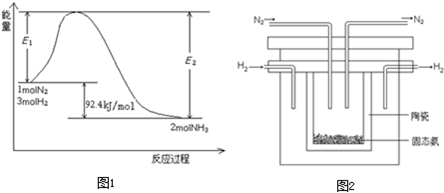
��2011?��ͷ����ģ�⣩��֪A��B��C����ѧ��ѧ�ij������ʣ�������һ��������������ת����ϵ��A
B
C
��1����A��ʹʪ��ĺ�ɫʯ����ֽ������CΪ����ɫ���壮��Aת��ΪB��Ӧ�Ļ�ѧ����ʽΪ
��
��2����D�Ǿ��������Եĵ��ʣ�AԪ�����ڶ������������Ԫ�أ���C�ĵ���ʽΪ
��
��3����D�ǽ�����C��Һ������ʱӦ��������D���������ǣ��ñ�Ҫ�����ֺ����ӷ���ʽ˵����
��������������ֹFe2+������ΪFe3+��2Fe3++Fe�T3Fe2+
��������������ֹFe2+������ΪFe3+��2Fe3++Fe�T3Fe2+
��
��4����D��һ�ֳ������������壻A��һ��ǿ���������ˮ��Һ�е���������������Ӿ�����10�����ӣ���Bת��ΪC�����ӷ���ʽΪ
CO2+CO32-+H2O=2HCO3-
CO2+CO32-+H2O=2HCO3-
��
��5����DΪ�ȼҵ����Ҫ��Ʒ��B�������ԣ���C��Һ�г�����������ڵ������ӵĻ�ѧʽΪ
AlO2-
AlO2-
��
��6����A��B��C��Ϊ�����D��һ�ֺ�ɫ��̬�ǽ������ʣ���B���ӵĽṹʽΪ
O=C=O
O=C=O
��
��7����A���������Ϊ75%����Һ������ҽ����������B������������ͭ���ȣ���ש��ɫ�������ɣ���A����B�Ļ�ѧ����ʽΪ
��



 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��


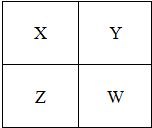 ��2011?��ͷ����ģ�⣩��֪X��Y��Z��W���ֶ�����Ԫ�������ڱ��е����λ����ͼ��ʾ������˵����ȷ���ǣ�������
��2011?��ͷ����ģ�⣩��֪X��Y��Z��W���ֶ�����Ԫ�������ڱ��е����λ����ͼ��ʾ������˵����ȷ���ǣ�������