
����22�֣�
��4�֣�����ʵ������У���������
A��ʵ��������ϩʱ���ھƾ���Ũ����Ļ��Һ�У����뼸Ƭ���Ƭ�����Ȼ���ʹҺ���¶�Ѹ������170��
B����֤������ˮ�����ʱ���������������������Һ��ϣ��������Һ�����ã���Һ��ֲ�μ���������Һ
C����ͭ˿�������״���ھƾ����ϼ��ȱ�ں�����������ˮ�Ҵ��У�����Ҵ�����Ϊ��ȩ��ʵ��
D�������еμ�����ϡ��ˮ���������������鱽��
E����ҵ�ƾ���ȡ��ˮ�ƾ�ʱ���ȼ���ʯ��Ȼ������������뽫�¶ȼƵ�ˮ������뷴ӦҺ�У��ⶨ��ӦҺ�¶�
��10�֣�ʵ��������ͼ��ʾװ���Ʊ��屽������֤�÷�Ӧ��ȡ����Ӧ��

(1) �ر�F��������C��������װ��������������ƿ����A�ڼ��������壬�ټ���������м����סA�ڣ�������ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ�� ��
(2) D�Թ���װ���� ���������� ��
(3) E�Թ���װ���� ��E�Թ��ڳ��ֵ�����Ϊ ��
(4) ��������ƿ�еķ�Ӧ��������ʱ(��ʱ�������Լ���)����F�������ر�C���������Կ����������� ��
(5) ��һ���õ����屽��Ҫ�����²������ƣ�
a���� bˮϴ�� c�ø������� d 10%NaOH��Һϴ�ӣ� eˮϴ
��ȷ�IJ���˳����
��8�֣�������ͼ��ʾװ�ý���ʵ�飬��A��μ���B�У�

��1�� ��BΪNa2CO3��ĩ��CΪC6H5ONa��Һ��ʵ���й۲쵽С�Թ�����Һ�ɳ������ǣ����Թ�C�л�ѧ��Ӧ�����ӷ���ʽ��________________ ��Ȼ�����ձ��м����ˮ���ɹ۲쵽�Թ�C�е����� ��
��2�� ��B����ʯ�ң��۲쵽C��Һ�����γɳ�����Ȼ������ܽ�.��������ȫ�ܽ⣬ǡ�ñ����ʱ���ر�E.Ȼ����С�Թ��м���������ȩ��Һ�������ձ��м�����ˮ������Ƭ�̣��۲쵽�Թܱڳ��ֹ�������������A�� �������ƣ���C�� ���ѧʽ��������ȩ�Ļ�Ϻ���Һ�з�Ӧ�Ļ�ѧ����ʽ�� _____________������D�ڴ�ʵ���е������� ��
��AC����ȫ��ȷ��4�֣����һ����2�֣��д���𰸵�0�֣�
��1�� (2��)
(2��)
(2) CCl4����1�֣����ջӷ����ı��������塣��1�֣�
(3) AgNO3��Һ(1��)�����ܿڲ���������������1�֣����ɵ���ɫ��������1�֣�
(4) ˮ������������ƿ�У�1�֣�
(5) bdeca (��edbca)��2�֣�
��1��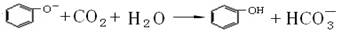 (2��)��Һ�ɻ��DZ���塣��1�֣�
(2��)��Һ�ɻ��DZ���塣��1�֣�
��2�� Ũ��ˮ��1�֣���AgNO3(1��)��CH3CHO��2Ag(NH3)2OH CH3COONH4��H2O��2Ag����3NH3 (2��)��ֹ������1�֣�.
CH3COONH4��H2O��2Ag����3NH3 (2��)��ֹ������1�֣�.
����������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ÿ��2�֣���22�֣�
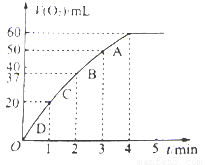
I.ʵ����0.01mol/L��KMnO4��������Һ��0.1mol/L��H2C2O4��Һ�������Ϻ�Ӧ���ʦ�[mol/(L �� s)]�뷴Ӧʱ��t��s���Ĺ�ϵ��ͼ��ʾ���ش��������⣺
��1���÷�Ӧ�Ļ�ѧ����ʽ��
��2��0��t2ʱ����ڷ�Ӧ���������ԭ���ǣ� ��
��3��t2��tʱ����ڷ�Ӧ���ʼ�С��ԭ���ǣ� ��
��4��ͼ����Ӱ���֡��������ʾt1��t3ʱ���� ��
����Mn2+���ʵ���Ũ�ȵ����� B��Mn2+���ʵ���������
C��SO42-���ʵ���Ũ�� D��MnO4-���ʵ���Ũ�ȵļ�С
II. Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ����������ͼ�ס�����ʾ��ʵ�顣��ش�������⣺
��1�����Է���������ͼ�ɹ۲� �����ԱȽϵó����ۡ���ͬѧ�����FeCl3��ΪFe2(SO4)3��Ϊ�������������� ��
��2����������������ͼ����ʾ��ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ͼ������A������Ϊ ��ʵ������Ҫ������������
��3������0.01mol MnO2��ĩ��60mL H2O2��Һ�У��ڱ�״���·ų����������ʱ��Ĺ�ϵ��ͼ��ʾ����ų�����������ΪV mL��
�ٷų�V/3 mL����ʱ����ʱ��Ϊ min��
�� ��H2O2��Һ��Ũ��Ϊ
��A��B��C��D���㷴Ӧ���ʿ�����˳��Ϊ > > >
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ӱ�ʡ������ѧ10-11ѧ���һ��ѧ����ĩ���ԣ���ѧ����B�� ���ͣ�ʵ����
��ÿ��2�֣���22�֣�
I.ʵ����0.01mol/L��KMnO4��������Һ��0.1mol/L��H2C2O4��Һ�������Ϻ�Ӧ���ʦ�[mol/(L �� s)]�뷴Ӧʱ��t��s���Ĺ�ϵ��ͼ��ʾ���ش��������⣺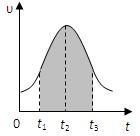
��1���÷�Ӧ�Ļ�ѧ����ʽ��
��2��0��t2ʱ����ڷ�Ӧ���������ԭ���ǣ� ��
��3��t2��tʱ����ڷ�Ӧ���ʼ�С��ԭ���ǣ� ��
��4��ͼ����Ӱ���֡��������ʾt1��t3ʱ���� ��
����Mn2+���ʵ���Ũ�ȵ����� B��Mn2+���ʵ���������
C��SO42-���ʵ���Ũ�� D��MnO4-���ʵ���Ũ�ȵļ�С
II.Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ����������ͼ�ס�����ʾ��ʵ�顣��ش�������⣺
��1�����Է���������ͼ�ɹ۲� �����ԱȽϵó����ۡ���ͬѧ�����FeCl3��ΪFe2(SO4)3��Ϊ�������������� ��
��2����������������ͼ����ʾ��ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ͼ������A������Ϊ ��ʵ������Ҫ������������
��3������0.01mol MnO2��ĩ��60mL H2O2��Һ�У��ڱ�״���·ų����������ʱ��Ĺ�ϵ��ͼ��ʾ����ų�����������ΪV mL��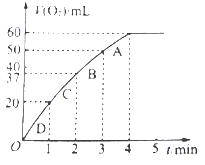
�ٷų�V/3 mL����ʱ����ʱ��Ϊ min��
�ڸ�H2O2��Һ��Ũ��Ϊ
��A��B��C��D���㷴Ӧ���ʿ�����˳��Ϊ > > >
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɽ��ʡɽ���и�һ5���¿���ѧ�Ծ���A������������ ���ͣ�ʵ����
����22�֣�ijʵ��С��������װ�ý������µ�ʵ�顣
��1��ʵ�������ͭ�����ֺ�ɫ�ͺ�ɫ�����������д����Ӧ�Ļ�ѧ����ʽ
�� ��
ʵ��С���ڲ��Ϲ������������£�Ϩ��ƾ��ƣ����ַ�Ӧ���ܼ������У�˵���÷�Ӧ�� �� ���� ��Ӧ��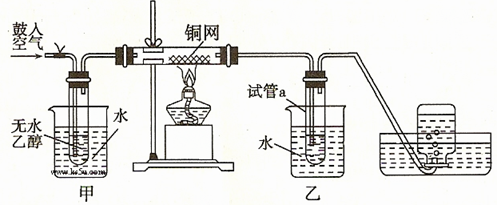
��2����������ˮԡ���ò���ͬ���������� ��
�ҵ������� ��
��3����Ӧ����һ��ʱ������ԇ��a�����ռ�����ͬ�����ʣ���ô�ռ������л����ǣ�����д��ṹ��ʽ�� ������ƿ���ռ��������������Ҫ�ɷ��� ������д����ķ���ʽ��
��4�����Թ�a���ռ�����Һ������ɫʯ����ֽ���飬��ֽ�Ժ�ɫ��ʵ��С���ͬѧ��Ϊ������Ǵ��ڸ��������ᡣ��ȥ�����ʣ�����ʹ�ó����г�����һ�ֻ����������Ϊ  ������д��ѧʽ�����ʽ��
������д��ѧʽ�����ʽ��
��5����д������ʵ���漰���л����Ҵ������������е�һ�־���Ӧ�ã� ��
��6����ʵ���Ŀ���ǣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���ӱ�ʡ10-11ѧ���һ��ѧ����ĩ���ԣ���ѧ����B�� ���ͣ�ʵ����
��ÿ��2�֣���22�֣�
I.ʵ����0.01mol/L��KMnO4��������Һ��0.1mol/L��H2C2O4��Һ�������Ϻ�Ӧ���ʦ�[mol/(L �� s)]�뷴Ӧʱ��t��s���Ĺ�ϵ��ͼ��ʾ���ش��������⣺

��1���÷�Ӧ�Ļ�ѧ����ʽ��
��2��0��t2ʱ����ڷ�Ӧ���������ԭ���ǣ� ��
��3��t2��tʱ����ڷ�Ӧ���ʼ�С��ԭ���ǣ� ��
��4��ͼ����Ӱ���֡��������ʾt1��t3ʱ���� ��
����Mn2+���ʵ���Ũ�ȵ����� B��Mn2+���ʵ���������
C��SO42-���ʵ���Ũ�� D��MnO4-���ʵ���Ũ�ȵļ�С
II. Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ����������ͼ�ס�����ʾ��ʵ�顣��ش�������⣺
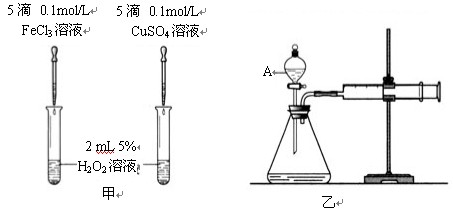
��1�����Է���������ͼ�ɹ۲� �����ԱȽϵó����ۡ���ͬѧ�����FeCl3��ΪFe2(SO4)3��Ϊ�������������� ��
��2����������������ͼ����ʾ��ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ͼ������A������Ϊ ��ʵ������Ҫ������������
��3������0.01mol MnO2��ĩ��60mL H2O2��Һ�У��ڱ�״���·ų����������ʱ��Ĺ�ϵ��ͼ��ʾ����ų�����������ΪV mL��
�ٷų�V/3 mL����ʱ����ʱ��Ϊ min��
�� ��H2O2��Һ��Ũ��Ϊ
��A��B��C��D���㷴Ӧ���ʿ�����˳��Ϊ > > >
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ������и�һ��ѧ�����п��Ի�ѧ�Ծ� ���ͣ������
��ÿ��1�֣���4�֣���ͨ��״���£���ͬѧȡ1 mol H2O���ȵ�100��ʱ��Һ̬ˮ������Ϊˮ��������ͼ��ʾ�����ù������� �仯��
�ڱ���ѹǿ���������£�ˮ��������� 
���������������������22.4L��
����ͬѧ��H2��O2��ȼ�յ�ʵ�飬��ʵ��������� �仯���ڸñ仯�����У�һ��������ȵ��� ������ţ���
| A����Ӧ�������Ŀ�������������Ŀ | B����Ӧ��ԭ�������ʵ�����������ԭ�������ʵ��� |
| C����Ӧ���������������������� | D����Ӧ���������������� |

 �ۿ̶��� ��Ũ�� ���ݻ�
�ۿ̶��� ��Ũ�� ���ݻ��鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com