
Ϊ̽���������Ƶ����ȶ��ԣ�ij�о���ѧϰС�齫��ˮ�������Ƹ���������
�ȣ����������Ⱥ�Ĺ�����������ͼ��ʾ��ʵ��װ�ý���ʵ�顣��ش������й����⣺
��1���������ϣ���ˮ�������Ƹ����������ȵ�600��ſ�ʼ�ֽ⣬�ҷֽ����ֻ������
������һ�ֹ��塣��������¶ȵ���600�棬��������ȴ����������л����μ�ϡ������
�������ڵμ�ϡ��������������� �����ʵ���Ũ�ȱ仯����Ϊ ��
�����ʵ���Ũ�ȱ仯����Ϊ ��
��2����������¶�Ϊ700�棬��������ȴ����������л����μ�ϡ�������������۲쵽��ƿ�г��ֵ���ɫ���������д������ݲ�������Ӧ���ɵ���ɫ���������ӷ���ʽΪ ����ʱ��B��C��װ���п��ܹ۲쵽������Ϊ ��
��3���ڣ�2���еμ������������ƿ�ڳ� �⣬��������һ��Ũ�Ƚϴ�������ӡ�Ϊ����������ӣ���ȡ������������ˮ�����Һ�������Ǽ���������ӵ�����ʵ�鷽������Ϊ�����ķ����� ����ס����ҡ�������˵����һ������������ԭ�� ��
�⣬��������һ��Ũ�Ƚϴ�������ӡ�Ϊ����������ӣ���ȡ������������ˮ�����Һ�������Ǽ���������ӵ�����ʵ�鷽������Ϊ�����ķ����� ����ס����ҡ�������˵����һ������������ԭ�� ��
�����ף�ȡ����������Һ���Թ��У��ȼ�ϡ ���ټ�
���ټ� ��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
�����ң�ȡ����������Һ���Թ��У��ȼ�ϡHCl���ټ� ��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
��4��д�� ������ȵ�600�����Ϸֽ�Ļ�ѧ����ʽ ��
������ȵ�600�����Ϸֽ�Ļ�ѧ����ʽ ��
��1������������С ��2�֣�
��2��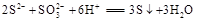 ��2�֣���
��2�֣���
B��Ʒ����Һ��ɫ��C�������Ա仯�������ݣ�2��,���B�������Ա仯�������ݣ�C�в�����ɫ���������� ��B��Ʒ����Һ��ɫ��C�в�����ɫ���������÷֣���
��3���ң�1�֣��������ȼ������ǿ�����Ե�ϡ �����ܽ�
�����ܽ�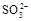 ������
������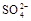 �������ж��Ƿ�����
�������ж��Ƿ�����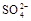 ��2�֣���
��2�֣���
��4��4Na2SO3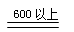 Na2S +3Na2SO4��2�֣�
Na2S +3Na2SO4��2�֣�
���������������1�������¶ȵ���600�棬��������û�зֽ⣬����ϡ���ᷴӦ�ֲ����У�SO32-+H+=HSO3-
HSO3-+H+=SO2��+H2O����Ũ������������٣���2�������������ȷֽ⣬����������ԭ��Ӧ����Ӧ����ʽΪ4Na2SO3 = Na2S +3Na2SO4������������ֵĻ�ɫ����ӦΪS���ʣ������������µ�SO32-��S2-�Ĺ��з�Ӧ������������Ӧ��SO2��H2S�������߲����棬�����ղ���������ֻ��������һ�֣�����ΪB��Ʒ����Һ��ɫ��C�������Ա仯�������ݣ�����ΪSO2����B�������Ա仯�������ݣ�C�в�����ɫ����������ΪH2S������3�������ȼ������ǿ�����Ե�ϡ �����ܽ�
�����ܽ�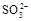 ������
������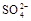 �������ж��Ƿ�����
�������ж��Ƿ�����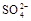 .��
.��
���㣺���黯ѧʵ���й����⡣


 ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʵ�����п�����ͼ��ʾװ����ȡ����ء��������ƺ�̽����ˮ�����ʡ�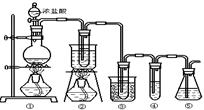
ͼ�У���Ϊ��������װ�ã��ڵ��Թ���ʢ��15mL30��KOH��Һ����������ˮԡ�У��۵��Թ���ʢ��15mL 8%NaOH��Һ�������ڱ�ˮԡ�У��ܵ��Թ��������ɫʯ����Һ����Ϊβ������װ�á�
����д���пհף�
��1����ȡ����ʱ������ƿ�����һ�����Ķ������̣�ͨ��______����д�������ƣ�����ƿ�м���������Ũ���ᡣʵ��ʱΪ�˳�ȥ�����е��Ȼ������壬���ڢ����֮�䰲װʢ��_________����д���б����ĸ���ľ���װ�á�
| A����ʯ�� | B������ʳ��ˮ | C��Ũ���� | D������̼��������Һ |
| ʵ������ | ԭ�� |
| ��Һ�������ɫ��Ϊ___ɫ | ������ˮ��Ӧ���ɵ�H+ʹʯ���ɫ |
| �����Һ��Ϊ��ɫ | ______________________________________ |
| Ȼ����Һ����ɫ��Ϊ___ɫ | _________________________________________ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ��ʵ�����Ʊ�����������һϵ�����ʵ���װ�ã��гּ������������ԣ���
��1���Ʊ�����ѡ�õ�ҩƷΪ����������̺�Ũ���ᣬ����ص����ӷ�Ӧ����ʽΪ ��
װ��B�б���ʳ��ˮ��������_________��ͬʱװ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������д����������ʱB�е����� ��
��2��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C��I��II��III���η��� ��
| | a | b | c | d |
| I | �������ɫ���� | �������ɫ���� | ʪ�����ɫ���� | ʪ�����ɫ���� |
| II | ��ʯ�� | �轺 | Ũ���� | ��ˮ�Ȼ��� |
| III | ʪ�����ɫ���� | ʪ�����ɫ���� | �������ɫ���� | �������ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ߴ��ȵ������ǵ��͵����ǽ������ϣ����Ʊ��뵼�����Ҫ���ϣ����ķ��ֺ�ʹ��������������һ�������������ߴ���ͨ�������·����Ʊ�����̼�ڸ����»�ԭ���������Ƶôֹ裨��Fe��Al��B��P�����ʣ����ֹ���������Ӧ�������Ȼ��裨��Ӧ�¶�Ϊ450��500�棩�����Ȼ��辭�ᴿ����������ԭ�ɵøߴ��衣������ʵ�����Ʊ����Ȼ����װ��ͼ��
�����Ϣ��a�����Ȼ�����ˮ����ˮ�⣻b���������������ڸ����¾���������ֱ�ӻ���������Ӧ���Ȼ��c���й����ʵ������������±���
| ���� | SiCl4 | BCl3 | AlCl3 | FeCl3 | PCl5 |
| �е�/�� | 57.7 | 12.8 | �� | 315 | �� |
| �۵�/�� | ��70.0 | ��107.2 | �� | �� | �� |
| �����¶�/�� | �� | �� | 180 | 300 | 162 |
| ���� | ����֮�� |
| �� | |
| �� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧС��������װ�ó�ȡ�ռ����������������о������ʡ�������������⡣
��1��װ��A�з�����Ӧ�����ӷ���ʽΪ_______________________________��
��2��������������������ӿڵ�����˳��Ϊa��___________________��g��
��3��װ��B��Ũ�����������__________________________��װ��C���Լ������___________________________________��
��4��ijͬѧ��Ϊ��������ȱ��β������װ�ã���������ķ����л�����װ�ò�ע���Լ���
| |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ��һλѧ����Ƶ�ʵ������ȡ���ռ���������֤�����ʵ�װ��ͼ��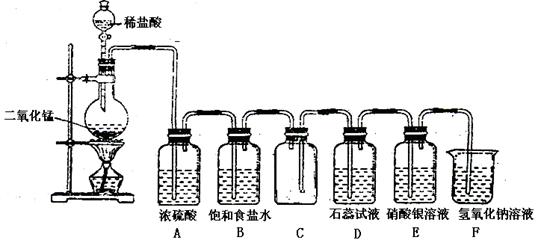
��1���ҳ��ڴ�װ��ͼ�г��ֵĴ�����Ը�����
��2��ʵ������У���Dװ���й۲쵽��ʵ�������� ����Eװ���й۲쵽��ʵ�������� ����Ӧ�Ļ�ѧ����ʽ�� ��
��3��Fװ�õ������� ����Ӧ�����ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ̽��ij��̼�Ͻ���Ũ�����ڼ��������µķ�Ӧ�IJ��ֲ��ﲢ�ⶨ��̼�Ͻ�����Ԫ�ص�����������ij��ѧ�С���������ͼ��ʾ��ʵ��װ�ã����������ʵ��̽����
|
|

 ��1����Բ����ƿ�м���m g��̼�Ͻ𣬲��������Ũ���ᣬδ��ȼ�ƾ���ǰ��A��B��������������ԭ���ǣ��ٳ����£�Fe��Ũ�����жۻ����� ��
��1����Բ����ƿ�м���m g��̼�Ͻ𣬲��������Ũ���ᣬδ��ȼ�ƾ���ǰ��A��B��������������ԭ���ǣ��ٳ����£�Fe��Ũ�����жۻ����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ѧϰС��ͨ���������Ϸ�����KClO3��Ũ����Ҳ�����Ʊ�������Ϊ�˸�С��������ͼװ����ʵ������ȡCl2��̽�����й����ʣ�
��1��װ��A�д���һ������������� ��
��2��д��װ��A�з�����Ӧ�Ļ�ѧ����ʽ�� ��
��3��Ϊ�˳�ȥ�����е��Ȼ��⣬��Ҫ��.A��B֮��װ��������װ��E��װ��E����ʢװ���Լ�Ϊ ��Ϊ��̽��ʹ��ɫ������ɫ������������������ˮ��Ӧ���������Ҫ������װ��������װ��E�����м���Ũ���ᣬװ��E������װ���е�����λ��Ϊ ��ѡ����ţ���
a A��B֮�� b��B��C֮��
c��C��D֮�� d��D֮��
��4��ȡ�¼���ƿB������˿����������״̬�����뵽����ƿB�У����Թ۲쵽�������� ���ܼ���÷�Ӧ��������Ԫ�صļ�̬���Լ� ��ѡ����ţ���ѡ����
a����ˮ b������ˮ������
c������ˮ������������Һ d������ˮ��KSCN��Һ
��5������һ����ѧѧϰС��ͻ�����������ָ��������װ�õ�ȱ�ݣ��������˸Ľ���ʩ���øĽ���ʩ�ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�±����������У�������ͼ����һ��ת����ϵ��ѡ���ǣ� ��
| ѡ�� | X | Y | Z |
| A | Na | NaOH | NaHCO3 |
| B | Cu | CuSO4 | Cu��OH��2 |
| C | C | CO | CO2 |
| D | Si | SiO2 | H2SiO3 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com