ʋ��ǣ�3Mg+SO2 2MgO+MgS��Ҫ��֤�ס��ҡ�����λͬѧ���Ʋ��Ƿ���ȷ����ͬѧ������ʵ��̽����
2MgO+MgS��Ҫ��֤�ס��ҡ�����λͬѧ���Ʋ��Ƿ���ȷ����ͬѧ������ʵ��̽����
��֪��MgSO3��MgS��������ˮ���������ᷢ�����ֽⷴӦ�ų����壻H2S����ͨ��CuSO4��Һ�г��ֺ�ɫ������
��ѡ�Լ���2mol/L HCl��2mol/L HNO3������ˮ��2mol/LNaOH��Ʒ����Һ������ʯ��ˮ��2mol/LCuSO4����������Ʒ��ѡ��
| ��� | ʵ�鲽�� | Ԥ������ͽ��� |
| �� | ȡ������Ӧ�����ù������Թ��� | |
| �� | ���Թ��еĹ��������μ� �� �Թܿ����ϴ����ܵĵ���������������ͨ��ʢ�� ���Թ��С� | ���Թ��е� �� ���ͬѧ�Ʋ���ȷ�� ���Թ��еĹ���δ��ȫ�ܽ⣬�� ������ͬѧ�Ʋ���ȷ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���ֳ�������X��Y��Z��E��F������ͬһԪ��M����֪Y�ǵ��ʣ�Z��NO(һ������)����һ������������������ת����ϵ�������ƶϲ��������� �� ��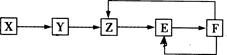
| A��MԪ�ؿ����Ƿǽ���Ԫ�أ�Ҳ�����ǽ���Ԫ�� |
| B��X������һ���⻯�� |
| C��E������һ����ɫ���� |
| D��X��Y�����Ƿ�������ԭ��Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��SO2����ͨ��BaCl2��Һ�����ͣ�δ���г������ɣ�����ͨ����һ��������������������ͨ�����������ǣ� ��
| A��NH3 | B��Cl2 | C��CO2 | D��NO2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�����й����ʵ����ʺ����ʵ�Ӧ�þ���ȷ����
| A��ͭ�Ľ��������Ա����IJ���ں��������װ����ͭ���Լ����丯ʴ |
| B���������費���κ��ᷴӦ������ʯӢ������������ |
| C��������Ũ���������������ۻ������ڳ�������������������Ũ���� |
| D���������Ⱦ��л�ԭ�ԣ�����������ˮ��ɱ������ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com