
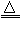
 2MgO+Si��ͬʱ�ᷢ������Ӧ��2Mg+ Si
2MgO+Si��ͬʱ�ᷢ������Ӧ��2Mg+ Si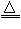 Mg2Si��
Mg2Si��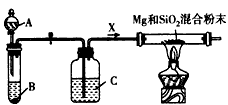


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

ͼ2-10
�Իش��������⣺
(1)�����к���Ca2+��Mg2+��![]() �����ʣ�����ʱ�����Լ�Ϊ��A.���B.BaCl2��Һ��C.NaOH��Һ��D.Na2CO3��Һ�������Լ���˳���ǣ�_______________________________________��
�����ʣ�����ʱ�����Լ�Ϊ��A.���B.BaCl2��Һ��C.NaOH��Һ��D.Na2CO3��Һ�������Լ���˳���ǣ�_______________________________________��
(2)��ⱥ��ʳ��ˮʱ�����Դ���������ĵ缫�Ϸ����ķ�ӦΪ_____________________�����Դ���������ĵ缫������ҺpH__________�������䡱��С��������1 mol���ӵĵ���Ϊ96
(3)��MgCl2��6H2O������ˮ����ˮMgCl2ʱ��MgCl2��6H2O������_________�����м�����ˮ���������������_____________________________��
(4)�����ˮMgCl2���õ�þ��������������____________��������ȴ��
A.H2 B.N
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(14��)������������һ����Ҫ�ĵ���Ԫ�����ϡ����ز��ϡ���ҵ�����ú������ϣ�������þ�Ͻ�Ϊ������ȡ���������ٸ������ղ�������ȡ��������������֪����ĸơ�þ������������ˮ���������й�������ʾ��ͼ�ش����⡣
(1)���������ж�ν��й��ˡ�ʵ���ҽ��й��˲������õ����������������������� ��
(2)������������������̼������Һ��pH��4.0~4.5���ң�����̼������Һ�������� ���ټ���NH4F��Ŀ���� ��
(3)��������NiC2O4��2H2O�����ȿ����и�����ˮ���ڸ�����������Сʱ���Ƶ�������������ͬʱ���һ�ֻ�����塣д�����������ȷֽ�Ļ�ѧ����ʽ ��
(4)��ҵ�ϻ����õ�ⷨ��ȡ��������������NaOH��Һ����NiCl2��ҺpH��7.5���������������ƺ���ö��Ե缫���е�⡣�������в�����Cl2��80%������������������ClO�����ٰѶ���������Ϊ��������д��ClO������Ni(OH)2�������������������ӷ���ʽ ��a mol������ȫ��ת��Ϊ������ʱ�����·��ͨ�����ӵ����ʵ����� ��
(5)��ⷨ��ȡ����������ʵ�ʹ����У���ʱ���һ�ֽᾧˮ�����֪1mol�������к���0.5 mol�ᾧˮ��ȡ�û�����20.2 g���г�ּ��ȣ���������������������0.2 molˮ����ýᾧˮ����Ļ�ѧʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ��ͨ��ͨ��������4�²�©��ȱר���⻯ѧ�Ծ����������� ���ͣ������
(14��)������������һ����Ҫ�ĵ���Ԫ�����ϡ����ز��ϡ���ҵ�����ú������ϣ�������þ�Ͻ�Ϊ������ȡ���������ٸ������ղ�������ȡ��������������֪����ĸơ�þ������������ˮ���������й�������ʾ��ͼ�ش����⡣
(1)���������ж�ν��й��ˡ�ʵ���ҽ��й��˲������õ����������������������� ��
(2)������������������̼������Һ��pH��4.0~4.5���ң�����̼������Һ�������� ���ټ���NH4F��Ŀ���� ��
(3)��������NiC2O4��2H2O�����ȿ����и�����ˮ���ڸ�����������Сʱ���Ƶ�������������ͬʱ���һ�ֻ�����塣д�����������ȷֽ�Ļ�ѧ����ʽ ��
(4)��ҵ�ϻ����õ�ⷨ��ȡ��������������NaOH��Һ����NiCl2��ҺpH��7.5���������������ƺ���ö��Ե缫���е�⡣�������в�����Cl2��80%������������������ClO�����ٰѶ���������Ϊ��������д��ClO������Ni(OH)2�������������������ӷ���ʽ ��a mol������ȫ��ת��Ϊ������ʱ�����·��ͨ�����ӵ����ʵ����� ��
(5)��ⷨ��ȡ����������ʵ�ʹ����У���ʱ���һ�ֽᾧˮ�����֪1mol�������к���0.5 mol�ᾧˮ��ȡ�û�����20.2 g���г�ּ��ȣ���������������������0.2 molˮ����ýᾧˮ����Ļ�ѧʽΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com