
 ����ͼ��ʾʵ��װ����ȡ�����������ش��������⣺
����ͼ��ʾʵ��װ����ȡ�����������ش��������⣺


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 �������ʵ�飺������ͼ��ʾ��װ����ȡ�����飻�ڽ��������������ʵ�飮���Թ�I�����μ���2mL ����ˮ��4mLŨ���ᡢ2mL 95%���Ҵ���3g�廯�Ʒ�ĩ�����Թܢ���ע������ˮ�����ձ���ע������ˮ�������Թ�I����״̬�����Ӻ���ȴ��
�������ʵ�飺������ͼ��ʾ��װ����ȡ�����飻�ڽ��������������ʵ�飮���Թ�I�����μ���2mL ����ˮ��4mLŨ���ᡢ2mL 95%���Ҵ���3g�廯�Ʒ�ĩ�����Թܢ���ע������ˮ�����ձ���ע������ˮ�������Թ�I����״̬�����Ӻ���ȴ��| �� |
| �� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ʵ���ҿ�������ͼ��ʾ��װ����ȡ�����������ش��������⣺
ʵ���ҿ�������ͼ��ʾ��װ����ȡ�����������ش��������⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
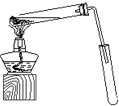 ij��ѧʵ��С������ͼ��ʾ��װ����ȡ������������������ �� �� �����Ƿ����������ʣ�����̨�����ӵ�֧������ʡ�ԣ�����֪���������ķе�Ϊ77.1�棬�Ҵ��е�Ϊ78.4�棬����ķе�Ϊ118�森�����Ҫ����գ�
ij��ѧʵ��С������ͼ��ʾ��װ����ȡ������������������ �� �� �����Ƿ����������ʣ�����̨�����ӵ�֧������ʡ�ԣ�����֪���������ķе�Ϊ77.1�棬�Ҵ��е�Ϊ78.4�棬����ķе�Ϊ118�森�����Ҫ����գ�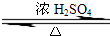 CH3COOCH2CH3+H2O
CH3COOCH2CH3+H2O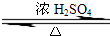 CH3COOCH2CH3+H2O
CH3COOCH2CH3+H2O�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 �����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺
�����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺ CH3CO18OCH2CH3+H2O
CH3CO18OCH2CH3+H2O CH3CO18OCH2CH3+H2O
CH3CO18OCH2CH3+H2O�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�����ɹŸ߶��ڶ�ѧ����ĩ���Ի�ѧ���� ���ͣ�ʵ����
�����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ������������ش��������⣺

��1���������Թ���ʢ�ŵ����Ҵ��� ��3�֣��� ��3�֣�����ȡ���������Ļ�ѧ����ʽΪ�� ��4�֣���
��2��Ũ����������� ����ˮ����3�֣���
��3������̼������Һ����Ҫ�����ǣ� ��4�֣���
��4��װ����ͨ�����ĵ���Ҫ���ڱ���̼������Һ��Һ ��֮�ϣ����ܲ�����Һ�У�Ŀ���Ƿ�ֹ ��3�֣���
��5����Ҫ���Ƶõ������������������Ӧ���õ�ʵ������� ��3�֣���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com