
 3Cu2++2NO��+4H2O
3Cu2++2NO��+4H2O Al(OH)3��+3NH4+
Al(OH)3��+3NH4+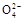 ����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����
����,��ô�ų���Ba2+�Ĵ���;���ڢ�������Cl-,���Ԣ���֤��ԭ��Һ���Ƿ�Cl-;���һ�����ڵ�������Al3+��NO3����SO42��,һ�������ڵ�������Fe3+��Ba2+��HCO3����


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| | �϶����ڵ����� | �϶�û�е����� | ����D | |
| ��ѧʽ�����ӷ��� | | | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ˮ�����c(OH��)=1��10��12mol/L����Һ�У�Ba2����K����Br����SiO32�� |
| B����������������Һ�У�SCN����Cl����K����NH4�� |
| C����ˮ�У�Na+��CO32����NH4����SO42�� |
| D��PH����7����Һ�� Na+��Ba2+��SO32����ClO�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A������ͨ��ˮ�У���Һ�����ԣ�Cl2+H2O 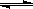 2H++Cl��+ClO�� 2H++Cl��+ClO�� |
| B����������������������Һ��Al+2OH-��AlO2-+H2�� |
| C����������Һ�еμ�Ba(OH)2��Һ����������ʵ������2Al3++3SO42-+3Ba2++6OH��=" 2" Al(OH)3��+3BaSO4�� |
| D������������ϡ���Fe+4H++NO3��=Fe3++NO��+2H2O |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ۢݢ� | B���ۢܢ� | C���ܢݢ� | D���٢ڢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ���� | �����Լ� | ʵ������ |
| ��һ����Һ | �μ������ĵ���KI��Һ | ��Һ����ɫ |
| �ڶ�����Һ | �μ��������ữ��BaCl2��Һ | �а�ɫ�������� |
| ��������Һ | �μ�NaOH��Һ�����ȣ������NaOH��Һ���(V)�����ɵij�������������������ʵ���(n)�Ĺ�ϵ����ͼ |  |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ������ | K����Na����Cu2����Al3�� |
| ������ | SO42����HCO3����NO3����OH�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��pH=0����Һ��:Na+��Fe2+��N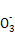 ��ClO- ��ClO- |
B��c(Fe3+)="0.1" mol/L����Һ��:K+��Ba2+��S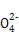 ��SCN- ��SCN- |
C��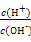 =1012����Һ��:N =1012����Һ��:N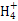 ��Al3+��S ��Al3+��S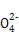 ��Cl- ��Cl- |
D����ˮ�����c(H+)=1��10-14 mol/L����Һ��:Ca2+��K+��Cl-��HS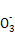 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com