
������ʵĵ���ƽ�⡢�����ˮ��ƽ�����������ܽ�ƽ������ڻ�ѧƽ�⡣
��.��֪H2A��ˮ�д�������ƽ�⣺H2A=H����HA����HA��??H����A2����
��1��������NaHA��Һ��pH________(�����)��ԭ����_________________��
A������7 B����7
C������7 D����ȷ��
��2��ij�¶��£�����0.1 mol��L��1��NaHA��Һ����εμ�0.1 mol��L��1KOH��Һ����Һ������(���Ի�Ϻ���Һ������仯)����ʱ�û����Һ�е����й�ϵһ����ȷ����________��
A��c(H��)��c(OH��)��1.0��10��14
B��c(Na��)��c(K��)��c(HA��)��2c(A2��)
C��c(Na��)��c(K��)
D��c(Na��)��c(K��)��0.05 mol��L��1
��3����֪������H2A�ĸ���(CaA)�ı�����Һ�д�������ƽ�⣺CaA(s)??Ca2��(aq)��A2��(aq)����H��0����Ҫʹ����Һ��Ca2��Ũ�ȱ�С���ɲ�ȡ�Ĵ�ʩ��________��
A�������¶� B�������¶�
C������NH4Cl���� D������Na2A����
��.����Cr2O72-�ķ�ˮ���Խϴ�ij������ˮ�к�5.0��10��3 mol��L��1��Cr2O72-��Ϊ��ʹ��ˮ���ŷŴ�꣬�������´�����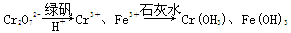
(1)�÷�ˮ�м����̷���H����������Ӧ�����ӷ���ʽΪ____________________��
(2)��������ķ�ˮ�в�����c(Fe3��)��2.0��10��13 mol��L��1���������Cr3����Ũ��Ϊ________��
(��֪��Ksp[Fe(OH)3]��4.0��10��38��Ksp[Cr(OH)3]��6.0��10��31)
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ͼ�Ӳ�ʺϽ��к�̼����(WC)��������(Co)�����������������õ�ⷨ�ɻ���WC��Co���������̼�ͼ���£�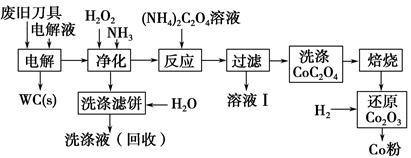
(1)���ʱ�Ͼɵ������������������������HCl��ҺΪ���Һ��������Ҫ�ĵ缫��ӦʽΪ ��
(2)�������������˱�����Ҫ�ɷ��� �����յ�ϴ��Һ����ˮ���Ƶ��Һ��Ŀ���ǻ����������е� ��
(3)��Һ�����Ҫ�ɷ��� ��ϴ��CoC2O4����ֶ����ղ�Ʒ���Ȳ�������Ӱ�죬������ʱ����ɻ�����Ⱦ��ԭ���� ��
(4)��Co2O3��ԭ��Co�۵Ļ�ѧ��Ӧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
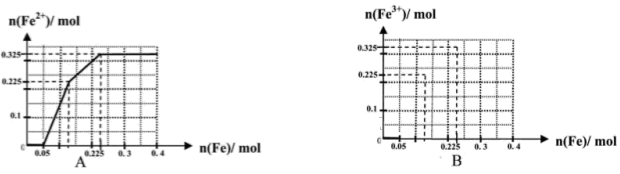
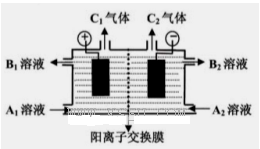
��һ����8�֣��ں������ʵ�����Ϊ0.1 mol FeCl3��H2SO4��Cu(NO3)2����Һ500 mL�У��������ۣ���Һ��n(Fe2��)�����n(Fe)�Ĺ�ϵ��ͼA(��ע��ʶͼ������ͼ��ÿС������Ϊ0.05mol)������ʾ����������ˮ�������Ӱ�죻��������������ԭʱ��������һ���������壩
��1��д��ͼ��n(Fe)��0.125mol-0.225 mol�ζ�Ӧ��Ӧ�����ӷ���ʽ____��
��2��д��ͼ��n(Fe)��0-0.05 mol�ζ�Ӧ��Ӧ�����ӷ���ʽ____��
��3������ͼB�л�����Һ��n(Fe3��)�����n(Fe)�ı仯��ϵͼ
��4������Ӧ���е����ȡ����Һ2mL�����Թ��У�Ϊ��֤������Һ����NO3�����ڣ��������Թ����ڵμ�_______��
| A������KMnO4��Һ | B�����������KSCN��Һ | C����ˮ | D������������Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���绷�����˽���ȫ���ֹʹ��������������ˮ����������������ø�Ч����ɫ���������������ȡ�����������һ�ּ��ױ�ը��ǿ���������壬������ˮ�����ȶ����ʻ���ɫ����������ʹ��ʱ���뾡����ϡ���������ϡ�ͣ�ͬʱҪ������ա�����ȡ�ʵ�����Ե�ⷨ�Ʊ�ClO2���������£���ش��������⡣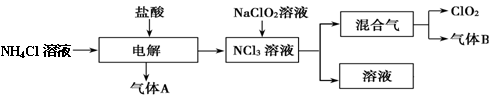
��1��ClO2������ԭ�� ����ǡ����ǡ���������8���ӽṹ����ͼ��ʾ��ⷨ�ƵõIJ�������������B��ʹʯ����Һ����ɫ����ȥ���������ѡ�� ��
A������ʳ��ˮ B����ʯ�� C��Ũ���� D������ˮ
��2���ȶ��Զ���������Ϊ�ƹ�������ȶ����������Ͳ�Ʒ������˵����ȷ���� ��
A���������ȿɹ㷺���ڹ�ҵ������ˮ����
B��Ӧ����ʳƷ��ҵ������Ч���ӳ�ʳƷ������
C���ȶ��Զ������ȵij��ִ�������˶������ȵ�ʹ�÷�Χ
D���ڹ������ͳ�Ʒ�������ڣ�Ҫ��ͨ��װ�úͼ�⼰����װ��
��3��ŷ������Ҫ��������������Ũ�����Ʊ�����ѧ��Ӧ����ʽΪ ���˷�ȱ����Ҫ�Dz��ʵ͡���Ʒ���Է��룬��������Ⱦ������
��4���ҹ��㷺���þ��������ϡ�͵�����������������ƣ�NaClO2����Ӧ�Ʊ�����ѧ����ʽ�� ���˷����ŷ�������ŵ��� ��
��5����ѧ��������о�����һ���µ��Ʊ����������������ữ�IJ��ᣨH2C2O4����Һ��ԭ�����ƣ���ѧ��Ӧ����ʽΪ ���˷���������������桢����İ�ȫ�ԣ�ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
I.���ݷ�Ӧ3Cu+8HNO3 3Cu(NO3)2+2NO��+4H2O���ش��������⣺
��1������������������ Ԫ�ر�������
��2���á�˫���š���ʾ�÷�Ӧ����ת�Ƶķ������Ŀ��
3Cu+8HNO3=3Cu(NO3)2+2NO��+4H2O
��3������2 mol HNO3����ԭʱ�������������ʵ�����Ϊ��������ת�Ƶĵ�����Ϊ������
II. ������6�����ʣ�K2SO4��K2SO3��I2��H2SO4��KIO3��H2O���һ��������ԭ��Ӧ����֪�ڷ�Ӧ��K2SO3ʧȥ���ӡ��뽫��6���������һ����ѧ����ʽ������ƽ����
2002��9��26�գ��й�������˾���Ƴ��˵�һ������ҹ���ȫ����֪ʶ��Ȩ�ĸ�����CPU оƬ�� ����о��1�š���оƬ����Ҫ�ɷֿ����Ƴɼ���������������������·���ϣ��������ڵ��µIJ� �������¶�ڼ��������У�������·�������ӷ���ʽ˵��ԭ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�⻯��ͭ(CuH)��һ����������,��CuSO4��Һ�͡���һ���ʡ���40��50 ��ʱ��Ӧ����������CuH���ȶ�,�ֽ�;����������ȼ��;��ϡ���ᷴӦ����������;Cu+�����������·����ķ�Ӧ��2Cu+ Cu2++Cu������������Ϣ,����Լ������յĻ�ѧ֪ʶ,�ش�:
Cu2++Cu������������Ϣ,����Լ������յĻ�ѧ֪ʶ,�ش�:
(1)��CuSO4��Һ�͡���һ���ʡ���CuH�ķ�Ӧ��,��������ԭ�۵����,�⡰��һ���ʡ��ڷ�Ӧ�������������������������������������
(2)д��CuH��������ȼ�յĻ�ѧ��Ӧ����ʽ
(3)CuH�ܽ���ϡ���������ɵ���������������,�����Ӧ�������˱�״����22.4 L������,����ԭ�����ӵõ��ӵ����ʵ���������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��.�ס���Ԫ�صĵ��ʺͻ�����Ӧ�ù㷺��
��1����Ԫ�ص�ԭ�ӽṹʾ��ͼ�� ��
��2��������뽹̿��ʯӢɰ���,�ڵ�¯�м��ȵ�1 500 �����ɰ���,��ӦΪ:
2Ca3��PO4��2+6SiO2 6CaSiO3+P4O10
6CaSiO3+P4O10
10C+P4O10 P4+10CO
P4+10CO
ÿ����1 mol P4ʱ,���� mol���ӷ���ת�ơ�
��3����������ƣ�Na2S2O3���dz��õĻ�ԭ������ά����C����ѧʽC6H8O6����ˮ��Һ�м������I2��Һ,ʹά����C��ȫ����,ʣ���I2��Na2S2O3��Һ�ζ�,�ɲⶨ��Һ��ά����C�ĺ����������ķ�ӦΪ:
C6H8O6+I2 C6H6O6+2H++2I-
C6H6O6+2H++2I-
2S2 +I2
+I2 S4
S4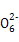 +2I-
+2I-
��һ�������ijά����C��Һ�м���a mol/L I2��ҺV1 mL,��ַ�Ӧ��,��Na2S2O3��Һ�ζ�ʣ���I2,����b mol/L Na2S2O3��ҺV2 mL������Һ��ά����C�����ʵ����� mol��
��4����������Һ��,����أ�KIO3�����������ƿɷ������·�Ӧ:
2I +5S
+5S +2H+
+2H+ I2+5S
I2+5S +H2O
+H2O
���ɵĵ�����õ�����Һ����,���ݷ�Ӧ��Һ������ɫ�����ʱ���������÷�Ӧ�����ʡ�
ijͬѧ���ʵ�����±���ʾ:
| | 0.01 mol/LKIO3������Һ�������ۣ������/mL | 0.01 mol/LNa2SO3��Һ�����/mL | H2O�����/mL | ʵ���¶�/�� | ��Һ������ɫʱ����ʱ��/s |
| ʵ��1 | 5 | V1 | 35 | 25 | |
| ʵ��2 | 5 | 5 | 40 | 25 | |
| ʵ��3 | 5 | 5 | V2 | 0 | |
 Ce��OH��4��+ ��
Ce��OH��4��+ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ʢ��KI��Һ���Թ��м�������CCl4��μ���ˮ��CCl4������ɫ������������Թ��еμ���ˮ����CCl4�����dz���������ɫ��
���������գ�
��1��д������ƽCCl4������ɫ�����ɫ�Ļ�ѧ��Ӧ����ʽ(���ϵ����1��������д)��
��2�����������еĻ�ԭ����________________��
��3����KI����KBr����CCl4����________ɫ�������μ���ˮ��CCl4�����ɫû�б仯��Cl2��HIO3��HBrO3��������ǿ������˳���� ��
��4���ӵ����к�����Ϊ25.4mg��50 mg/kg����ȡ�ӵ���(��KIO3��ʳ��)1000 kg������KI��Cl2��Ӧ��KIO3��������Ҫ����Cl2 L(��״��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ԫ��ʮ����Ҫ���䵥�ʼ��仯�����ڿ��С�������������ʮ�ֹ㷺����;��
��1����Ԫ�غ�������Ų�ʽΪ�� ��ԭ�Ӻ���� �ֲ�ͬ�ܼ��ĵ��ӣ���ԭ�Ӻ���������� �ֲ�ͬ����չ������ �ֲ�ͬ�˶�״̬�ĵ��ӡ�
��2�����¡��˹��̵������о����������³�ѹ�����������£�N2�ڴ���������ˮ������Ӧ��
2N2 (g)+6H2O (l) 4NH3 (g)+3O2 (g) - Q �������Ӧ��ƽ�ⳣ��Kֵ��÷�Ӧ ��ѡ���ţ���
4NH3 (g)+3O2 (g) - Q �������Ӧ��ƽ�ⳣ��Kֵ��÷�Ӧ ��ѡ���ţ���
A��һ��������Ӧ�����ƶ�
B����ƽ���ƶ�ʱ����Ӧ������������С
C��һ�����淴Ӧ�����ƶ�
D����ƽ���ƶ�ʱ�淴Ӧ�����ȼ�С������
��3�����������Ӧ�����������ܱ������н��У�����Ӧ�ﵽƽ��������жϴ������
A�������������ƽ������������ʱ����仯 B��ͨ�����ϡ����������߷�Ӧ������
C��������������ܶȲ���ʱ����仯 D��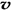 ��N2��/
��N2��/ 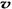 ��O2��=2:3
��O2��=2:3
��4�����������Ϊ���ȼ�ϵ���Ҫ�����塣��������ڸ�ѹ��450���������Ѹ�ٷֽ⣬��Ӧ�Ļ�ѧ����ʽ4NH4ClO4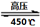 6H2O��+ 2N2��+ 4HCl��+ 5O2�����������������뻹ԭ��������ʵ���֮���� ��ÿ�ֽ�1mol������泥�ת�Ƶĵ�����Ŀ�� ��
6H2O��+ 2N2��+ 4HCl��+ 5O2�����������������뻹ԭ��������ʵ���֮���� ��ÿ�ֽ�1mol������泥�ת�Ƶĵ�����Ŀ�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com