
��12�֣���ʽ̼��þ[һ����xMgCO3?yMg(OH)2?zH2O��ʾ]�ܶ�С������������ˮ������ˮ���������ļ��Է�Ӧ���������Ҵ��������²���ֽ⣬������Ʒ���������ϡ�������������ͼ��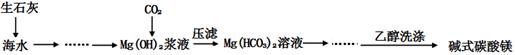
��1���ش��������⣺
����ʯ������ʯ��ʯ�뽹̿����������ɣ���̿�������ǣ� ��
��������þ��Һ��CO2��Ӧ�Ļ�ѧ����ʽΪ��
��
�ۼ�ʽ̼��þ���Ҵ�ϴ�ӵ�Ŀ���ǣ� ��
��2����֪����ʽ̼��þ�ֽⷴӦ����ʽΪ��
xMgCO3?yMg(OH)2?zH2O 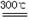 ��x+y��MgO+xCO2��+��y+z��H2O
��x+y��MgO+xCO2��+��y+z��H2O
ijͬѧ��46.6g��ʽ̼��þ��Ʒ����Ӳ�ʲ������г�����������أ�������������Ϊ20.0g����������������ͨ��������Ũ���ᣬ��ͨ�������ļ�ʯ�ң���ʯ����������17.6g������ʵ��ⶨ�����ݣ�ȷ����ʽ̼��þ[xMgCO3��yMg(OH)2?zH2O]�Ļ�ѧʽ��Ҫ��д��������̣���
��1������ȼ�ϡ��ṩ���� ��2�֣�
�� Mg(OH)2+2CO2 = Mg(HCO3)2 ��2�֣�
�۳�ȥ�������ʡ����ٹ������� ��2�֣�
��2��������ã�
n(Mg2+)=20.0g/40g��mol-1=0.5mol ��1�֣�
n(CO2)=17.6g/44g��mol-1= 0.4mol ��1�֣�
��Ԫ���غ�ã�
m(MgCO3)=0.4mol��84g��mol-1=33.6g��1�֣�
m��Mg(OH)2��=(0.5mol-0.4mol)=5.8g��1�֣�
n(H2O)=44.6g-33.6g-5.8g/18g��mol-1=0.4mol��1�֣�
x:y:z=4:1:4
���ʽ̼��þ�Ļ�ѧʽΪ��4MgCO3��Mg(OH)2��4H2O��1�֣����������̺���Ҳ���֣�
���������������1����CaCO3���·ֽ�����CaO��CO2�����Խ�̿�������ǣ���ȼ�ϡ��ṩ������
�ڸ�������ͼ��CO2��Mg(OH)2��Ӧ��IJ��ᆳѹ�˵õ�Mg(HCO3)2������CO2��Mg(OH)2��Ӧ�Ļ�ѧ����ʽΪ��Mg(OH)2+2CO2 = Mg(HCO3)2
����Ϊ��ʽ̼��þ��ˮ���������ļ��Է�Ӧ���������Ҵ����������Ҵ�ϴ�ӿ��Գ�ȥ�������ʡ����ٹ������ġ�
��2����������ΪCaO����ʯ������CO2������������ΪCO2���������ɵã�
n(Mg2+)=20.0g/40g��mol-1=0.5mol ��1�֣�
n(CO2)=17.6g/44g��mol-1= 0.4mol ��1�֣�
��Ԫ���غ�ã�
m(MgCO3)=0.4mol��84g��mol-1=33.6g��1�֣�
m��Mg(OH)2��=(0.5mol-0.4mol)=5.8g��1�֣�
n(H2O)=44.6g-33.6g-5.8g/18g��mol-1=0.4mol��1�֣�
x:y:z=4:1:4
���ʽ̼��þ�Ļ�ѧʽΪ��4MgCO3��Mg(OH)2��4H2O��1�֣�
���㣺���⿼�黯ѧ���̵ķ�������ѧ����ʽ����д��ϴ�ӷ�������ѧ���㡣


 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ijѧ��������Ϊa g���߳�Ϊb cm�������ζ�п��Ƭ��������ᷴӦ�ⶨ�Ʋ��ȣ�����ʵ������ͼʾ�⣬��Ӧ��Ϻ���Ƭ����Ϊc g(��֪п���ܶ�Ϊ�� g/cm3)�����������������
| A��t1ʱ��п�Ʋ�����ȫ��Ӧ |
B��п�Ʋ�ĺ��Ϊ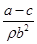 |
| C����Ӧ����Ҳ��Ӱ��OA�εķ�Ӧ���ʵ�����֮һ |
| D����ԭ��ط�Ӧ�����������ĵ缫��ӦʽΪZn - 2e �� Zn2+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ�鲻�ܴﵽĿ�ĵ���
| A����AlCl3��Һ������ˮ�Ʊ�Al(OH)3 |
| B����NH4Cl��Ca(OH) 2�����ϼ����Ʊ�NH3 |
| C����NaOH��Һ��ȥ���е��� |
| D��������ͭ�۳�ȥFeCl2��Һ�е�FeCl3���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ƽ������[KNH4Fex(SO4)y(OH)z]������ˮ��ϡ���ᣬ��ȡ�Ƽ��������ʾ��ͼ���£�
��1����ҺX��________��
��2��������Һ���Ƿ����K���IJ�����________________________��
��3���Ƽ�������Ļ�ѧʽ��ͨ������ʵ��ⶨ��
�ٳ�ȡһ����������Ʒ����ϡ�������ܽ⣬��������Һת��������ƿ�����Ƴ�100.00 mL��ҺA��
����ȡ25.00 mL��ҺA�����������ữ��BaCl2��Һ��������ȫ�����ˡ�ϴ�ӡ����������أ��õ���ɫ����9.32 g��
����ȡ25.00 mL��ҺA����������NaOH��Һ�����ȣ��ռ�����״��������224 mL��ͬʱ�к��ɫ�������ɡ�
�ܽ���������ó������ˡ�ϴ�ӡ����գ����յù���4.80 g��
ͨ������ȷ���Ƽ�������Ļ�ѧʽ(д���������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
ijС�������ͼװ�ã����ּг�װ������ȥ������̽����ʪ��Cl2��Na2CO3��Ӧ�õ��������ʵijɷ֡�
��1��A����ȡCl2�����ӷ���ʽ .
��2��ͨ��һ������ʪ��Cl2��Ӧ����⣬D��ֻ��Cl2Oһ�����壬C��ֻ��һ�������⣬ͬʱ����NaHCO3�ȣ�ijͬѧ��C�����ù�������ijɷֽ���̽����
������������衣
����1���������ֳɷ֣�NaHCO3�� ��
����2���������ֳɷ֣�NaHCO3�� ��
����Ʒ���������ʵ�顣д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ�
��ѡʵ���Լ�������������ˮ��ϡHNO3��BaCl2��Һ������ʯ��ˮ��AgNO3��Һ���Թܡ�С�ձ��� ��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ��
��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��12�֣�ijͬѧ��ͬ����Ԫ�����ʵݱ����ʵ��ʱ���Լ������һ��ʵ�鷽��������¼���й�ʵ���������±������������ͬѧ���������ʵ�鱨��:
| ʵ�鷽�� | ʵ������ |
| ����ɰֽ�����þ�����ˮ��Ӧ������Ӧ����Һ�еμӷ�̪ | ��A������ˮ�棬�۳�С����ˮ�����ζ�����֮��ʧ����Һ��� |
| �������Ƶ�H2S������Һ�еμ����Ƶ���ˮ | ��B���������壬���ڿ�����ȼ�գ���Һ��Ϊdz��ɫ |
| ��������з�̪��Һ����ˮ��Ӧ | ��C����Ӧ��ʮ��ǿ�ң�������������ڿ�����ȼ�� |
| ��þ����2mol/L���ᷴӦ | ��D�����ҷ�Ӧ��������ȼ���� |
| ��������2mol/L���ᷴӦ | ��E�����ɰ�ɫ��״����������������ʧ |
| ����AlCl3��Һ�еμ�NaOH��Һ������ | ��F�����ɵ���ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣��л��������ұ���GB2760��2011���涨���Ѿ���SO2���ʹ����Ϊ0.25g/L��ij��ȤС������9ͼ1װ�ã��г�װ���ԣ��ռ�ij���Ѿ���SO2�����Ժ������Բⶨ��
��1������A��������_______��ˮͨ��A�Ľ���Ϊ_________��
��2��B�м���300.00ml���Ѿƺ��������ᣬ����ʹSO2ȫ���ݳ�����C��H2O2��ȫ��Ӧ���仯ѧ����ʽΪ______________��
��3����ȥC�й�����H2O2��Ȼ����0.0900mol/LNaOH����Һ���еζ����ζ�ǰ������ʱ��Ӧѡ����9��ͼ2�е�_____�����ζ��յ�ʱ��Һ��pH��8.8����ѡ���ָʾ��Ϊ__________������50ml�ζ��ܽ���ʵ�飬���ζ����е�Һ���ڡ�10�����������Һ������������ţ�____���٣�10ml���ڣ�40ml���ۣ�10ml���ܣ�40ml��
��4���ζ����յ�ʱ������NaOH��Һ25.00ml�������Ѿ���SO2����Ϊ��__g/L��
��5���òⶨ�����ʵ��ֵƫ�ߣ�����ԭ����������װ������Ľ���ʩ_______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУѧ����ѧʵ��С�飬Ϊ��֤�ǽ���Ԫ�ط���������ǿ����͵��������һ��ʵ��װ�ã������ּӳ�װ������ȥ��
��1��д��A�з�Ӧ�����ӷ���ʽ ��
��2��B�г��ֻ�ɫ��������������������ӷ���ʽ ��
��3���Դ�ԭ�ӽṹ�ǶȽ����ȵ������Դ������ԭ�� ��
��4��D�и�����г��ֵ�����ѧ����ʽ ��
��5����ͬѧ��ΪD�е�������˵���ȵ������Դ��ڵ�����Ҫ��C֮ǰ��װϴ��װ�ã��뻭����װ��ͼ ����ע��ʢװ�Լ�����
��6������ʲô������֤��������Cl2��S����һ�������ʵ˵�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������Ũ�������ܷ����ۻ�����ij��ȤС���ͬѧ���ֽ�һ����������Ũ�������ʱ���۲쵽����ȫ�ܽ⣬�������������塣Ϊ�ˣ��������������װ����֤�����������塣
��1��֤����Ӧ����������������SO2���ɵ������� ��
��2��֤�������к���������ʵ�������� ��
��3��Ϊ�˽�һ��̽����Ӧ��A��Һ����Ԫ�صļ�̬�����ǽ��������µļ��裺
����1����Һ����Ԫ�ؼ���Fe3+Ҳ��Fe2+
����2����Һ����Ԫ��ֻ��Fe3+
����3����Һ����Ԫ��ֻ��________________
���ڼ���1�������Լ���0.01 mol/L����KMnO4��Һ��ϡ��ˮ��Һ��0.1 mal/L KI��Һ��
������Һ��KSCN��Һ������ˮ����̽����������Һ������ɱ������ݡ�
��ʵ��̽����
| ʵ����� | Ԥ������ | ���� |
| ȡ��Ӧ���A��Һ��װ��a��b���Թܣ�����٣���a�Թ��е��� �� | | |
| ����ڣ���b�Թ��е��� �� | | ��Һ����Fe3+ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com