
��ҵ���û���̿�������������ˮ�е�I-��ȡI2������������ͼ��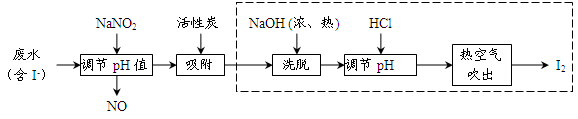
��1�����ˮ�м���NaNO2������pH<4��д����Ӧ�����ӷ���ʽ__________________��
��2���û���̿�������ɵĵ��ʵ��Ŀ����__________________________����Ũ����NaOHϴ�������ĵ�Ļ�ѧ����ʽΪ___________________________________��
��3���ӵ���pH�����õ���Һ�л�ȡ�⣬��ҵ�Ͽ����ȿ�����������ԭ����___________����ȡ��������Һ��ʵ�����з���õ����ʵ⣬��Ҫ���еIJ���Ϊ__________________��
��4�����������ɵ�NOβ���ж�����ҵ�Ͻ�����O2��һ������ͨ��NaOH�Ʊ�NaNO2��д����Ӧ�Ļ�ѧ����ʽ��________________________________��
��5����ҵ����������̻�������ͼ�������棬������ͼ���̵��ŵ�___________________________��д��һ������
��1��2NO2-+ 4H++ 2I- �� 2NO + I2 + 2H2O ��2�֣�
��2���������ʵ⣨1�֣� 3I2 + 6NaOH �� NaIO3+ 5NaI+ 3H2O��2�֣�
��3�����ʵ���ˮ���ܽ��С����������1�֣� ��ȡ ��Һ ����3�֣�
��4��4NaOH+ O2+ 4NO �� 4NaNO2+ 2H2O��2�֣�
��5�������˻�ѧҩƷ��Ͷ������ʹ�ɱ���͡������������ڼ��٣���ҩƷ���豸��ʴ�Եȣ���1�֣��������ɣ�
���������������1����������ͼ�ż���NaNO2ʱ��NO���ɣ�������������ԭ��Ӧ���������ӷ���ʽ2NO2-+ 4H++ 2I- �� 2NO + I2 + 2H2O��
��2���û���̿�������ɵĵ��ʵ��Ŀ���Ǹ������ʵ⡣��������ͼ��������ʱ���е����ɣ���֪��Ũ����NaOHϴ�������ĵ�Ļ�ѧ����ʽΪ3I2 + 6NaOH �� NaIO3+ 5NaI+ 3H2O��
��3���ӵ���pH�����õ���Һ�л�ȡ�⣬��ҵ�Ͽ����ȿ�����������ԭ���ǵ��ʵ���ˮ���ܽ��С������������ȡ��������Һ��ʵ�����з���õ����ʵ��Ⱥ���еIJ���Ϊ ��ȡ����Һ������
��4������������ԭ��Ӧԭ������Ӧ�Ļ�ѧ����ʽ4NaOH+ O2+ 4NO �� 4NaNO2+ 2H2O��
��5������Դ�����úͻ�������˼�����𣬿����Ǽ����˻�ѧҩƷ��Ͷ������ʹ�ɱ���͡������������ڼ��٣���ҩƷ���豸��ʴ�Եȣ�
���㣺���⿼�����ӷ���ʽ����д�����ʵ��ᴿ��ʵ�鷽�������ۡ�


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��δ�ʯ���л�ø��������ȼ��һֱ�ǻ�ѧ��̽���Ŀ��⣬��ʯ�ͷ���õ������ͽ����ѻ����Ի�ø��������ȼ�͡�
����һ��ʯ���Ǻ���20��30��̼ԭ�ӵ������Ļ��������³ʹ�̬��
���϶���ʯ�ʹ��ѻ���ͨ��ʹ��Al2O3��������
ij�о���ѧϰС����ʵ������ģ��ʯ�͵Ĵ��ѻ���װ����ͼ��ʵ������пɹ۲쵽��ƿ�й���ʯ�����ۻ����Թܢ���������Һ�����ᣬ�Թܢ������Ը��������Һ��ɫ��ʵ������Թܢ���Һ����ζ���������͵���ζ��
(1)��װ���������ӵ�˳��Ӧ��ѭ��ԭ��Ϊ__________________��
Ϊ��֤ʵ��ɹ���ʵ��ǰ������еIJ�����_________________��
װ���нϳ����ܵ�������_____________________��
(2)�Թܢ�������Һ������˵����______��
(3)�Թܢ�����Һ��ɫ˵����________________��
(4)�ܷ����Թܢ��е�Һ����ȡ��ˮ�е��壬������_______________��
(5)д����ʮ���ѻ��õ������ϩ�Ļ�ѧ����ʽ________________��
(6)ʯ���ѻ�����Ҫ������_____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��9�֣����������( Na2S2O3)�����������Լ������ﻹԭ���������ȡ������ֽ⡣��ҵ�Ͽ��÷�Ӧ��2Na2S+Na2CO3+4SO2=3Na2S2O3 +CO2�Ƶá�ʵ����ģ��ù�ҵ���̵�װ����ͼ��ʾ��
�ش��������⣺
��1��b�з�Ӧ�����ӷ���ʽΪ__________________��c���Լ�Ϊ_____________________��
��2����Ӧ��ʼ��c�����л��Dz��������ֱ���塣�˻�������____________________��
��3��d�е��Լ�Ϊ____________��
��4��ʵ����Ҫ����SO2�������ʣ����Բ�ȡ�Ĵ�ʩ��______________________��д����������
��5��Ϊ�˱�֤��������ƵIJ�����ʵ����ͨ���SO2,���ܹ�����ԭ����_______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���������ؾ���Kx[Fe��C2O4��y]��3H2O��������Ӱ����ɫӡˢ��ʵ�����Ʊ����������ؾ����ʵ���������£�
��1����������ϡ�����Ʊ�FeSO4��7H2O��������______������������___________________________________________________��
��2�����������У���������һ������FeC2O4��2H2O��������ˮϴ�ӡ���������Ƿ�ϴ�Ӹɾ��ķ�����_________________________________________________________________��
��3���ⶨ���������ز�Ʒ��Fe3��������C2O42-������ʵ�鲽�����£�
����1��ȷ��ȡ���Ʋ��������ؾ���a g��Լ1.5 g�������250 mL����Һ��
����2������Һ����ȡ25.00 mL����Һ����ƿ�У�����6 mol��L��1 HCl 10 mL��������70��80 �棬������SnCl2TiCl3���ϻ�ԭ����Fe3��ȫ����ԭΪFe2��������MnSO4��Һ10 mL����75��80 ������0.010 00 mol��L��1 KMnO4����Һ�ζ����յ㣨Cl�������뷴Ӧ������C2O42-ȫ��������CO2��Fe2��ȫ��������Fe3����¼�����
����3������
����4���ظ���������2������3���Ρ�
�ٲ���2����ʱ������Ҫʹ����ͼ��ʾ�����е�________������ţ���
�ڲ���2��MnSO4��Һ��������________���ζ��յ��������______________________________________��
���ڲ������Լ������ǰ���£�����3��Ŀ����_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о���ѧϰС������H2��ԭ���ⶨ����ͭ��������CuO��Cu2O��Ϸ�ĩ��ͭԪ�ص�������������ͼ�Dzⶨװ�õ�ʾ��ͼ��A�е��Լ������ᡣ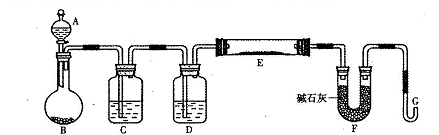
��ش��������⡣
��1����ƿ��װ����Լ���п�������н�������Ӧ�����ӷ���ʽ�� ��
��2���Լ�C��ˮ����������__ __��
��3���Լ�D��__ __���������Ǹ���������
��4�����Ӻ�װ�ú��ʵ�����������_ __�������в�����ţ�
�ټ����������ȣ��ڼ���E���ۼ���װ�������ԣ��ܵ�ȼG����������壻�ݴ�Aƿ��εμ�Һ��
��5����֪��C(s)+1/2O2( g)�� CO(g) ��H=��110.5kJ��mol��1,
Cu2O(s) +1/2O2(g) �� 2CuO(s) ��H=��145 kJ��mol��1
����̿���ڸ��������»�ԭCuO����Cu2O���Ȼ�ѧ����ʽ��____ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ҵ������м��ԭ���Ʊ��⻯�Ƶ���Ҫ��������ͼ��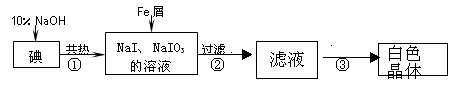
��1����Ԫ��λ�����ڱ��е� ���ڣ��� �壻
��2����Ӧ�ٵĻ�ѧ����ʽ ��
��3���жϷ�Ӧ���е��Ƿ�����ȫ��Ӧ�IJ����� ��
��4������Һ�ڵ����·����NaI��Ʒ�Ĺ����У�Ҫ��ֹNaI����������ȡ�Ĵ�ʩ����� ��
��5��ijͬ�Ʋⲽ����еõ��İ�ɫ������NaI��NaIO3��NaOH�Ļ���������·������м��飬ʵ�����������Ʋ���ȷ��
��֪��IO3-ʮ5I-+6H+=3I2+3H2O��NaIO3ˮ��Һ�����ԡ�
��ѡ�Լ���lmol��LH2SO4��2mol��LHNO3��������Һ����̪��Һ��ʯ����Һ������ˮ��������������Ʒ��ѡ��
| ʵ�鷽�� | ʵ������ | ���� |
| ����ɫ��������ˮ������2�ε�����Һ | �õ���ɫ��Һ |  |
| ȡ������ҺҺ���Թ�A�У� . | ��Һ����ɫ | ��Һ�к�IO3һ |
| ��ȡ������ҺҺ���Թ�B�У� . | . | ��Һ�к�����0Hһ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������һЩʵ�����Ʊ�����ͭ��ʵ����ơ�
ʵ��һ����ͭ��Ũ���ᷴӦ��ȡ����ͭ��
ʵ�����������Ũ����ֶ�μ��뵽ͭ����ϡ����Ļ�����У�����ʹ֮��Ӧ��ȫ��ȡ����ͭ����װ������ͼ�е�ͼI��ͼ��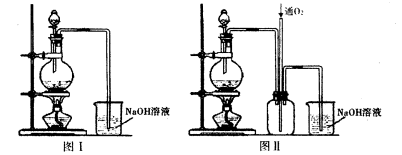
ʵ��������ͭ����ij�����з������գ�ʹͭ�۳�������������Ӧ��������ͭ���ٽ�����ͭ��ϡ���ᷴӦ������������ɵõ�����ͭ���塣
ʵ���ģ���ͭ�ۺ�ϡ����Ļ�����м���H2O�õ�����ͭ��
��ش�ʵ���е��й����⡣
��1��ʵ��һ�з�����Ӧ�Ļ�ѧ��Ӧ����ʽΪ____�����ַ�����ȱ����____��
��2��ʵ���װ��ͼ�з�Һ©����ʢװ��Һ����____��ͼ����ͼI�ĸĽ�װ�ã�����ͼIIװ�õ��ŵ��� ��
��3��ʵ�������������յ����������� ����ʵ���еĺ�������ָ����__ ��
��4��ʵ�����з�����Ӧ�Ļ�ѧ����ʽΪ____��
��5���������ʵ������⣬���������һ��ֻ��ͭ��ϡ����Ϊԭ����ȡ����ͭ�ķ�����д����ѧ��Ӧ����ʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧС�������ͼװ��ģ���ⱥ��ʳ��ˮ�Ʊ�������ͨ��������ԭ����ͭ�ⶨCu�����ԭ��������ͬʱ���������������ԣ�ͼ�мгֺͼ��������Ѿ���ȥ����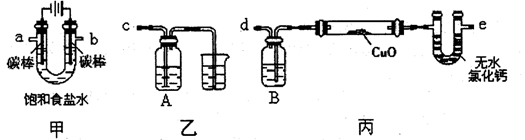
��1��д��װ�ü��з�Ӧ�����ӷ���ʽ ��Ϊ�������ʵ�飬��ȷ��������ʽΪa���� ��b���� ������ĸ����
��2����װ�����ձ���Һ��������� ��Aƿ��ʢװ����Һ����� ������ĸ����
a��I-������Һ b��NaOH��Һ
c��FeCl2��KSCN�����Һ d��Na2SO3��Һ
�ڼ���װ�ñ��е�����ͭ��ĩ֮ǰ������Ҫ����װ�õ������Ի�����еı�Ҫ������ ��
��3������װ�ñ��ⶨCu�����ԭ���������������ַ������ٲ�÷�Ӧǰ��ϴ��ƿB������Һ��������m1���ڲ�÷�Ӧǰ��U�ιܼ����й���������m2������Ϊ�����ķ���Ϊ ����١��ڡ����������ò�÷�Ӧ��Ӳ�ʲ�������ʣ����������m3�ķ�������֪O�����ԭ������Ϊ16��ʵ��������ͭ��Ʒ����Ϊm����ⶨCu�����ԭ�������ı���ʽΪ ���÷����ڷ�Ӧ��Ӳ�ʲ�������ȴ������û��һֱͨ�������ᵼ�²ⶨCu�����ԭ������ ���ƫ����ƫС������Ӱ�족���������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ��ѧ����С��Ϊ�˼���̼���ƺ�̼���������ְ�ɫ���壬�ò�ͬ�ķ�����������ʵ�飬��ͼ����ʾ��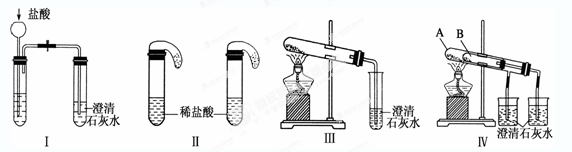
��1��ֻ����ͼ����ʾʵ�飬�ܹ��ﵽʵ��Ŀ�ĵ���(��װ�����)___ _______��
��2��ͼ����ʾʵ����ܼ������������ʣ��䶼�����ķ�Ӧ�Ļ�ѧ����ʽΪ_____________ ___________����ʵ�����ȣ�ʵ������ŵ���__________________________��
��3������ʵ�����֤̼���ƺ�̼�����Ƶ��ȶ��ԣ����Թ�B��װ��Ĺ��������________��
��4����ͬѧ��Ϊ�����������鷽���⣬�����ֹ�����ɵ���Һ�м������ʯ��ˮҲ�����������ǣ�����˵����_____________�����ȷ�������ġ�
��5����̼��������Һ�������ij���ʯ��ˮ��ϲ���ַ�Ӧ�������ӷ���ʽΪ_________________________����ʣ����Һ�����ʻ�ѧʽΪ___________
��6��������ƿ��Һ����֪�ֱ���K2CO3��NaHCO3������д�����ֲ�ͬ�ļ�����
��________________________________________________________________________��
��_______________________________________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com