
[s1] ��֪��H2��g��+F2��g��=2HF��g����H=-270kJ/mol������˵����ȷ���� �� ��
A��2L����������ֽ��1L������1L��������270kJ����
B��1mol������1mol ������Ӧ����2mol Һ̬������ų�����С��270kJ
C������ͬ�����£�1mol ������1mol �����������ܺʹ���2mol���������������
D��1������������1���������ӷ�Ӧ����2��������������ӷų�270kJ������
[s1]24��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��㶫ʡ�����и������в������ۻ�ѧ�Ծ��������棩 ���ͣ������
�Դ�����Ⱦ��SO2��NOx�����о�������Ҫ�������塣��ش��������⣺
��1��Ϊ����SO2���ŷţ�����ȡ�Ĵ�ʩ�ǽ�úת��Ϊ�������ȼ�ϡ�
��֪��H2��g����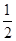 O2��g����H2 O��g�� ��H����241.8kJ��mol��1
O2��g����H2 O��g�� ��H����241.8kJ��mol��1
C��s����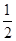 O2��g����CO��g�� ��H����110.5kJ��mol��1
O2��g����CO��g�� ��H����110.5kJ��mol��1
д����̿��ˮ������Ӧ���Ȼ�ѧ����ʽ�� ��
��2����֪����������NO�ķ�ӦΪ��N2��g����O2��g�� 2NO��g��
��H
2NO��g��
��H 0����1.0 mol������0.80 mol N2��0.20 mol O2��1300oCʱ��1.0 L�ܱ������ھ���5s��Ӧ�ﵽƽ�⣬���NOΪ8.0��10��4 mol��
0����1.0 mol������0.80 mol N2��0.20 mol O2��1300oCʱ��1.0 L�ܱ������ھ���5s��Ӧ�ﵽƽ�⣬���NOΪ8.0��10��4 mol��
��5s�ڸ÷�Ӧ��ƽ�����ʦͣ�NO�� �� ������2λ��Ч���֣�����1300oC ʱ���÷�Ӧ��ƽ
�ⳣ������ʽK�� ��
�����������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ��ԭ���� ��
��3������β����NO��CO��ת��������������һ��ʱ�������������ı���������ѧ��Ӧ���ʡ���ͼ��ʾ��������������ʱ����Ӧ2NO��g����2CO��g��
 2CO2��g����N2��g�� �У�NO��Ũ��
2CO2��g����N2��g�� �У�NO��Ũ��
c��NO�����¶ȣ�T���������������S����ʱ�䣨t���ı仯���ߡ�

�ٸ÷�Ӧ�ġ�H 0 ���������������
���������ı����S1��S2������ͼ�л���c��NO�� ��T1��S2�����´ﵽƽ������еı仯���ߣ�������Ӧ��ע����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������������09-10ѧ��߶���ѧ����ĩ���ԣ���ѧ�� ���ͣ������
[s1] ��֪25�桢101kpaʱ��һЩ���ʵ�ȼ����Ϊ��
|
��ѧʽ |
CO(g) |
H2(g) |
CH3OH(l) |
CH4(g) |
|
��H/(kJ/mol)
|
-283.0 |
-285.8 |
-726.5 |
-890.3 |
��ش��������⡣
��д����������CH3OH(l)��ȫȼ�յ��Ȼ�ѧ����ʽ�� ��
�Ƹ��ݸ�˹����������з�Ӧ���Ȼ�ѧ����ʽ��
CO(g)+ 2H2(g)= CH3OH(l)����H= ��
(3)����H2��CH4�Ļ������112 L����״������ʹ����ȫȼ������CO2(g)��H2O(l)�����ų�����3242.5 kJ����ԭ���������H2��CH4�����ʵ���֮���ǣ�
A��1��1 B��1��3 C��1��4 D��2��3
[s1]22��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
[s1] ��֪25�桢101kpaʱ��һЩ���ʵ�ȼ����Ϊ��
| ��ѧʽ | CO(g) | H2(g) | CH3OH(l) | CH4(g) |
| ��H/(kJ/mol)
| -283.0 | -285.8 | -726.5 | -890.3 |
��ش��������⡣
��д����������CH3OH(l)��ȫȼ�յ��Ȼ�ѧ����ʽ�� ��
�Ƹ��ݸ�˹����������з�Ӧ���Ȼ�ѧ����ʽ��
CO(g)+ 2H2(g)= CH3OH(l)����H= ��
(3)����H2��CH4�Ļ������112 L����״������ʹ����ȫȼ������CO2(g)��H2O(l)�����ų�����3242.5 kJ����ԭ���������H2��CH4�����ʵ���֮���ǣ�
A��1��1 B��1��3 C��1��4 D��2��3
[s1]22��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��9�·ݰ��⾫����2�� ���ͣ���ѡ��
[s1] ��֪��H2��g��+F2��g��=2HF��g����H=-270kJ/mol������˵����ȷ���� �� ��
A��2L����������ֽ��1L������1L��������270kJ����
B��1mol������1mol ������Ӧ����2mol Һ̬������ų�����С��270kJ
C������ͬ�����£�1mol ������1mol �����������ܺʹ���2mol���������������
D��1������������1���������ӷ�Ӧ����2��������������ӷų�270kJ������
[s1]24��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com