
 Al2O3+6Cu��
Al2O3+6Cu�� Al2O3+6Cu��
Al2O3+6Cu��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�꽭��ʡ�����ѧ�߶�10�½���ϰ��ѧ�Ծ����������� ���ͣ������
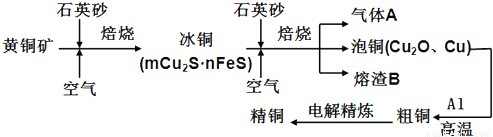
��12�֣���ҵ���ɻ�ͭ����Ҫ�ɷ�CuFeS2��ұ��ͭ����Ҫ�������£�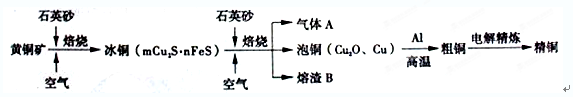
������A�еĴ�����Ⱦ���ѡ�������Լ��е�____________���ա�
a��ŨH2SO4 b��ϡHNO3 c��NaOH��Һ d����ˮ
����ϡH2SO4 ��������B��ȡ����������Һ��������Һ�л�����Fe3���ķ�����_______________________��ע���Լ�������
������ͭұ����ͭ�Ļ�ѧ��Ӧ����ʽΪ__________________________________��
����CuSO4��ҺΪ�������Һ���д�ͭ����Al��Zn��Ag��Pt��Au�����ʣ��ĵ�⾫��������˵����ȷ����____________��
a���������������仯��� b���������Һ����ɲ������仯
c����Һ��Cu2���������ƶ� d������������ɻ���Ag��Pt��Au�Ƚ���
�����÷�Ӧ2Cu��O2��2H2SO4��2CuSO4��2H2O���Ʊ�CuSO4�������÷�Ӧ���Ϊԭ��أ��������缫��ӦʽΪ________________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com