
 H= ?241��8kJ/mol
H= ?241��8kJ/mol  H= ?110��5kJ/mol
H= ?110��5kJ/mol 2SO3(g)��
2SO3(g)�� 2NO(g)
2NO(g)  H��0
H��0 H��0��
H��0�� CO2(g)��H2(g)����420��ʱ��ƽ�ⳣ��K=9������Ӧ��ʼʱ��CO��H2O��Ũ�Ⱦ�Ϊ0��1mol/L����CO�ڴ˷�Ӧ�����µ�ת����Ϊ ��
CO2(g)��H2(g)����420��ʱ��ƽ�ⳣ��K=9������Ӧ��ʼʱ��CO��H2O��Ũ�Ⱦ�Ϊ0��1mol/L����CO�ڴ˷�Ӧ�����µ�ת����Ϊ �� H=+131��3kJ/mol (3��)(����ʽ���±���ȷ�ɸ�1��)
H=+131��3kJ/mol (3��)(����ʽ���±���ȷ�ɸ�1��)  G=
G= H��T
H��T S,
S,  G��0,����ʵ�֡� (2��)
G��0,����ʵ�֡� (2��) H1= ?241��8kJ/mol ��C(s)+1/2O2(g)="CO(g)"
H1= ?241��8kJ/mol ��C(s)+1/2O2(g)="CO(g)"  H2= ?110��5kJ/mol���ݸ�˹���ɣ��÷���ʽ2��ȥ����ʽ1���ɵã�д����̿��ˮ������Ӧ��H2��CO���Ȼ�ѧ����ʽ��1��C(s)+H2O(g)=CO(g)+H2(g)
H2= ?110��5kJ/mol���ݸ�˹���ɣ��÷���ʽ2��ȥ����ʽ1���ɵã�д����̿��ˮ������Ӧ��H2��CO���Ȼ�ѧ����ʽ��1��C(s)+H2O(g)=CO(g)+H2(g)  H=+131��3kJ/mol ����2���ɷ�Ӧ����ʽ��֪����2mol��SO3ʱ����a kJ����õ�2a kJ��������������4molSO3�����ڷ�Ӧ�ǿ��淴Ӧ����������ȫ���е��ף�����Ҫ�õ�4molSO3��SO2��O2���ʵ�������Ҫ����4mol�� 2mol����D���ϣ�Bѡ���м�������Ӧ�����淴Ӧ�����շų�������С��2akJ����ѡD����3����A�����ڷ�Ӧǰ��������䣬��ѹǿʼ�ղ��䲻����Ϊƽ���ж����ݣ����� B�����ڷ�Ӧǰ���������������ʵ��������ı䣬�ʻ������ƽ����Է����������䣬�ʲ�����Ϊƽ���ж����ݣ�����ѡCD�����ɷ�Ӧ2CO(g)=2C(s)+O2(g)
H=+131��3kJ/mol ����2���ɷ�Ӧ����ʽ��֪����2mol��SO3ʱ����a kJ����õ�2a kJ��������������4molSO3�����ڷ�Ӧ�ǿ��淴Ӧ����������ȫ���е��ף�����Ҫ�õ�4molSO3��SO2��O2���ʵ�������Ҫ����4mol�� 2mol����D���ϣ�Bѡ���м�������Ӧ�����淴Ӧ�����շų�������С��2akJ����ѡD����3����A�����ڷ�Ӧǰ��������䣬��ѹǿʼ�ղ��䲻����Ϊƽ���ж����ݣ����� B�����ڷ�Ӧǰ���������������ʵ��������ı䣬�ʻ������ƽ����Է����������䣬�ʲ�����Ϊƽ���ж����ݣ�����ѡCD�����ɷ�Ӧ2CO(g)=2C(s)+O2(g)  H��0����֪�÷�Ӧ���������ؼ��ķ�Ӧ������
H��0����֪�÷�Ӧ���������ؼ��ķ�Ӧ������ G=
G= H��T
H��T S,
S,  G��0,����ʵ�֡�
G��0,����ʵ�֡� 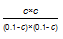 =9 ���c=0��075����һ����̼�ڴ������µ�ƽ��ת����=75%���ʴ�Ϊ��75%��
=9 ���c=0��075����һ����̼�ڴ������µ�ƽ��ת����=75%���ʴ�Ϊ��75%��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������



| ʵ���� | T(0C) | NO��ʼŨ�� ��mol/L�� | CO��ʼŨ�� ��mol/L�� | �����ı� �����(m2/g) |
| �� | 280 | 1��20��10-3 | 5��80��10-3 | 82 |
| �� | a | b | c | 124 |
| �� | 350 | d | e | 124 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ��ѧ�� | H��H | N��H | N��N |
| ����kJ��mol��1 | 436 | 391 | 945 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 C(s)=
C(s)= CO2(g)��2Fe(s) ��H����234��1 kJ/mol
CO2(g)��2Fe(s) ��H����234��1 kJ/mol O2(g)=Fe2O3(s) �Ħ�H�ǣ� ��
O2(g)=Fe2O3(s) �Ħ�H�ǣ� ��| A����824��4 kJ/mol | B����627��6 kJ/mol |
| C����744��7 kJ/mol | D����169��4 kJ/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CH3OH(g)��H2O(g) ��H����49.0kJ/mol
CH3OH(g)��H2O(g) ��H����49.0kJ/mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


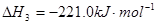
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��2 molH2(g)��1molO2(g)����������2mol H2O (l)������ |
| B��1 mol H2O (g)�ֽ��H2(g)��O2 (g)������241.8kJ���� |
| C��1mol H2O (l)���1mo1 H2O (g)������88 kJ���� |
| D����������H2O (g)��H2O(l������������ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com