
����Ŀ��ijʵ��С����0.50 mol/L NaOH��Һ��0.50 mol/L������Һ�����к��ȵIJⶨ��
������0.50 mol/L NaOH��Һ
��ʵ���д�ԼҪʹ��245 mL NaOH��Һ��������Ҫ����NaOH����_____g��
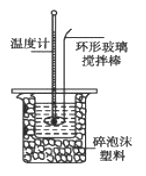
�ⶨϡ�����ϡ���������к��ȵ�ʵ��װ����ͼ��ʾ��
��1��д���÷�Ӧ���Ȼ�ѧ����ʽ���к���Ϊ57.3 kJ/mol����_____________��
��2��ȡ50 mL NaOH��Һ��30 mL������Һ����ʵ�飬ʵ���������±���
������д�±��еĿհף�
��ʼ�¶�t1/�� | ��ֹ�¶� t2/�� | �¶Ȳ�ƽ��ֵ ��t2-t1��/�� | |||
H2SO4 | NaOH | ƽ��ֵ | |||
1 | 26.2 | 26.0 | 26.1 | 30.1 | ______ |
2 | 27.0 | 27.4 | 27.2 | 33.3 | |
3 | 25.9 | 25.9 | 25.9 | 29.8 | |
4 | 26.4 | 26.2 | 26.3 | ||
�ڽ�����Ϊ0.50 mol/L NaOH��Һ��0.50 mol/L������Һ���ܶȶ���1 g/cm3���кͺ�������Һ�ı�����c=4.18 J/(g�����������к��ȡ�H=______________ȡС�����һλ����
������ʵ����ֵ�����57.3 kJ/mol��ƫ�����ƫ���ԭ������ǣ�______��
A��ʵ��װ�ñ��¡�����Ч����
B����ȡNaOH��Һ�����ʱ���Ӷ���
C���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
D�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
���𰸡�5.01/2H2SO4(aq����NaOH(aq����1/2Na2SO4(aq����H2O(l�� ��H����57.3kJ/mol4.0-53.5kJ/molacd
��������
���⿼��һ�����ʵ���Ũ����Һ���Ƶ��йؼ��㣬�Ȼ�ѧ����ʽ����д�Լ��к��ȵļ��㣬�������ȣ���Ŀ�ѶȲ���ע�������к��ȵĸ�������Ȼ�ѧ����ʽ����д�������Լ��ⶨ��Ӧ�ȵ��������⡣
I����1������ʵ������û��245mL����ƿ��Ӧѡ��250mL������ƿ������250mL����Һ��������������ƹ��������m=cVM=0.50mol/L��0.25L��40g/mol=5.0g��
II����1�������к��ȵĶ��壬ϡǿ�ᡢϡǿ�Ӧ����1molˮʱ�ų�57.3kJ��������ϡ�������������ϡ��Һ�ֱ���ǿ�ᡢǿ���÷�Ӧ���Ȼ�ѧ����ʽΪ1/2H2SO4(aq����NaOH(aq����1/2Na2SO4(aq����H2O(l�� ��H����57.3kJ/mol��
��2�������ݱ����ṩ����Ϣ����2���������ϴ���ȥ�����¶Ȳ�ƽ��ֵ=��4.0+3.9+4.1����3=4.0��C����50mL0.50mol/L����������30mL0.50mol/L������Һ�����кͷ�Ӧ����ˮ�����ʵ���Ϊ0.05L��0.50mol/L=0.025mol����Һ������Ϊ��80mL��1g/mL=80g���¶ȱ仯��ֵΪ��T=4����������0.025molˮ�ų�������ΪQ=mc��T=80g��4.18J/��g������4.0��=1337.6J����1.3376kJ������ʵ���õ��к�����H=-1.3376kJ��0.025mol=-53.5 kJ/mol��
��a��ʵ��װ�ñ��¡�����Ч������ã�����ᵼ������ɢʧ��ʹ�ⶨ���ƫС��a����ȷ��b����ȡNaOH��Һ�����ʱ���Ӷ������ᵼ�������������������ƫ�ų�������ƫ�ߣ������57.3kJ/mol��b�����c������һ�ο��ٽ�NaOH��Һ����ʢ�������С�ձ��У����ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ��У��ᵼ������ɢʧ��c��ȷ��d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�Ҫ���¶ȼƻ����ٲⶨH2SO4��Һ���¶ȣ�����������²��С��d����ȷ����ѡacd��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ��ʾװ���У��ס��ҡ��������ձ����ηֱ�ʢ��100 g 5.00%��NaOH��Һ��������CuSO4��Һ��100 g 10.00%��K2SO4��Һ���缫��Ϊʯī�缫��

(1)��ͨ��Դ������һ��ʱ���ñ���K2SO4Ũ��Ϊ10.47%������c�缫�������ӡ��ݴ˻ش����⣺
����Դ��N��Ϊ____________________����
���缫b�Ϸ����ĵ缫��ӦΪ____________________��
����ʽ����缫b�����ɵ������ڱ�״���µ������________________��
���缫c�������仯��__________g��
�����ǰ�����Һ���ᡢ���Դ�С�Ƿ����仯��������ԭ����Һ______________________________������Һ______________________________������Һ______________________________��
(2)�����������ͭȫ����������ʱ����ܷ�������У�Ϊʲô�� ________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ѧ������������أ������й�˵��������ǣ� ��
A.�����յķ����������ֲ�˿��������ά
B.ʳ���ͷ������Ȼ�����������������к�����
C.������ɱ�����в�������Ϊ���������ȱ���
D.ҽ�������ƾ����Ҵ���Ũ��Ϊ95%
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������֪O3����ΪV���νṹ��O3��ˮ�е��ܽ�Ⱥ�O2�Ƚ�
A.O3��ˮ�е��ܽ�Ⱥ�O2һ�� B.O3��ˮ�е��ܽ�ȱ�O2С
C.O3��ˮ�е��ܽ�ȱ�O2Ҫ�� D.û�취�Ƚ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ���¶��£����ڿ��淴ӦN2(g) +3H2(g) ![]() 2NH3(g)������Ӧ���ȣ�����������������˵����Ӧ�Ѵﻯѧƽ��״̬���ǣ� ��
2NH3(g)������Ӧ���ȣ�����������������˵����Ӧ�Ѵﻯѧƽ��״̬���ǣ� ��
A. ���������ڻ��������ܶȲ��ٱ仯
B. NH3������������H2����������֮��Ϊ2:3
C. ��ѹ�����ڻ������������ʵ������ٱ仯
D. ��λʱ���ڶ���a mol N��N����ͬʱ����6amol N-H��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������ʾ��ij���е���Ҫ������Ⱦ��ΪSO2��NOx��CO�ȣ�����Ҫ��ԴΪȼú��������β�������������о���
��1��Ϊ����ȼú��SO2���ŷţ��ɽ�úת��Ϊ���ȼ��ˮú����CO��H2����
��֪��![]() ��H��241.8kJ��mol��1��
��H��241.8kJ��mol��1��
![]() ��H����110.5kJmol��1
��H����110.5kJmol��1
д����̿��1molˮ������Ӧ����ˮú�����Ȼ�ѧ����ʽ��________��
��2������β����NO���ڷ��������������ɵģ���ӦΪN2��g����O2��g��![]() 2NO��g�� ��H��0��
2NO��g�� ��H��0��
������0.8molN2��0.2molO2�����ƿ�����ɣ��Ļ���������ij�ܱ������У�����1300����Ӧ�ﵽƽ�⣬�������8��10��4molNO��������¶��´˷�Ӧ�Ļ�ѧƽ�ⳣ��K��________������Ƽ���������
�������������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ��ԭ����________��
��3��������ͼ��ʾװ�ã��缫��Ϊ���Ե缫��������SO2�������������ų�����Һ����NO2��

���缫A�ĵ缫��ӦʽΪ________��
�缫B�ĵ缫��ӦʽΪ________��
�����������£��������ų�����Һ����NO2��ʹ��ת��Ϊ�����壬ͬʱ����SO32�����÷�Ӧ�����ӷ���ʽΪ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ����ģ��ϳ�������������£�

����ͼ��ѡ����ȡ����ĺ���װ�ã�

��1��װ��C������Ϊ_________________��ʵ����ͨ������װ��C�Ʊ�_________��
A��H2 B��H2S C��CO2 D��O2
��2��ʵ������װ��D�Ʊ�NH3�Ļ�ѧ����ʽΪ_______________________________��
��3������װ��B�Ʊ�SO2������ѡ���Լ�Ϊ___________��
A��Ũ���ᡢ�������ƹ��� B��Ũ���ᡢͭƬ
C��ϡ���ᡢ��������Һ D��Ũ���ᡢ��м
��4��SO2��O2ͨ����װ�ã���װ�õ����ó��˿��Կ���SO2��O2�������⣬������__________��

��5������һ������SO2(g)��O2(g)�ֱ�ͨ�뵽���Ϊ2L�ĺ����ܱ������У��ڲ�ͬ�¶��½��з�Ӧ�õ����±��е��������ݣ�
ʵ���� | �¶�/�� | ��ʼ��/mol | ƽ����/mol | ||
SO2 | O2 | SO2 | O2 | ||
1 | T1 | 4 | 2 | x | 0.8 |
2 | T2 | 4 | 2 | 0.4 | y |
����x=_______mol��y=_______mol��T1______T2���>������=����<��)
��6��SO2β������NaOH��Һ���գ��õ�Na2SO3��NaHSO3�����Ρ���һ�����ʵ�����SO2������������Һ��Ӧ��������Һ��Na2SO3��NaHSO3�����ʵ���֮��Ϊ2:3����μӷ�Ӧ��SO2��NaOH�����ʵ���֮��Ϊ________��
A. 5:7 B��1:2 C��9:4 D��9:13
��7�������백ˮ��Ӧ��������炙�������泥��ֳ�ȡ(NH4)2SO4��NH4HSO4�������Ʒ7.58 g�����뺬0.1 molNaOH����Һ��������Ӧ����OH-+H+��H2O, ��OH-+NH4+��NH3��+H2O����ַ�Ӧ������1792 mL����״��������֪�����ȷ�Ӧ������Ʒ��(NH4)2SO4�����ʵ���Ϊ_______mol��NH4HSO4�����ʵ���Ϊ_______mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������йص绯ѧԭ����˵���������ǣ� ��
A���Ȼ������۵���������ͣ���˹�ҵ����ò��õ�������Ȼ������Ʊ�������
B�����ʱ��ͨ���Ѵ��ƵĽ�����Ʒ���������ѶƲ����������
C������ұ�����ơ��ơ�þ�������������õĽ�������ⷨ������Ψһ���еĹ�ҵ����
D���Դ��ʹ�������ǽ��еġ���������������������������Ӧ����ԭ���ԭ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����и����Ȼ�ѧ����ʽ�У���ѧ��Ӧ�Ħ�Hǰ�ߴ��ں��ߵ���( )
��C(s)��O2(g)===CO2(g) ��H1
C(s)��![]() O2(g)===CO(g) ��H2
O2(g)===CO(g) ��H2
��S(s)��O2(g)===SO2(g) ��H3
S(g)��O2(g)===SO2(g) ��H4
��H2(g)��![]() O2(g)===H2O(l) ��H5
O2(g)===H2O(l) ��H5
2H2(g)��O2(g)===2H2O(l) ��H6
��CaCO3(s)===CaO(s)��CO2(g) ��H7
CaO(s)��H2O(l)===Ca(OH)2(s) ��H8
A���٢� B���٢� C���ڢۢ� D���٢ڢ�
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com