
ij��ȤС���������ͼʵ��װ�ý���ʵ�顣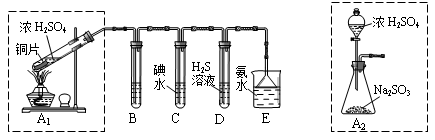
��̽��������Ⱦ��SO2������
��1��Ϊ��ʵ����ɫ������Ŀ�꣬�ܷ�����ͼA2����A1װ�� ����ܡ�����
��2��B��C��D�ֱ����ڼ���SO2��Ư���ԡ���ԭ�Ժ������ԣ���B����ʢ�Լ�
Ϊ ��C�з�Ӧ�����ӷ���ʽΪ ��D�з�Ӧ�Ļ�ѧ����ʽ
Ϊ ��
��̽��ͭƬ��ŨH2SO4��Ӧ�IJ���
ʵ�������������ͭƬ���渽�ź�ɫ���塣�������ϵ�֪���˺�ɫ�����
�ܺ���CuO��CuS��Cu2S��������CuS��Cu2S��������ϡ���ᣬ�ڿ��������ն�ת��ΪCu2O��SO2����С��ͬѧ�ռ�һ������ɫ���壬������ʵ�鷽��̽����ɷ֣�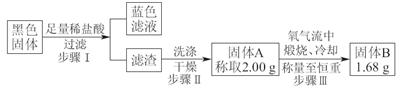
��3������� �м�������ϴ�Ӹɾ���ʵ�鷽����____________________________��
��4����ɫ����ijɷ���________________��
�������
�ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH-��SO32-��SO42-��HSO3-�������ӡ�
��5����ˮ���չ���SO2�ķ�Ӧ�����ӷ���Ϊ ��
��1���ܣ�1�֣�
��2��Ʒ����Һ��1�֣� SO2��I2��2H2O��SO42-��2 I-��4H+��2�֣� SO2��2H2S="3S��+" 2H2O��2�֣�
��3��ȡ���һ��ϴ��Һ�������μ�����AgNO3��Һ����������������ϴ�Ӹɾ���2�֣���
��4��CuO��CuS��Cu2S��2�֣�
��5��NH3��H2O��SO2��NH4+��HSO3-��2�֣�
���������������1��A2װ�ò���Ҫ���ȣ��ҿ��Կ��Ƽ���Ũ����������Ʒ�Ӧ���У�����������2������SO2Ư����һ����Ʒ���Լ���C��SO2��ⵥ�ʷ�Ӧ��D����H2S��Ӧ���з�Ӧ����3��ȡ���һ��ϴ��Һ�������μ�����AgNO3��Һ����������������ϴ�Ӹɾ���ǿ�����һ��ϴ��Һ����4�����ݲ���������ɫ��Һ�ó����庬��CuO������������պ������ı仯���㣺��ȫΪCuS�����õ�����������ӦΪ2��96��2��144=1��5g����ȫΪCu2S�����õ�����������ӦΪ2��160��144=1��8g�����������ʾ����С���5������SO2Ӧ������������李�
���㣺����ʵ�鼰ʵ��������й����⡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��������ƣ�Na2S2O3�������������ƺ����ͨ�����Ϸ�Ӧ�Ƶá���֪��Na2S2O3��������Һ�в����ȶ����ڡ�
��1��ij�о�С����Ƶ��Ʊ�Na2S2O3��5H2O��װ�úͲ��ֲ����������¡�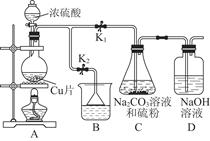
��.��K1�ر�K2����Բ����ƿ�м�������Ũ���ᣬ���ȡ�
��.C�л��Һ��������������Ӧһ��ʱ�����۵������٣���C����Һ��pH �ӽ�7ʱ����K2���ر�K1��ֹͣC�еķ�Ӧ��ֹͣ���ȡ�
��.����C�еĻ��Һ��
��.����Һ���� �� �����ˡ�ϴ�ӡ���ɣ��õ���ƷNa2S2O3��5H2O��
�٢��У�����C����Һ��pH�ӽ�7ʱ��ֹͣC�еķ�Ӧ����ԭ���� �������ӷ���ʽ��ʾ����
�ڢ��У����������� �� ��
��װ��B��ʢ�ŵ��Լ��ǣ��ѧʽ�� ��Һ��
����һС����ʵ���з��֣�����������������������º����������ԣ����ֲ��������������⣬�����Ʋ���ܵ�ԭ�� ��
��2������Na2S2O3��Һ�ⶨ��ˮ��Ba2����Ũ�ȣ��������£�ȡ��ˮ25.00 mL�������ʵ�����ȼ������� K2Cr2O7��Һ����BaCrO4���������ˡ�ϴ�Ӻ�������ϡ�����ܽ⣬��ʱCrO42-ȫ��ת��ΪCr2O72-���ټӹ�KI��Һ����ַ�Ӧ��û����ҺV mL������ƽ���ֳ�4�ȷݣ����������Һ��ָʾ������0.001 0 mol��L��1��Na2S2O3��Һ���еζ�����Ӧ��ȫʱ��������ݼ�¼���±���ʾ��
| ��� | 1 | 2 | 3 | 4 |
| ����Na2S2O3�� | | | | |
| ��Һ�����/mL | 18.02 | 17.98 | 18.00 | 20.03 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���������������Ҫ��������������������й㷺Ӧ�á�̽�������������ȡ�����ʶ����ŷdz���Ҫ�����塣
��1����ҵ���û�����FeS2������SԪ��Ϊ��1�ۣ��ڸ����º�������Ӧ�Ʊ�SO2��
�÷�Ӧ�б�������Ԫ����__________����Ԫ�ط��ţ������÷�Ӧת��5.50mol����ʱ�����ɵĶ��������ڱ�״���µ����Ϊ________L��
��2����ʵ���ҿ���70����Ũ������������Ʒ�Ӧ��ȡ������������ܿ��Ʒ�Ӧ�ٶȣ���ͼ�п�ѡ�õķ���װ����_______����д��ĸ����
������������������Ʒ�Ӧ��ȡ3.36L����״�������������������4.0���������ƣ������������������������ƣ����������ȡ����������_______g������һλС������
��3��ij��ѧ��ȤС���������ͼװ����֤��������Ļ�ѧ���ʡ�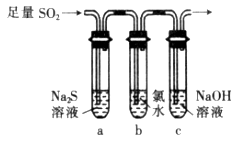
����˵������������������Ե�ʵ������Ϊ___________________________��
��Ϊ��֤��������Ļ�ԭ�ԣ���ַ�Ӧ��ȡ�Թ�b�е���Һ�ֳ����ݣ��ֱ��������ʵ�飺
���������һ����Һ����Ʒ����Һ����ɫ��ȥ
��������ڶ�����Һ����BaCl2��Һ��������ɫ����
���������������Һ�м���AgNO3��Һ���а�ɫ��������
���������к�������________������������Թ�b�з�����Ӧ�����ӷ���ʽΪ______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʵ�������ö������̸�Ũ���ᷴӦ�Ʊ����﴿��������������������ͼ��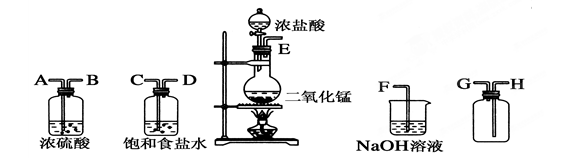
(1)����������������ȷ˳��(����ӿڴ�����ĸ)��__��__��__��__��__��__��__��__����2�֣�
(2)װ���У�����ʳ��ˮ��������______________��NaOH��Һ��������____________����4�֣�
(3)д�����л�ѧ��Ӧ�ķ���ʽ��
�����巢��װ���н��еķ�Ӧ��________________________________________����2�֣�
��NaOH��Һ�з����ķ�Ӧ��__________________________________________ ����2�֣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ��ʵ������ȡ���ռ�Cl2��װ�á�A��Cl2����װ�ã���E��Ӳ�ʲ�������װ��ͭ˿����FΪ����Ĺ��ƿ���ձ�GΪβ������װ�á�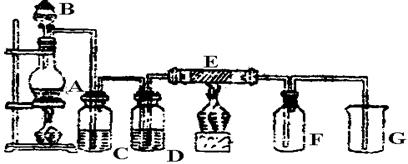
�Իش�
��1��A�з����Ļ�ѧ��Ӧ�����ӷ���ʽΪ ��
��2��C��G�и�װ��ҩƷ��C__________��G__________��
��3��E��Ӧ����ʽΪ ��
��4��д��G�з�Ӧ�����ӷ���ʽ ��
��5������Cl2��SO2�����ʵ������Ư�����ü������û�ѧ����ʽ����ԭ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijͬѧ�������װ���Ʊ����ռ��������Ȼ�����Ӳ�ʲ�����E��װ��ϸ��˿����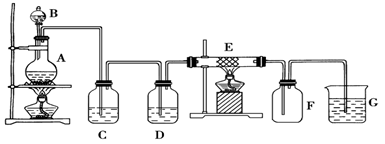
�Իش�
��1������װ��A�������Եķ�����
��2��װ��A�з�Ӧ�Ļ�ѧ����ʽΪ
��3��װ��C�������ǣ� ________________��װ��D�е��Լ��ǣ�____ _______��
��4���ɼ���װ��E�����ɵ������������ӵķ����������� ��
��5����������ͨ��ʯ����Һ�У��۲�������ǣ� ��
��6��װ�� G�з�����Ӧ�����ӷ���ʽΪ��________________ _ ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧ��ȤС����ʵ������ȡƯ�ۣ���̽��������ʯ���鷴Ӧ�������Ͳ��
��֪���ٶ���������Ũ���ᷴӦ���Ʊ�������ͬʱ����MnCl2��
�������ͼ�ķ�Ӧ�Ĺ����зų��������¶Ƚϸ�ʱ�������ͼ�ܷ������·�Ӧ��6Cl2 + 6Ca(OH)2 5CaCl2 + Ca(ClO3)2 + 6H2O
5CaCl2 + Ca(ClO3)2 + 6H2O
����ȤС�����������ʵ��װ�ã�����ʵ�顣
�� �� �� ��
��ش��������⣺
��1���ټ�װ���з�����Ӧ�Ļ�ѧ����ʽ�� ��
����װ���е��Լ��� �������� ��
�۸���ȤС����300mL 12mol/L������17.4g MnO2�Ʊ��������������������������ʯ���鷴Ӧ���������������Ƶñ�������� L��Ca(ClO)2 g��
��2��С���Ա���֣�������Ca(ClO)2����������С������ֵ���������ۺ���Ϊ����������δ��ʯ���鷴Ӧ���ݳ����Լ��¶������ǿ���ԭ��Ϊ��̽����Ӧ�����Բ����Ӱ�죬������ȡһ������ʯ���飬���������ٵ�ͨ�������������ó���ClO����ClO3�� �������ӵ����ʵ�����n���뷴Ӧʱ�䣨t���Ĺ�ϵ���ߣ����Ա�ʾΪ��ͼ��������������ˮ�ķ�Ӧ����
��ͼ�����ߢ��ʾ ���ӵ����ʵ����淴Ӧʱ��仯�Ĺ�ϵ��
����ȡʯ�����к���Ca(OH)2�����ʵ���Ϊ mol��
����ȡһ����ڵ����ʵ���Ca(OH)2��ʯ���飬�Խϴ������ͨ��������������Ӧ���ò�����Cl�������ʵ���Ϊ0.35mol��������� = ��
= ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ����ȡ��ˮ�Ȼ�ͭ��ʵ��װ��ͼ����Ũ����μӵ�ʢ�ж������̷�ĩ��Բ����ƿ�С���ش��������⣺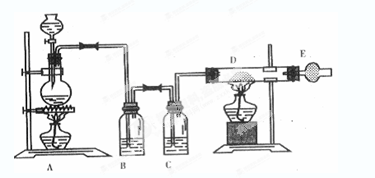
(1)ʢ��Ũ�������������Ϊ�ߣߣߣ���
(2)��ƿ�з�����Ӧ�Ļ�ѧ����ʽ ��
(3)Cƿ�е��Լ����ߣߣߣ������������ߣߣߣ���
(4)������D�з�����Ӧ�Ļ�ѧ����ʽ ����Ӧ�������ߣߣ���
(5)�����E��ʢ�м�ʯ��(CaO+NaOH)�����������ߣߣߣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���������ҹ���������᳧��ȡ�������Ҫԭ�ϡ�ij��ѧ��ȤС���ij������ʯ����Ҫ �ɷ�ΪFeS2)������Ԫ�غ����ⶨ��ʵ��̽������ҵ���������̽����
I ����m1,g�û�������Ʒ�������в������������������ͼ��ʾװ�ã��гֺͼ���װ�� ʡ�ԣ���ʯӢ����,��a�����ϵػ���ͨ���������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��4FeS2 + 11O2 2Fe2O3 + 8SO2
2Fe2O3 + 8SO2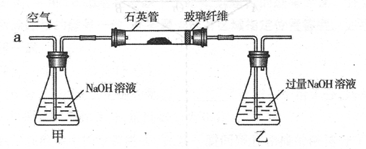
II��Ӧ������,����ƿ�е���Һ�������´���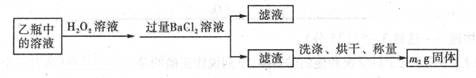
��1��I�У���ƿ�ڷ�����Ӧ�����ӷ���ʽ��____________��__________��
��2��II�У�����H2O2��Һ��������������____________________________
��3���û�����ʯ����Ԫ�ص���������Ϊ____________________________
��4�������ڴ���Ӧ���������Ƚ�������Ŀ�ģ�______________��
��5����ҵ�����г��ð��������ᷨ����β�������Դﵽ������Ⱦ���������õ�Ŀ��,�� ������ѧ����ʽ��ʾ�䷴Ӧԭ��_______________��______________
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com