
Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺
![]() ʵ��һ����ʢ�б�����ˮ����ƿ�м������ʯ����������۲쵽��ˮ�Ļ���ɫ��ȥ��ͬʱ�����������塣
ʵ��һ����ʢ�б�����ˮ����ƿ�м������ʯ����������۲쵽��ˮ�Ļ���ɫ��ȥ��ͬʱ�����������塣
![]() ʵ�����ȡ����ʵ��һ����ƿ����Һ����������ʵ�飺
ʵ�����ȡ����ʵ��һ����ƿ����Һ����������ʵ�飺
![]() ��1������Һ������ɫ�������м�ǿ��Ư�����á���2������Һ�еμ�̼������Һ������ɫ��������3������Һ�еμ�����ɲ����������塣��4������Һ���Ⱥ����Dz������������塣
��1������Һ������ɫ�������м�ǿ��Ư�����á���2������Һ�еμ�̼������Һ������ɫ��������3������Һ�еμ�����ɲ����������塣��4������Һ���Ⱥ����Dz������������塣
![]()
 ʵ��������ѧ������������װ���ռ�ʵ����в�������������һ�����顣
ʵ��������ѧ������������װ���ռ�ʵ����в�������������һ�����顣
![]() �ش��������⣺
�ش��������⣺
![]() ��1����ʵ����У���Һ������ɫ�������м�ǿ��Ư������˵������Һ�к��� ��д��ѧʽ������Һ�еμ�̼������Һ������ɫ����˵������Һ�к��� ���ӣ���Һ�еμ���������������� ����Һ���Ⱥ�������ǵ�ԭ������Ϊ������ ���ʡ�
��1����ʵ����У���Һ������ɫ�������м�ǿ��Ư������˵������Һ�к��� ��д��ѧʽ������Һ�еμ�̼������Һ������ɫ����˵������Һ�к��� ���ӣ���Һ�еμ���������������� ����Һ���Ⱥ�������ǵ�ԭ������Ϊ������ ���ʡ�
![]() ��2��ʵ�����У�ϴ��װ���ڵ�ϴ��Һһ���� ��Һ��
��2��ʵ�����У�ϴ��װ���ڵ�ϴ��Һһ���� ��Һ��
![]() ��3���ڷ�Ӧ�������ͬ����£���ѧ�������ü���װ��A�ͼ���װ��B�ֱ��ϴ��װ���������ռ����塣ѡ�ü���װ�� ��ѡ�A����B�������ռ�����������塣�����飬��ѧ������ѡ�ü���װ��B������һ��ʵ�顣��ѡ��װ��B��ԭ����
��3���ڷ�Ӧ�������ͬ����£���ѧ�������ü���װ��A�ͼ���װ��B�ֱ��ϴ��װ���������ռ����塣ѡ�ü���װ�� ��ѡ�A����B�������ռ�����������塣�����飬��ѧ������ѡ�ü���װ��B������һ��ʵ�顣��ѡ��װ��B��ԭ����
![]() ��
��
![]() ��4����ѧ��ͨ������ʵ�飬д����ˮ��̼��Ʒ�Ӧ�Ļ�ѧ����ʽΪ ��
��4����ѧ��ͨ������ʵ�飬д����ˮ��̼��Ʒ�Ӧ�Ļ�ѧ����ʽΪ ��
��1��HClO Ca2+ CO2 CaCO3 ��2������NaHCO3
![]() ��3��A ѡ��Bװ�ò�����Ϊ��㡣��ѡ��Aװ�ý������Ƴ��Թ�ʱ������ɴ������������Ҳ���Ҳ�����㡣��4��2CaCO3+Cl2+H2O
��3��A ѡ��Bװ�ò�����Ϊ��㡣��ѡ��Aװ�ý������Ƴ��Թ�ʱ������ɴ������������Ҳ���Ҳ�����㡣��4��2CaCO3+Cl2+H2O![]() Ca(HCO3)2+2HClO+CaCl2
Ca(HCO3)2+2HClO+CaCl2
�����ܽ���ˮ��������Ӧ��C12+H2O![]() HCl+HClO�����ɵ�������CaCO3������Ӧ��2 H++CaCO3
HCl+HClO�����ɵ�������CaCO3������Ӧ��2 H++CaCO3![]() Ca2++CO2��+ H2O����ʵ��һû�й۲쵽��ƿ����Һ����ǵ����������������壬��CaCO3+CO2+H2O
Ca2++CO2��+ H2O����ʵ��һû�й۲쵽��ƿ����Һ����ǵ����������������壬��CaCO3+CO2+H2O![]() Ca(HCO3)2��
Ca(HCO3)2��


 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺
Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺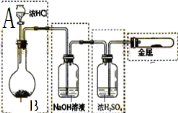
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��ɽ��ʡ̩�����������и�����һ���¿���ѧ�Ծ����������� ���ͣ�ʵ����
(6��)Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺
ʵ��һ����ʢ�б�����ˮ����ƿ�м������ʯ����������۲쵽��ˮ�Ļ���ɫ��ȥ��ͬʱ�����������塣
ʵ�����ȡ����ʵ��һ����ƿ����Һ����������ʵ�飺
�ٸ���Һ������ɫ�������м�ǿ��Ư�����á��ڸ���Һ�еμ�̼������Һ������ɫ�������۸���Һ�еμ�����ɲ����������塣�ܸ���Һ���Ⱥ����Dz������������塣
�ش��������⣺
(1)��ʵ����У���Һ������ɫ�������м�ǿ��Ư������˵������Һ�к���_________(д��ѧʽ)����Һ�еμ�̼������Һ������ɫ����˵������Һ�к���______���ӣ���Һ�еμ����������������________����Һ���Ⱥ�������ǵ�ԭ������Ϊ������________��
(2)��ѧ��ͨ������ʵ�飬д����ˮ��̼��Ʒ�Ӧ�Ļ�ѧ����ʽΪ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡ̩���и�����һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺
ʵ��һ����ʢ�б�����ˮ����ƿ�м������ʯ����������۲쵽��ˮ�Ļ���ɫ��ȥ��ͬʱ�����������塣
ʵ�����ȡ����ʵ��һ����ƿ����Һ����������ʵ�飺
�ٸ���Һ������ɫ�������м�ǿ��Ư�����á��ڸ���Һ�еμ�̼������Һ������ɫ�������۸���Һ�еμ�����ɲ����������塣�ܸ���Һ���Ⱥ����Dz������������塣
�ش��������⣺
(1)��ʵ����У���Һ������ɫ�������м�ǿ��Ư������˵������Һ�к���______________(д��ѧʽ)����Һ�еμ�̼������Һ������ɫ����˵������Һ�к���______���ӣ���Һ�еμ����������������________����Һ���Ⱥ�������ǵ�ԭ������Ϊ������________��
(2)��ѧ��ͨ������ʵ�飬д����ˮ��̼��Ʒ�Ӧ�Ļ�ѧ����ʽΪ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡ̩���и�����һ���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
(6��)Ϊ���о���ˮ�ܷ��̼��Ʒ�����ѧ��Ӧ��ijѧ���������ʵ�飺
ʵ��һ����ʢ�б�����ˮ����ƿ�м������ʯ����������۲쵽��ˮ�Ļ���ɫ��ȥ��ͬʱ�����������塣
ʵ�����ȡ����ʵ��һ����ƿ����Һ����������ʵ�飺
�ٸ���Һ������ɫ�������м�ǿ��Ư�����á��ڸ���Һ�еμ�̼������Һ������ɫ�������۸���Һ�еμ�����ɲ����������塣�ܸ���Һ���Ⱥ����Dz������������塣
�ش��������⣺
(1)��ʵ����У���Һ������ɫ�������м�ǿ��Ư������˵������Һ�к���_________(д��ѧʽ)����Һ�еμ�̼������Һ������ɫ����˵������Һ�к���______���ӣ���Һ�еμ����������������________����Һ���Ⱥ�������ǵ�ԭ������Ϊ������________��
(2)��ѧ��ͨ������ʵ�飬д����ˮ��̼��Ʒ�Ӧ�Ļ�ѧ����ʽΪ��
��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com