
��18�֣�ij��ѧʵ��С��ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�
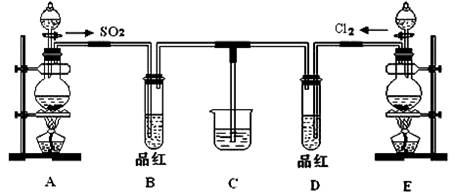
��1����װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2��ʵ������װ��E�Ʊ�Cl2���÷�Ӧ�Ļ�ѧ����ʽΪ ������Ӧ����1 mol��HCl����������Ӧ��ת�Ƶĵ�������Ϊ___ _______��
��3���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B ��
D ��
��ֹͣͨ�����ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ��
B ��
D ��
��4����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ���� ��
��5��SO2��Cl2�����ڴ�����Ⱦ���Ҫ����β����������װ��C��������װ��C�е��Լ���______________�������ƣ�����д��SO2��Cl2�ֱ�����Լ���Ӧ�����ӷ���ʽ ��
��
��1����Һ©���IJ�����û��ȡ������2�֣�
��2��MnO2+4HCl��Ũ��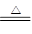 MnCl2+Cl2��+2H2O
��2�֣� 6.02��1023��2�֣�
MnCl2+Cl2��+2H2O
��2�֣� 6.02��1023��2�֣�
��3����Ʒ����Һ��ɫ��1�֣� Ʒ����Һ��ɫ��1�֣�
����ɫ��Ʒ����Һ�ָֻ��ɺ�ɫ��1�֣� ����������1�֣�
��4��Cl2+SO2+2H2O��2HCl+H2SO4��2�֣�
��5���������ƣ�2�֣� SO2��2OH����SO32����H2O��2�֣�
2OH�� + Cl2 = Cl�� + ClO�� + H2O��2�֣�
����������


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��18�֣�ij��ѧʵ��С��ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�
��1����װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2��ʵ������װ��E�Ʊ�Cl2���÷�Ӧ�Ļ�ѧ����ʽΪ ������Ӧ����1 mol��HCl����������Ӧ��ת�Ƶĵ�������Ϊ___ _______��
��3���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B ��
D ��
��ֹͣͨ�����ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ��
B ��
D ��
��4����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ���� ��
��5��SO2��Cl2�����ڴ�����Ⱦ���Ҫ����β����������װ��C��������װ��C�е��Լ���______________�������ƣ�����д��SO2��Cl2�ֱ�����Լ���Ӧ�����ӷ���ʽ ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡ����һ�и�һ��ѧ����ĩ������ѧ�Ծ���B������������ ���ͣ�ʵ����
��18�֣�ij��ѧʵ��С��ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�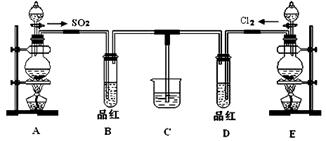
��1����װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B ��D ��
��ֹͣͨ�����ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ��
B ��D ��
��3����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ���� ��
��4��SO2��Cl2�����ڴ�����Ⱦ���Ҫ����β����������װ��C��������װ��C�е��Լ���_____�������ƣ�����д��SO2��Cl2�ֱ�����Լ���Ӧ�����ӷ���ʽ �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���������һ�и�һ��ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
��18�֣�ij��ѧʵ��С��ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�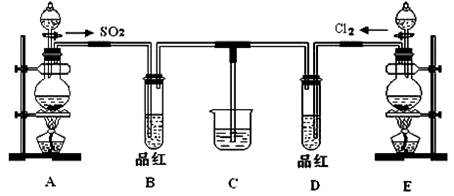
��1����װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2��ʵ������װ��E�Ʊ�Cl2���÷�Ӧ�Ļ�ѧ����ʽΪ ������Ӧ����1 mol��HCl����������Ӧ��ת�Ƶĵ�������Ϊ___ _______��
��3���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B ��
D ��
��ֹͣͨ�����ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ��
B ��
D ��
��4����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ���� ��
��5��SO2��Cl2�����ڴ�����Ⱦ���Ҫ����β����������װ��C��������װ��C�е��Լ���______________�������ƣ�����д��SO2��Cl2�ֱ�����Լ���Ӧ�����ӷ���ʽ ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015��ɽ��ʡ��һ��ѧ����ĩ������ѧ�Ծ���B���������棩 ���ͣ�ʵ����
��18�֣�ij��ѧʵ��С��ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�ã�
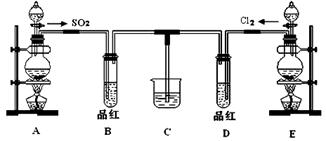
��1����װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�
B ��D ��
��ֹͣͨ�����ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�Ϊ��
B ��D ��
��3����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ���� ��
��4��SO2��Cl2�����ڴ�����Ⱦ���Ҫ����β����������װ��C��������װ��C�е��Լ���_____�������ƣ�����д��SO2��Cl2�ֱ�����Լ���Ӧ�����ӷ���ʽ �� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com