
 ��У���˿��ֿ���ϵ�д�
��У���˿��ֿ���ϵ�д� �ƸԴ��ž�ϵ�д�
�ƸԴ��ž�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����a��3b��2c��/2 kJ?mol��1 | B����a��3b��2c��/6 kJ?mol��1 |
| C����a��3b��2c��/6 kJ?mol��1 | D����a��3b��2c��/3 kJ?mol��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����317.3 kJ/mol | B����379.3 kJ/mol |
| C����332.8 kJ/mol | D��317.3 kJ/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 H =-566kJ��mol��1 N2(g)+O2(g)="2NO" (g)
H =-566kJ��mol��1 N2(g)+O2(g)="2NO" (g)  H =+180kJ��mol��1��2CO(g)+2NO(g)=N2(g)+2CO2(g)��
H =+180kJ��mol��1��2CO(g)+2NO(g)=N2(g)+2CO2(g)�� H��
H��| A����386 kJ��mol��1 | B����386 kJ��mol��1 |
| C����746 kJ��mol��1 | D����746 kJ��mol��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| A������ | B��ϩ�� | C��Ȳ�� | D������ͬϵ�� |
 CH3OH(g)��H2O(g) ��H����49.0 kJ/mol��
CH3OH(g)��H2O(g) ��H����49.0 kJ/mol��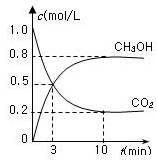
 CO2(g)+ 2H2O(l) ��H1���D 890.3 kJ/mol
CO2(g)+ 2H2O(l) ��H1���D 890.3 kJ/mol H2O(l) ��H2����285.8 kJ/mol
H2O(l) ��H2����285.8 kJ/mol| �ܽ��(S)/g | �ܶȻ�(Ksp) | ||
| Ca(OH)2 | Ba(OH)2 | CaCO3 | BaCO3 |
| 0.16 | 3.89 | 2.9��10-9 | 2.6��10-9 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
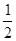 O2(g) �� H2O(g)����H1��a kJ��mol-1
O2(g) �� H2O(g)����H1��a kJ��mol-1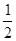 O2(g) ��H2��b kJ��mol-1
O2(g) ��H2��b kJ��mol-1| A��2b��c | B��b��285.8 |
| C��a����b? | D��������ȼ����Ϊa kJ��mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 ����(N2H4) Ϊȼ�ϡ���������Ϊ��ȼ������֪���и����ʷ�Ӧ���Ȼ�ѧ����ʽ��
����(N2H4) Ϊȼ�ϡ���������Ϊ��ȼ������֪���и����ʷ�Ӧ���Ȼ�ѧ����ʽ��| A��N2H4(g)+2H2O2(l)= N2(g)+4H2O(l)��H����817.63 kJ��mol��1 |
| B��N2H4(g)+2H2O2(l)= N2(g)+4H2O(g)��H����817.63 kJ��mol��1 |
| C��N2H4(g)+2H2O2(l)= N2(g)+4H2O(l)��H����641.63 kJ��mol��1 |
D��N2H4(g)+2H2O2(l)= N2(g)+4H2O( g)��H����641.63 kJ��mol��1 g)��H����641.63 kJ��mol��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com