
| ������ | NaF | MgF2 | SiF4 |
| �۵�/K | 1266 | 1534 | 183 |
| | C��O | C=O | C��O |
| CO | 357��7 | 798��9 | 1071��9 |
| | N��N | N=N | N��N |
| N2 | 154��8 | 418��4 | 941��7 |
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
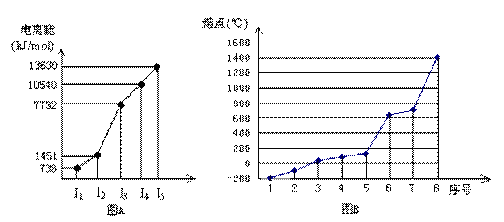
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

| A���٢ۢ� | B���ڢۢ� | C���ۢ٢� | D���ڢ٢� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
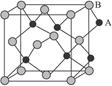
| A��AB | B��A2B | C��AB2 | D��A2B3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������Ԫ����ɵķ�����һ��ֻ�м��Լ� |
| B��ԭ�Ӿ����й��ۼ�Խǿ���۵�Խ�� |
| C�������з��Ӽ�������Խ����Խ�ȶ� |
| D���ɼ��Թ��ۼ���ϵķ���һ���Ǽ��Է��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��O2��S��Br2 | B��CO2��KCl��SiO2 | C��Na��W��NaCl | D��H2O��H2S��H2Se |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������þ | B���Ȼ��ƾ��� | C���� | D������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��22.4LCO2��22.4L CO������ԭ����Ŀ֮�� |
| B����Na2O2�������������������ӵ����ʵ���֮�� |
| C����ͬ�¶��£�0.2mol��L-1������Һ��0.1mol��L-1�����е�c��H+��֮�� |
| D��Һ����ڡ�0���̶�ʱ��50ml��ʽ�ζ��ܺ�25ml��ʽ�ζ�����ʢ��Һ�����֮�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com