
(1)����������ѧ��ѧʵ���г��õ�����������ʵ������У�һ�㲻��Ҫ�ò���������________(��д���)��
����pH��ֽ�ⶨNa2CO3��Һ��pH
������һ�����ʵ���Ũ�ȵ��Ȼ�����Һ
�۽������Ȼ���������Һ�����ˮ���Ʊ�������������
��̽��Ba(OH)2��8H2O�����NH4Cl���巴Ӧ�����е������仯
��ʵ���������Ƶ�FeSO4��Һ��Ԥ��������NaOH��Һ�Ʊ�Fe(OH)2��ɫ����
(2)�á����ڡ�����С�ڡ����ڡ���գ�
�ٶ�ȡ��Ͳ��Һ������ʱ������ƫ�ߣ���ȡ�������________ʵ���������
����������ƽ��ȡ10.4 gʳ�Σ��������ʳ�ε�λ�õߵ�������ȡʳ�ε�����_____10.4 g��
������500 mL 0.1 mol/L NaOH��Һ������ʱ���ӿ̶��ߣ�������Һ�����ʵ���Ũ�� _______0.1 mol/L��
�����к͵ζ�����ijNaOH��Һ��Ũ�ȣ���ȡ����Һʱδ�ø���Һ��ϴ�ζ��ܣ���õ���ҺŨ��________ʵ��Ũ�ȡ�
(1) �ۢ� (2)�ٴ��� ��С�� �۴��� ��С��
��������
�����������1���������ǻ�ѧʵ���г��õ������������������ڽ��衢���˻�ת��Һ��ʱ���������ݸ�ʵ���ʵ��Ŀ��ȷ���Ƿ���Ҫ��������
��2���ٸ�����Ͳ�Ķ������µ�����ֵ���������ݸ���ʱ�Ķ�����Һ��Ĺ�ϵ���з�����
�ڸ�����ƽ�ij���ԭ��������=����+����Ķ�����������
�۸��ݸ���ʱ�Ŀ̶�����Һ��Ĺ�ϵ���з�������Ĵ�С���ٸ���c��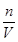 �������������ʵ����ʵ��������Һ�������Ӱ���жϣ����nƫ���VƫС������������ҺŨ��ƫ�ߣ�
�������������ʵ����ʵ��������Һ�������Ӱ���жϣ����nƫ���VƫС������������ҺŨ��ƫ�ߣ�
�ܸ�����ȡ����Һʱĩ�ø���Һ��ϴ�ζ��ܣ�����ҺʱŨ�Ƚ���С��һ���������Һ�����ʵ�������С�����ı�Һ�����ƫС������Һ��Ũ��ƫС��
��1������pH��ֽ�ⶨNa2CO3��Һ��pH���ò�����պȡ��������Һ����ֽ���в����ʢٴ���
������һ�����ʵ���Ũ�ȵ��Ȼ�����Һʱ���ܽ���Ҫ���������裬ת����Ҫ�������������ʢڴ���
�۽������Ȼ���������Һ�����ˮ���Ʊ������������壬����ֱ��Һ���Ϊ���ɫֹͣ���ȣ����貣�������裬�ʢ���ȷ��
��̽��Ba��OH��2•8H2O�����NH4Cl���巴Ӧ�����е������仯���ò���������������Ļ����ʢܴ���
��ʵ���������Ƶ�FeSO4��Һ��Ԥ��������NaOH��Һ�Ʊ�Fe��OH��2��ɫ������������NaOH��Һ�ĵιܲ���Һ�����£����貣�������ʢ���ȷ����ѡ�ۢݡ�
��2��������Ͳ�Ķ������µ�����ֵ��������ʱ�Ķ�����Һ��ߣ����Զ�ȡ�����������ʵ���������
������ƽ�ij���ԭ�������̣�����+����Ķ�������������ƽ��ȡ10.4gʳ�Σ��������ʳ�ε�λ�õߵ����Ƶõ�����Ϊ9.6gʳ�Σ�
������ʱ�Ŀ̶��߱�Һ��ߣ���Һ�����ƫС��Ũ��ƫ��
������ȡ����Һʱĩ�ø���Һ��ϴ�ζ��ܣ����Դ���ҺʱŨ�Ƚ���С��һ���������Һ�����ʵ�������С�����ĵı�Һ���ƫС����õ���ҺŨ��С��ʵ��Ũ�ȡ�
���㣺���黯ѧʵ�����������������
����������ʱ�߿��еij������ͣ������е��Ѷȵ����⣬���������ǿ�����ض�ѧ������֪ʶ�Ĺ��̺ͼ��飬����������ѧ���淶�Ͻ���ʵ����������Ͷ��ֲ�����������ѧʵ�����������ƽʱ��ѧϰ��ע����ۺ��ܽᣬ����������á�


 ͨ��ѧ��Ĭд����ϵ�д�
ͨ��ѧ��Ĭд����ϵ�д� ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ����С�ձ��ڼ����������ữ��ϡKI������Һ����С�ձ����ڱ����·���
����С�ձ��ڼ����������ữ��ϡKI������Һ����С�ձ����ڱ����·����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��A��B��C��D��E��ƿ��Һ�����Ƕ�����ѧ��ѧ�г��õ����Լ���B,C,D������������Һ���ֽ�������ʵ�飺
��A�д̼�����ζ����պ��Ũ����IJ������ӽ�Aʱ������ɫ������
��A�ֱ����������Һ�У�ֻ��B��C���г������ɣ��ҳ�����ɫ�ֱ�Ϊ���ɫ�Ͱ�ɫ�����а�ɫ��������������������Ҳ���������������ơ�
��պ��A�IJ�������պ��E��Ũ��Һ�IJ������ӽ�ʱҲ������ɫ��������E�����¸���������Щ�����Ӵ��ᷢ���ۻ�����
��D��Һ������ɫ��Ӧʱ����Ϊ��ɫ��
��������ʵ����Ϣ����ش��������⣺
��1����д��������Һ���ʵĻ�ѧʽ��A D E
��2����д��A��C��Ӧ�����ӷ���ʽ
��3����д������B��Һ�����ӷ���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��A��B��C��D��E��ƿ��Һ�����Ƕ�����ѧ��ѧ�г��õ����Լ���B,C,D������������Һ���ֽ�������ʵ�飺
��A�д̼�����ζ����պ��Ũ����IJ������ӽ�Aʱ������ɫ������
��A�ֱ����������Һ�У�ֻ��B��C���г������ɣ��ҳ�����ɫ�ֱ�Ϊ���ɫ�Ͱ�ɫ�����а�ɫ��������������������Ҳ���������������ơ�
��պ��A�IJ�������պ��E��Ũ��Һ�IJ������ӽ�ʱҲ������ɫ��������E�����¸���������Щ�����Ӵ��ᷢ���ۻ�����
��D��Һ������ɫ��Ӧʱ����Ϊ��ɫ��
��������ʵ����Ϣ����ش��������⣺
��1����д��������Һ���ʵĻ�ѧʽ��A D E
��2����д��A��C��Ӧ�����ӷ���ʽ
��3����д������B��Һ�����ӷ���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ��㶫ʡ÷�ض�ɽ��ѧ��һ��ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ������
��A��B��C��D��E��ƿ��Һ�����Ƕ�����ѧ��ѧ�г��õ����Լ���B,C,D������������Һ���ֽ�������ʵ�飺
��A�д̼�����ζ����պ��Ũ����IJ������ӽ�Aʱ������ɫ������
��A�ֱ����������Һ�У�ֻ��B��C���г������ɣ��ҳ�����ɫ�ֱ�Ϊ���ɫ�Ͱ�ɫ�����а�ɫ��������������������Ҳ���������������ơ�
��պ��A�IJ�������պ��E��Ũ��Һ�IJ������ӽ�ʱҲ������ɫ��������E�����¸���������Щ�����Ӵ��ᷢ���ۻ�����
��D��Һ������ɫ��Ӧʱ����Ϊ��ɫ��
��������ʵ����Ϣ����ش��������⣺
��1����д��������Һ���ʵĻ�ѧʽ��A D E
��2����д��A��C��Ӧ�����ӷ���ʽ
��3����д������B��Һ�����ӷ���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��㶫ʡ��һ��ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ������
��A��B��C��D��E��ƿ��Һ�����Ƕ�����ѧ��ѧ�г��õ����Լ���B,C,D������������Һ���ֽ�������ʵ�飺
��A�д̼�����ζ����պ��Ũ����IJ������ӽ�Aʱ������ɫ������
��A�ֱ����������Һ�У�ֻ��B��C���г������ɣ��ҳ�����ɫ�ֱ�Ϊ���ɫ�Ͱ�ɫ�����а�ɫ��������������������Ҳ���������������ơ�
��պ��A�IJ�������պ��E��Ũ��Һ�IJ������ӽ�ʱҲ������ɫ��������E�����¸���������Щ�����Ӵ��ᷢ���ۻ�����
��D��Һ������ɫ��Ӧʱ����Ϊ��ɫ��
��������ʵ����Ϣ����ش��������⣺
��1����д��������Һ���ʵĻ�ѧʽ��A D E
��2����д��A��C��Ӧ�����ӷ���ʽ
��3����д������B��Һ�����ӷ���������
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com