
 2N (g)
2N (g) 2H (g)
2H (g)
 ��3������һ��DZ�ڵ������Դ������������ȼ�ϵ�ص�ȼ�ϡ���ص��ܷ�ӦΪ��
��3������һ��DZ�ڵ������Դ������������ȼ�ϵ�ص�ȼ�ϡ���ص��ܷ�ӦΪ�� 2NH3(g)����H =" ��92.2" kJ��mol-1�� 391��2���� 200�棻�� AC���� 75%��3��2NH3��6e�� + 6OH��= N2 + 6H2O
2NH3(g)����H =" ��92.2" kJ��mol-1�� 391��2���� 200�棻�� AC���� 75%��3��2NH3��6e�� + 6OH��= N2 + 6H2O 2NH3��g������H=-92.2kJ/mol���ڷ�Ӧ�ʱ�=��Ӧ����ѻ�ѧ�����յ�����-�������γɻ�ѧ���ų�������=3��436+945.8-6��N-H������=-92.2��N-H������=391kJ��2���ϳɰ���ӦΪ���ȷ�Ӧ����Ӧ�¶�Խ��Խ�����ڷ�Ӧ�Ľ��У�����a��NH3�����ʵ���������ߣ��䷴Ӧ�¶�Ӧ�����ͣ���aΪ200���A����ʱ�����NH3����ʹƽ�������ƶ�����A��ȷ��B�������ܼӿ췴Ӧ���ʣ������ܸı�ת���ʣ���B����Cƽ�ⳣ��K���¶��йأ������������أ�ͬһ�¶��µ�ƽ�ⳣ����ȣ��ϳɰ���ӦΪ���ȷ�Ӧ�����¶�Խ��ƽ�ⳣ��ԽС����C��ȷ��ѡAC������M��NH3�����ʵ���Ϊ60%��ʣ�����ʵ���Ϊ40%����n(N2):n(H2)=1:3Ͷ�ϣ���ʣ��H2���ʵ���Ϊ30%����ΪN2��g��+3H2��g��
2NH3��g������H=-92.2kJ/mol���ڷ�Ӧ�ʱ�=��Ӧ����ѻ�ѧ�����յ�����-�������γɻ�ѧ���ų�������=3��436+945.8-6��N-H������=-92.2��N-H������=391kJ��2���ϳɰ���ӦΪ���ȷ�Ӧ����Ӧ�¶�Խ��Խ�����ڷ�Ӧ�Ľ��У�����a��NH3�����ʵ���������ߣ��䷴Ӧ�¶�Ӧ�����ͣ���aΪ200���A����ʱ�����NH3����ʹƽ�������ƶ�����A��ȷ��B�������ܼӿ췴Ӧ���ʣ������ܸı�ת���ʣ���B����Cƽ�ⳣ��K���¶��йأ������������أ�ͬһ�¶��µ�ƽ�ⳣ����ȣ��ϳɰ���ӦΪ���ȷ�Ӧ�����¶�Խ��ƽ�ⳣ��ԽС����C��ȷ��ѡAC������M��NH3�����ʵ���Ϊ60%��ʣ�����ʵ���Ϊ40%����n(N2):n(H2)=1:3Ͷ�ϣ���ʣ��H2���ʵ���Ϊ30%����ΪN2��g��+3H2��g�� 2NH3��g������������Ӧ��H2���ʵ���Ϊ90%����M���Ӧ��H2ת������
2NH3��g������������Ӧ��H2���ʵ���Ϊ90%����M���Ӧ��H2ת������ %=75%����3��ԭ����и���ʧȥ���ӣ����ϼ����ߣ�����������Ӧ����Ӧʽ2NH3��6e��+6OH��=N2+6H2O��
%=75%����3��ԭ����и���ʧȥ���ӣ����ϼ����ߣ�����������Ӧ����Ӧʽ2NH3��6e��+6OH��=N2+6H2O��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 O2(g)=H2O��1�� ��H2=-285��8 kJ/mol
O2(g)=H2O��1�� ��H2=-285��8 kJ/mol| A����H=+244��1kJ/mol | B����H=-488��3 kJ/mol |
| C����H=-996��6 kJ/mol | D����H=+996��6 kJ/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A��NaOH(aq)+HCl(aq)��NaCl(aq)+H20(l) =" +28.7" kJ��mol-1 =" +28.7" kJ��mol-1 |
B��NaOH(aq)+HCl(aq)��NaCl(aq)+H20(l) =" -28.7" kJ��mol-1 =" -28.7" kJ��mol-1 |
C��NaOH(aq)+HCl(aq)��NaCl(aq)+H20(l) =" +57.4" kJ��mol-1 =" +57.4" kJ��mol-1 |
D��NaOH(aq)+HCl(aq)��NaCl(aq)+H20(l) =" -57.4" kJ��mol-1 =" -57.4" kJ��mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
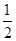 O2(g) ===CO(g)����H1 C(s)+O2(g) ===CO2(g)����H2
O2(g) ===CO(g)����H1 C(s)+O2(g) ===CO2(g)����H2| A���� | B���٢� | C���٢� | D���ڢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 [Cu��NH3��3]Ac?CO��aq������H��0��
[Cu��NH3��3]Ac?CO��aq������H��0�� CH3OH��g��+H2O��g�� ��H����49.0kJ/mol
CH3OH��g��+H2O��g�� ��H����49.0kJ/mol CH3OH��g�� ��H<0
CH3OH��g�� ��H<0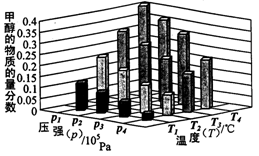
 CH3OCH3��g��+H2O��g�� ��H<0
CH3OCH3��g��+H2O��g�� ��H<0�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 2NH3 ��H ="-92.4" kJ/mol,��ʼ���ǽ�N2��H2�������20mol (�����1��1)����5L�ϳ�����.��ӦǰѹǿΪP0,��Ӧ������ѹǿ��P��ʾ����Ӧ������P/P0��ʱ��t�Ĺ�ϵ��ͼ��ʾ����ش��������⣺
2NH3 ��H ="-92.4" kJ/mol,��ʼ���ǽ�N2��H2�������20mol (�����1��1)����5L�ϳ�����.��ӦǰѹǿΪP0,��Ӧ������ѹǿ��P��ʾ����Ӧ������P/P0��ʱ��t�Ĺ�ϵ��ͼ��ʾ����ش��������⣺
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 2NO(g) ��H����֪�÷�Ӧ�� T ��ʱ��ƽ�ⳣ��K��9.0��
2NO(g) ��H����֪�÷�Ӧ�� T ��ʱ��ƽ�ⳣ��K��9.0�� 2NO2(g) ��H1 2NO2(g)
2NO2(g) ��H1 2NO2(g)  O2+2NO(g) ��H2 ��H= ���ú���H1����H2�ı���ʽ��ʾ����
O2+2NO(g) ��H2 ��H= ���ú���H1����H2�ı���ʽ��ʾ����| A������1 mol N2ͬʱ����1 mol O2 |
| B����������ܶȲ��� |
| C���������ƽ����Է����������� |
| D��2v��(N2)��v��(NO) |
 2NO(g)�ġ�K-T������c(NO)-t��ͼ����ͼA������֪�÷�ӦΪ ��Ӧ������ȡ����ȡ�������ͼB��֪����a��Ӧ��������ȣ�b�ı������������ ��
2NO(g)�ġ�K-T������c(NO)-t��ͼ����ͼA������֪�÷�ӦΪ ��Ӧ������ȡ����ȡ�������ͼB��֪����a��Ӧ��������ȣ�b�ı������������ ��
 2NO(g)________________(����ڻ�ѧƽ��״̬������������Ӧ������С������淴Ӧ������С�)��ƽ��ʱ��N2�ڻ�����������ٷ���Ϊ���٣����ڴ����д�����������̣��������2λ��Ч���֣�
2NO(g)________________(����ڻ�ѧƽ��״̬������������Ӧ������С������淴Ӧ������С�)��ƽ��ʱ��N2�ڻ�����������ٷ���Ϊ���٣����ڴ����д�����������̣��������2λ��Ч���֣��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com