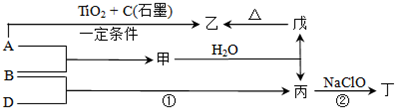
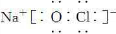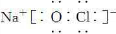�ס��ҡ�����������Ϊ���ֶ�����Ԫ�أ���ԭ���������������붡��������ֱ�ͬ���壬���ԭ�������������ȴ������2���������ҿ�����ԭ�Ӹ�����3��1�γɻ�����A����ÿ��A�����к���10�����ӣ���ش�
��1�����ԭ�ӽṹʾ��ͼ��
���ҵ��ʷ��ӵĵ���ʽ��
��
��2��A����ˮ������Һ�ʼ��Ե�ԭ���ǣ��õ��뷽��ʽ��ʾ��
NH3?H2O?NH4++OH-
NH3?H2O?NH4++OH-
��
��3�����붡���γ�һ�����ӻ�����û�������H
2O��Ӧ�õ�ǿ����Һ��H
2����÷�Ӧ�У��������뻹ԭ�������ʵ���֮����
1��1
1��1
��
��4��X��Y��ZΪ����ǿ����ʣ��ֱ�����������Ԫ���е�������ɣ�X��Y��Z��ϡ��Һ֮���������ת����ϵ��
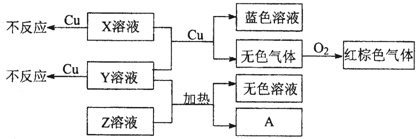
��Y��Z��Ϻ���ȵõ�A���ӷ���ʽ��
��
�ڽ���Cu��X��Y�Ļ����Һ��Ӧ�����ӷ���ʽ��
3Cu+8H++2NO3-=3Cu2++2NO��+4H2O
3Cu+8H++2NO3-=3Cu2++2NO��+4H2O
��



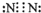
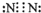

 ���ҵ���ΪN2������ʽΪ
���ҵ���ΪN2������ʽΪ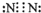 ��
�� ��
��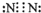 ��
��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�