
| A���ڢۢ� | B���ۢ� | C���٢ڢ� | D���٢ڢۢ� |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ݢ� | B���٢ڢ� | C���٢ڢݢ� | D�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ʵ��ʣ���ҩƷһ�㲻�ܷŻ�ԭ�Լ�ƿ |
| B���ܽ�ʱ�ò��������� |
| C����ȼ�������ȼǰ�����鴿 |
| D������ʱ����������������ֽ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ñ���Na2CO3��Һ�ɳ�ȥ������̼�е��Ȼ��� |
| B��ͭ���к�������������ͭ���ɼ���ϡ�������� |
| C�����ͻ�ú�ʹ���ڴ���������ɫ����ƿ�� |
| D������Ũ�����Ũ�������ʱ��Ӧ��Ũ���������ӵ�Ũ�����У�����ʱ�������ȴ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Բ����ƿ����ֱ���þƾ��Ƽ��� |
| B��������Ũ����Һմ��Ƥ���ϣ�Ҫ�����ô���ˮ��ϴ��Ȼ��Ϳ������ |
| C���ƾ����Ż�ʱ����ˮ���� |
| D������������Һʱ����������Ͳ�м���һ�������ˮ�����ڽ�����������������Ũ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��450 mL��52��7 g | B��500 mL��58��5 g |
| C��450 mL��58��5 g | D��500 mL��52��7 g |
�鿴�𰸺ͽ���>>
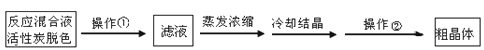
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 Na2S2O3��
Na2S2O3��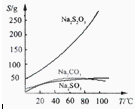
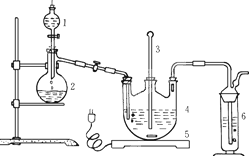
| A��BaCl2��Һ | B��ŨH2SO4 |
| C������KMnO4��Һ | D��NaOH��Һ |
 6I-+Cr2O72-+14H+=3I2+2Cr3++7H2O���ټ��뼸�ε�����Һ������������Na2S2O3��Һ�ζ���������Ӧ�� I2+2S2O32-=2I-+S4O62-���ζ��յ������Ϊ ��
6I-+Cr2O72-+14H+=3I2+2Cr3++7H2O���ټ��뼸�ε�����Һ������������Na2S2O3��Һ�ζ���������Ӧ�� I2+2S2O32-=2I-+S4O62-���ζ��յ������Ϊ ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������0��1mol��L-1��H2SO4��Һʱ����ȡŨ����С��ע��ʢ����������ˮ������ƿ�� |
| B����Ҫ��ȥCO2�к��е�����SO2���壬��ͨ��ʢ�б���NaHCO3��Һ��ϴ��ƿ |
| C����ʵ������ʹ�ý�����ʱ�������Ż𣬿�������ϸɳ��� |
| D������ҺpHʱ�����øɾ��IJ�����պȡ��Һ�ε����ڱ������ϵ�pH��ֽ�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com