




| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
[��ѧ��ѡ��ѧ�뼼��]
H2O2��ˮ��Һ��һ�ֳ��õ�ɱ������
��1��H2O2�ɵĹ�ҵ�Ʒ�����PtΪ������ʯīΪ����������������Һ���ٽ�������ˮ�⡣��ѧ����ʽΪ��
![]() ��д��������������Һ��������������Ӧ����
��д��������������Һ��������������Ӧ����
������ ��
������ ��
��2��H2O2�µĹ�ҵ�Ʒ����Ƚ��һ��������ԭ���ٽ��м����ȥ����������H2O2����Ӧ����ʽΪ
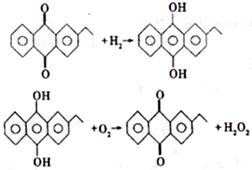
�һ������ڴ˱仯�����е������Լ���ɵĹ�ҵ�Ʒ�����¹�ҵ�Ʒ����ŵ���
��
��3��H2O2��ʵ�����Ʒ�֮һ�ǽ������������뵽ϡ�����У��÷�Ӧ�Ļ�ѧ����ʽΪ
��4��д��һ�ֶ����ⶨH2O2ˮ��Һ��H2O2�����Ļ�ѧ����ʽ
��5��д������(4)��Ӧԭ���ļ�Ҫʵ�鲽��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
[��ѧ��ѡ��ѧ�뼼��](15��)
H2O2��ˮ��Һ��һ�ֳ��õ�ɱ������
��1��H2O2�ɵĹ�ҵ�Ʒ�����PtΪ������ʯīΪ����������������Һ���ٽ�������ˮ�⡣��ѧ����ʽΪ��
![]() ��д��������������Һ��������������Ӧ����
��д��������������Һ��������������Ӧ����
������ ��
������ ��
��2��H2O2�µĹ�ҵ�Ʒ����Ƚ��һ��������ԭ���ٽ��м����ȥ����������H2O2����Ӧ����ʽΪ
�һ������ڴ˱仯�����е������Լ���ɵĹ�ҵ�Ʒ�����¹�ҵ�Ʒ����ŵ���
��
��3��H2O2��ʵ�����Ʒ�֮һ�ǽ������������뵽ϡ�����У��÷�Ӧ�Ļ�ѧ����ʽΪ
��4��д��һ�ֶ����ⶨH2O2ˮ��Һ��H2O2�����Ļ�ѧ����ʽ
��5��д������(4)��Ӧԭ���ļ�Ҫʵ�鲽��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ����ʦ���С�����ʦ���С�����ʡ2010������ڶ������������ۣ���ѧ���� ���ͣ������
[��ѧ��ѡ��ѧ�뼼��] (15��)
H2O2��ˮ��Һ��һ�ֳ��õ�ɱ������
��1��H2O2�ɵĹ�ҵ�Ʒ�����PtΪ������ʯīΪ����������������Һ���ٽ�������ˮ�⡣��ѧ����ʽΪ��
 ��д��������������Һ��������������Ӧ����
��д��������������Һ��������������Ӧ����
������ ��
������ ��
��2��H2O2�µĹ�ҵ�Ʒ����Ƚ��һ��������ԭ���ٽ��м����ȥ����������H2O2����Ӧ����ʽΪ

�һ������ڴ˱仯�����е������Լ���ɵĹ�ҵ�Ʒ�����¹�ҵ�Ʒ����ŵ���
��
��3��H2O2��ʵ�����Ʒ�֮һ�ǽ������������뵽ϡ�����У��÷�Ӧ�Ļ�ѧ����ʽΪ
��4��д��һ�ֶ����ⶨH2O2ˮ��Һ��H2O2�����Ļ�ѧ����ʽ
��5��д������(4)��Ӧԭ���ļ�Ҫʵ�鲽��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡģ���� ���ͣ������
 2NH3(g)����H= -92.4 kJ/mol��
2NH3(g)����H= -92.4 kJ/mol��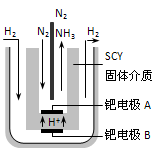
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com