
(15��)��ú������ƷC8H10�ϳɸ߷��Ӳ���I��·�����£�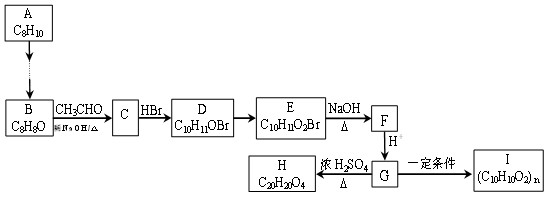
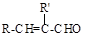 ��֪����1��B��C��D���ܷ���������Ӧ���ұ����ϵ�һ±���������֡�H����������Ԫ����
��֪����1��B��C��D���ܷ���������Ӧ���ұ����ϵ�һ±���������֡�H����������Ԫ����
��2��RCHO��R'CH2CHO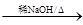 �� H2O
�� H2O
�����������������գ�
��1��A�������� ; D�к��й����ŵ����� ��
��2��D��E�ķ�Ӧ������ ��
��3��C��H�Ľṹ��ʽ�ֱ�Ϊ �� ��
��4��E��F�Ļ�ѧ����ʽ�� ��
��5��G��I�Ļ�ѧ����ʽ�� ��
��6����������������G��ͬ���칹���� �֣�
д����������һ�ֵĽṹ��ʽ
A���ܷ���������Ӧ��ˮ�ⷴӦ����1mol��ͬ���칹��������2mol��NaOH
B�����б�������������FeCl3��ɫ��
C��������

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(��С��15��)�ö������ȣ�ClO2���������ƣ�Na2FeO4Ħ������Ϊ166 g��mol��1�������;�ˮ�������ͳ�ľ�ˮ��Cl2�Ե�ˮ���������dz�������ˮ�����¼�����ClO2��Na2FeO4��ˮ���������зֱ𱻻�ԭΪCl����Fe3����
(1)����Ե�λ���������������õ��ĵ���������ʾ����Ч�ʣ���ô��ClO2��Na2FeO4��Cl2��������ɱ����������Ч���ɴ�С��˳���� �� �� ��
(2)������֮�����ܾ�ˮ��������������ǿ�������⣬��һ��ԭ������ǣ�
��

(3)����������һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ��59�棬�е�Ϊ11.0�棬������ˮ��ClO2���Կ����������ᣨHClO2�������ᣨHClO3���Ļ����������ҵ�����Գ�ʪ��KClO3�Ͳ�����60��ʱ��Ӧ�Ƶá�ijѧ������ͼ��ʾ��װ��ģ�ҵ��ȡ���ռ�ClO2������AΪClO2�ķ���װ�ã�BΪClO2������װ�ã�CΪβ������װ�á����ʣ�
��A���ֻ�Ӧ�����¶ȿ��ƣ���ˮԡ���ȣ�װ�ã�B���ֻ�Ӧ����ʲôװ�� ��
��C��Ӧװ���Լ�Ϊ ��C�з�����Ӧ�Ļ�ѧ����ʽΪ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����������ѧ����8���¿���ѧ�Ծ����������� ���ͣ�ʵ����
(15��)����ͼװ�ý���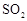 ת��Ϊ
ת��Ϊ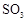 ��ת���ʲⶨʵ�飺
��ת���ʲⶨʵ�飺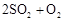
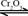
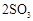 ��
��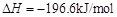 ����֪��
����֪��
| | �۵㣨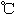 �� �� | �е㣨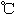 �� �� |
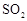 | -72.4 | -10 |
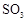 | 16.8 | 44.3 |

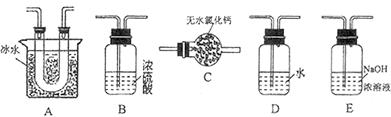
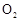 ��Ϊʹ
��Ϊʹ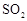 �нϸߵ�ת���ʣ�ʵ��ʱ����Ũ��������ȴ������Ⱥ�˳���� ��
�нϸߵ�ת���ʣ�ʵ��ʱ����Ũ��������ȴ������Ⱥ�˳���� ��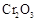 �������ʱ��Ӧ���ƾ����ƿ�һ����ټ��ȡ����Է��¶ȹ��ߣ���������ԭ���� �����ô�������
�������ʱ��Ӧ���ƾ����ƿ�һ����ټ��ȡ����Է��¶ȹ��ߣ���������ԭ���� �����ô�������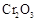 ���ķ�Ӧ��ʱ��
���ķ�Ӧ��ʱ��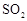 ��ת���ʻ� ������ߡ��������͡����䡱����
��ת���ʻ� ������ߡ��������͡����䡱����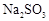 �������Ũ�����������ģ���Ӧ����ʱ����ͨ��
�������Ũ�����������ģ���Ӧ����ʱ����ͨ�� һ��ʱ�䣬�Ƶâ�װ�õ���������11.3g����ʵ����
һ��ʱ�䣬�Ƶâ�װ�õ���������11.3g����ʵ���� ��ת����Ϊ %������С�����һλ����
��ת����Ϊ %������С�����һλ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ�����ڶ����������ۻ�ѧ�Ծ��������棩 ���ͣ������
(15��)��ú������ƷC8H10�ϳɸ߷��Ӳ���I��·�����£�
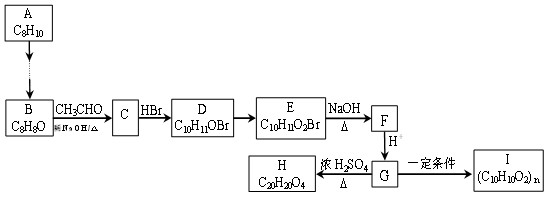
 ��֪����1��B��C��D���ܷ���������Ӧ���ұ����ϵ�һ±���������֡�H����������Ԫ����
��֪����1��B��C��D���ܷ���������Ӧ���ұ����ϵ�һ±���������֡�H����������Ԫ����
��2��RCHO��R'CH2CHO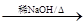 �� H2O
�� H2O
�����������������գ�
��1��A�������� ; D�к��й����ŵ����� ��
��2��D��E�ķ�Ӧ������ ��
��3��C��H�Ľṹ��ʽ�ֱ�Ϊ �� ��
��4��E��F�Ļ�ѧ����ʽ�� ��
��5��G��I�Ļ�ѧ����ʽ�� ��
��6����������������G��ͬ���칹���� �֣�
д����������һ�ֵĽṹ��ʽ
A���ܷ���������Ӧ��ˮ�ⷴӦ����1mol��ͬ���칹��������2mol��NaOH
B�����б�������������FeCl3��ɫ��
C��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(��С��15��)�ö������ȣ�ClO2���������ƣ�Na2FeO4Ħ������Ϊ166 g��mol��1�������;�ˮ�������ͳ�ľ�ˮ��Cl2�Ե�ˮ���������dz�������ˮ�����¼�����ClO2��Na2FeO4��ˮ���������зֱ𱻻�ԭΪCl����Fe3����
(1)����Ե�λ���������������õ��ĵ���������ʾ����Ч�ʣ���ô��ClO2��Na2FeO4��Cl2��������ɱ����������Ч���ɴ�С��˳���� �� �� ��
(2)������֮�����ܾ�ˮ��������������ǿ�������⣬��һ��ԭ������ǣ� ��

(3)����������һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ��59�棬�е�Ϊ11.0�棬������ˮ��ClO2���Կ����������ᣨHClO2�������ᣨHClO3���Ļ����������ҵ�����Գ�ʪ��KClO3�Ͳ�����60��ʱ��Ӧ�Ƶá�ijѧ������ͼ��ʾ��װ��ģ�ҵ��ȡ���ռ�ClO2������AΪClO2�ķ���װ�ã�BΪClO2������װ�ã�CΪβ������װ�á����ʣ�
��A���ֻ�Ӧ�����¶ȿ��ƣ���ˮԡ���ȣ�װ�ã�B���ֻ�Ӧ����ʲôװ�� ��
��C��Ӧװ���Լ�Ϊ ��C�з�����Ӧ�Ļ�ѧ����ʽΪ�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com